Clear Sky Science · en
Glioblastoma cells that evade chemoradiotherapy-induced cell death exhibit a bifurcated glycolytic program
Why this brain cancer study matters
Glioblastoma is one of the deadliest brain cancers in adults, and almost always grows back after surgery, chemotherapy, and radiation. This study asks a crucial question: what is special about the small group of tumor cells that survive treatment and later rekindle the disease? By tracking how these cells handle sugar over time, the researchers uncover a hidden survival strategy that may explain why current treatments so often fail—and suggest new ways to cut off the tumor’s lifeline.
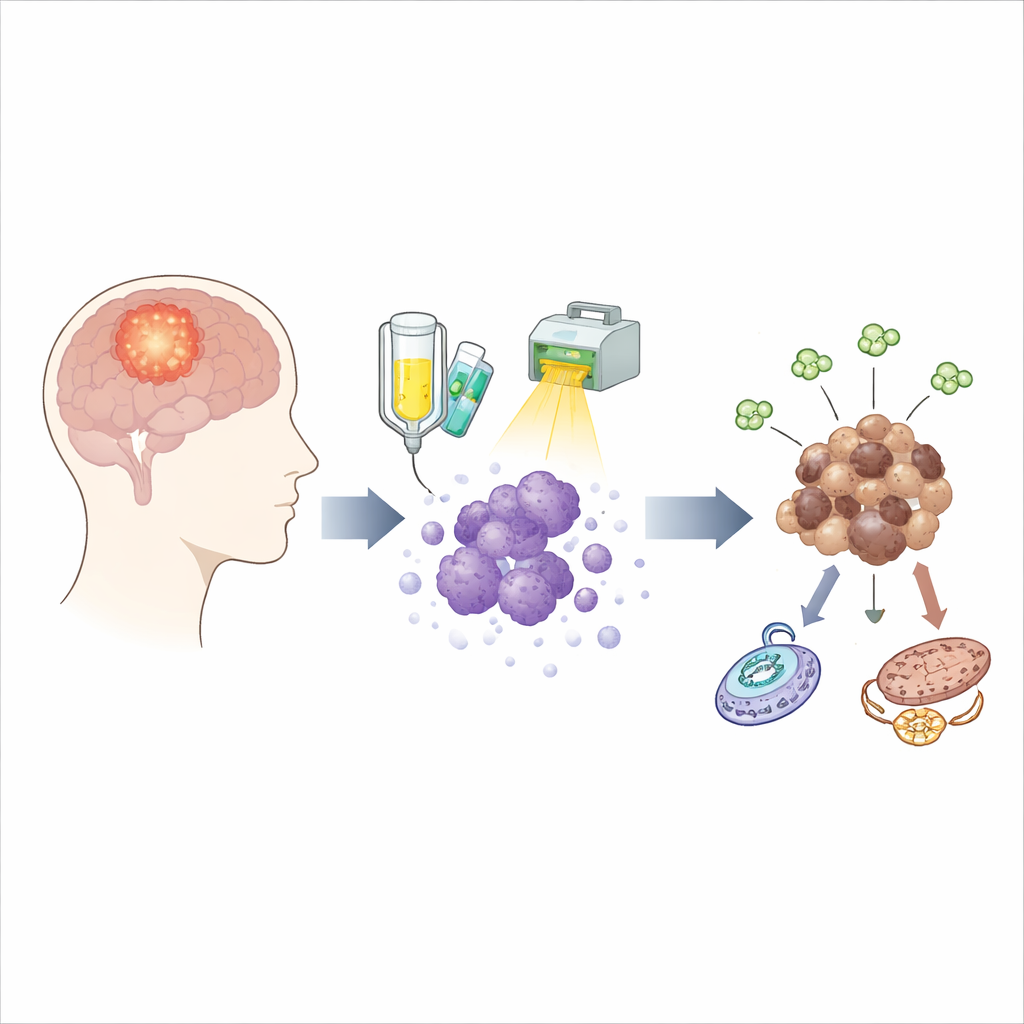
The hidden survivors after treatment
Standard care for glioblastoma combines a drug called temozolomide with radiation. While this combo kills the majority of tumor cells, a stubborn minority survives and can seed a new tumor months later. To catch these survivors in the act, the team used tumor cells taken directly from ten different patients and exposed them to a treatment schedule designed to closely mimic what people receive in the clinic. They sampled living cells at several points during and after treatment, and also studied matching tumors grown in mice, allowing them to follow how survivors change over time rather than just comparing tumors before and after relapse.
