Clear Sky Science · en
CAFs-derived LAM332 promotes CTCs formation and survival via ITGA3 and contributes to the metastasis of pancreatic ductal adenocarcinoma
Why this research matters for patients
Pancreatic cancer is notorious for spreading early and quietly, often leaving patients with few treatment options by the time it is found. This study digs into one of the most dangerous steps in that spread: how cancer cells break away from the original tumor, survive in the bloodstream, and seed new tumors in organs like the liver and lungs. By uncovering a specific "conversation" between tumor cells and their surrounding support cells, the authors point to new ways to predict, and potentially block, deadly metastasis.
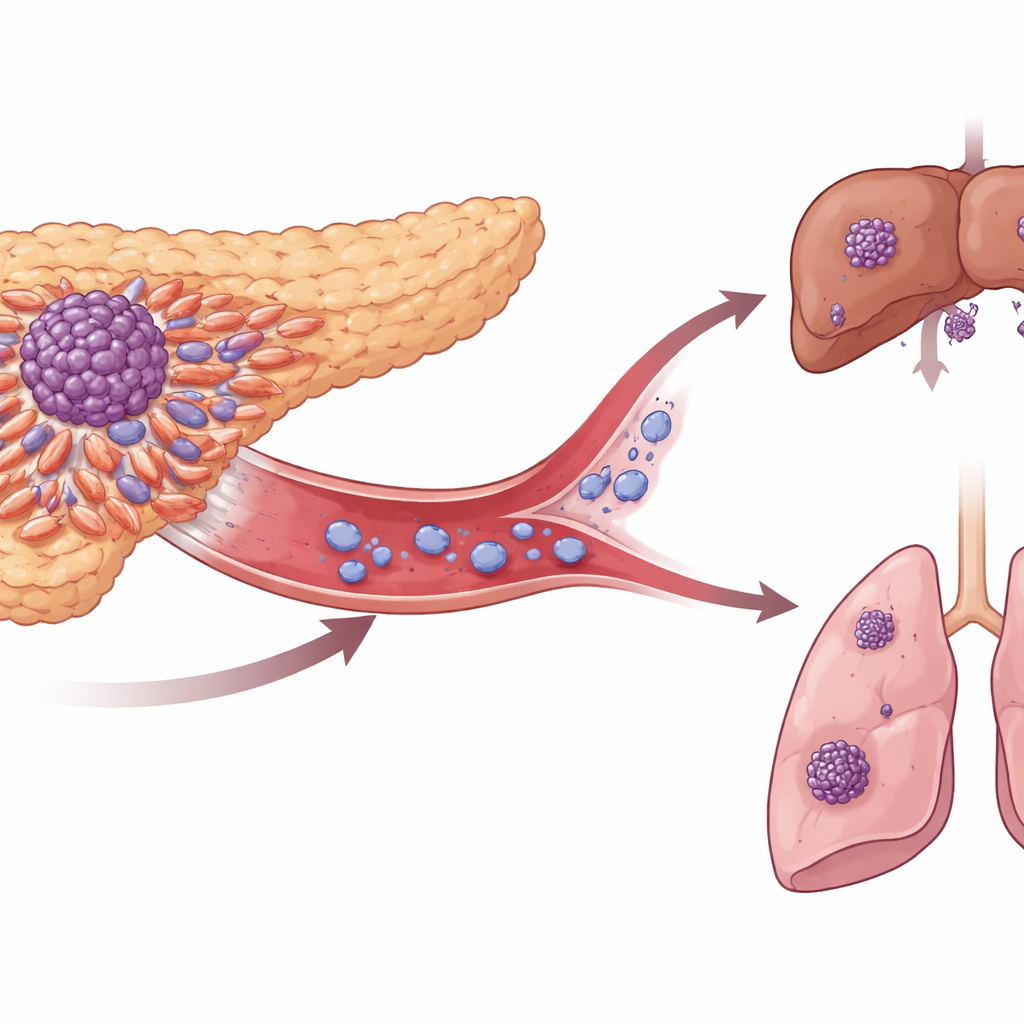
Hidden travelers in the blood
When pancreatic tumors spread, they do so through circulating tumor cells—rogue cells that escape into the blood. The team analyzed tumor samples and large genetic and protein datasets from patients with and without such cells in their vessels, and from those with localized versus metastatic disease. They repeatedly found one surface molecule, called ITGA3, standing out. Tumors and circulating cells with higher ITGA3 were more common in advanced cancers, especially at the invasive rim of the tumor where cells first break away. Patients whose tumors had more ITGA3 tended to have more circulating tumor cells, more aggressive features such as invasion into nearby tissues and blood vessels, and shorter survival.
How tumor cells gain the ability to break free
To understand what ITGA3 actually does, the researchers manipulated its levels in pancreatic cancer cell lines and implanted these cells in mice. When ITGA3 was reduced, cancer cells grew more slowly, moved less, and were less able to chew through surrounding tissue. The cells also shifted back toward a more orderly, "epithelial" state, regaining tight cell–cell contacts and losing the flexible, migratory traits that help them escape. In mice, tumors lacking ITGA3 were smaller and showed fewer signs of invasive behavior. These findings suggest that ITGA3 acts as a key switch that helps tumor cells loosen from the main mass, enter blood vessels, and begin their journey as circulating tumor cells.
Clumping together to survive the journey
Life in the bloodstream is harsh for solitary cancer cells, but clusters of tumor cells appear to survive and seed metastases far better. The study found that ITGA3 is especially abundant on cells within such clusters. In lab dishes designed to prevent cells from sticking to plastic, cancer cells with normal ITGA3 readily formed tight clusters and resisted cell death, whereas cells with ITGA3 turned off stayed mostly separate and died more easily. In mouse models where tumors grew in the pancreas, ITGA3 loss led to fewer circulating clusters, fewer metastatic colonies in the liver and lungs, and a lower overall burden of distant disease. This shows that ITGA3 not only helps cells detach, but also helps them stick to each other and endure the blood’s hostile conditions.