How tumor cells change the way they use sugar
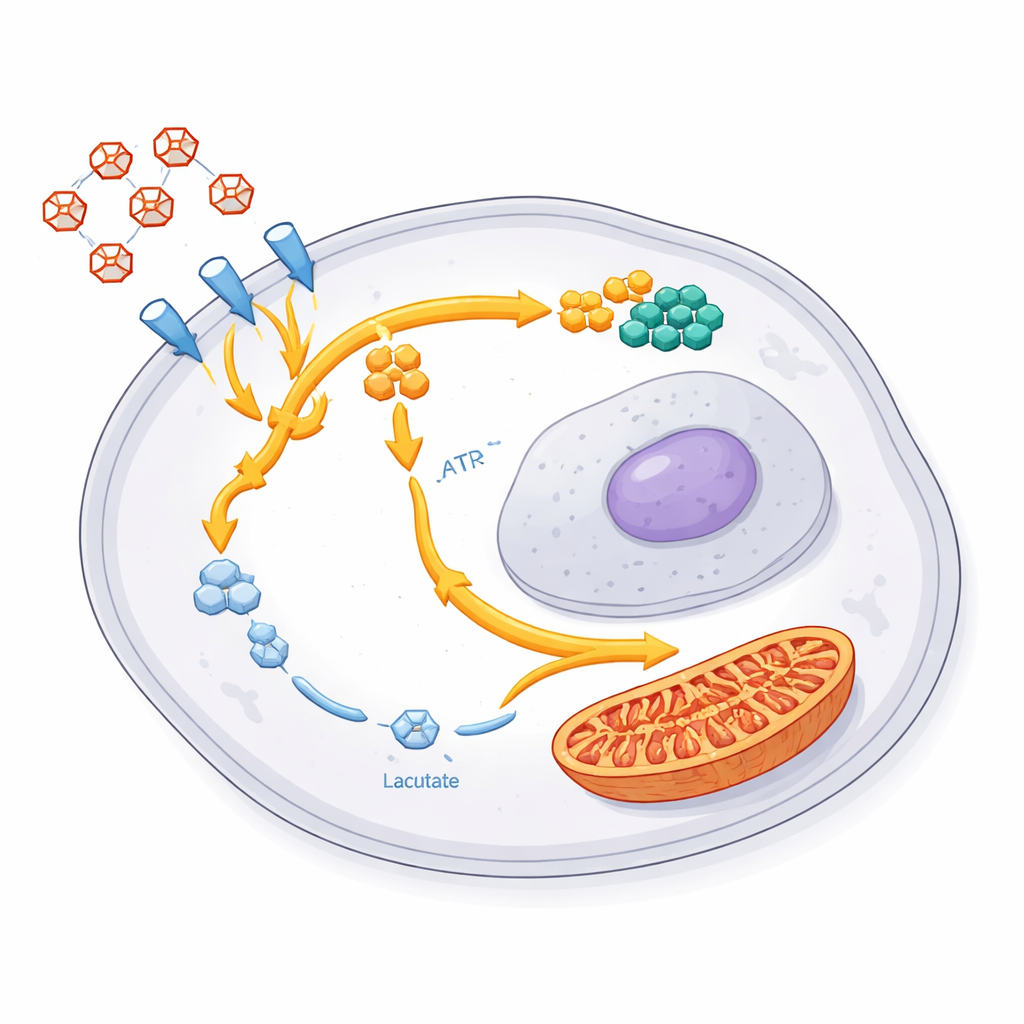
Brain tissue consumes a remarkable share of the body’s sugar, and glioblastoma cells are especially hungry for it. The researchers found that, as treatment progressed, the surviving cells pulled in even more glucose from their surroundings. Yet, in a twist, they did not simply burn this extra fuel faster. Instead, these cells reduced their production of lactate, the typical waste product of fast sugar breakdown in cancer. Measurements of key proteins showed a striking split pattern: components that bring sugar into the cell and start breaking it down ramped up, while those that handle the later steps and make lactate dropped. This “bifurcated” program means sugar enters and begins to be processed, but the usual exit route to lactate is partly shut.
Rerouting fuel into building blocks and power plants
Where does the diverted sugar go? Using specially labeled glucose and advanced metabolite measurements, the team showed that surviving cells shunt much of this carbon into two main destinations. One is a side route called the pentose phosphate pathway, which generates the raw materials needed to make DNA and RNA building blocks. In these cells, many nucleotide components and related genes were boosted, suggesting a strong push toward rebuilding and preparing for renewed growth. The second destination is the cell’s power stations, the mitochondria. Enzymes of the mitochondrial energy cycle increased, and earlier work from the same group showed greater import of sugar-derived fuel into these structures. Together, this reveals a coordinated rewiring: less wasteful fermentation to lactate, more investment in repair, growth materials, and flexible energy production.
A shared survival playbook across tumors and in animals
Glioblastoma tumors differ greatly from patient to patient, which often foils one-size-fits-all treatments. Despite this diversity, every one of the ten patient-derived models showed the same basic shift after chemoradiotherapy: increased early sugar-handling steps, decreased late steps and lactate output, and higher mitochondrial activity. Importantly, the same signature appeared in mouse brain tumors grown from patient cells and treated with adapted versions of standard therapy. Recurrent tumors in these animals still carried the altered sugar program, indicating that this is not a fleeting effect but a stable trait of cells that manage to outlive treatment and rebuild the cancer.
What this means for future treatment
To a layperson, the central message is that surviving glioblastoma cells do not simply “hide” from treatment; they actively rewire how they use sugar to stay alive. They draw in more glucose, avoid turning it into waste, and instead channel it into making DNA building blocks and powering mitochondria—helping them repair damage, maintain a flexible, stem-like state, and eventually regrow the tumor. By revealing this shared metabolic survival state, the study points to new therapeutic ideas: targeting the early sugar-processing steps, the DNA-building side route, or the mitochondrial reliance of these cells could make standard chemoradiotherapy more complete, leaving fewer escapees behind to restart the disease.
Citation: Martell, E., Kuzmychova, H., Chawla, U. et al. Glioblastoma cells that evade chemoradiotherapy-induced cell death exhibit a bifurcated glycolytic program. Cell Death Dis 17, 348 (2026). https://doi.org/10.1038/s41419-026-08646-9
Keywords: glioblastoma, cancer metabolism, glucose utilization, therapy resistance, pentose phosphate pathway