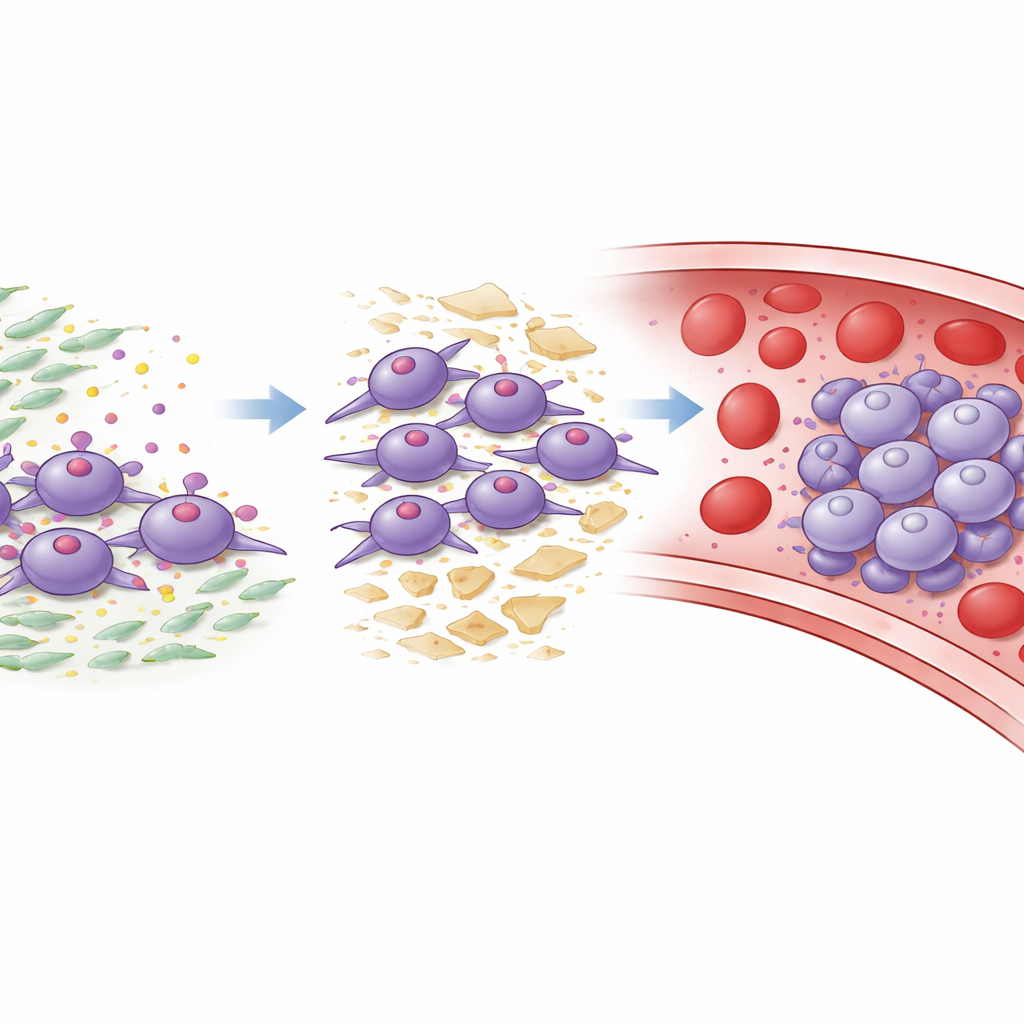
The role of supportive stromal cells
Pancreatic tumors are packed with cancer-associated fibroblasts, a type of support cell that builds and remodels the tissue scaffold around the tumor. The authors discovered that these fibroblasts are a major source of a sticky protein called laminin-332. Tumor cells bearing ITGA3 use it as a docking point for laminin-332. In cell and animal experiments, laminin-332 binding to ITGA3 triggered signals inside tumor cells that boosted growth, movement, tissue invasion, and the shift toward a more mobile, aggressive state. It also promoted clustering and reduced cell death when cells were in suspension, mimicking conditions in the bloodstream. Blocking laminin-332 with antibodies, or interfering with ITGA3 or its downstream signaling partners, largely reversed these malignant behaviors and cut down on metastases in mice.
What this could mean for future treatments
Taken together, the work outlines a clear chain of events: fibroblasts in and around pancreatic tumors release laminin-332; this protein latches onto ITGA3 on tumor cells; the signal that follows helps cells detach, clump into protective clusters, survive in the circulation, and colonize distant organs. For a layperson, the key message is that the researchers have mapped a specific "supply line" that tumors use to launch and protect their spreading cells. Because this pathway depends on components found mainly in the tumor and its stroma, it offers an attractive target for new therapies. Drugs or antibodies that block laminin-332, ITGA3, or the signals they trigger could help reduce the number and resilience of circulating tumor cells, potentially slowing or preventing deadly metastasis in pancreatic cancer.
Citation: Tang, H., Shi, W., Tan, S. et al. CAFs-derived LAM332 promotes CTCs formation and survival via ITGA3 and contributes to the metastasis of pancreatic ductal adenocarcinoma. Cell Death Dis 17, 369 (2026). https://doi.org/10.1038/s41419-026-08642-z
Keywords: pancreatic cancer, metastasis, circulating tumor cells, tumor microenvironment, cancer-associated fibroblasts