Clear Sky Science · en
HNF4α-HKDC1 axis orchestrates a metabolic rewiring to promote migration and metastasis in advanced gastric cancer
Why this matters for patients
Most deaths from stomach cancer occur not because of the original tumor, but because cancer cells travel to distant parts of the body and take root there. This paper uncovers a hidden molecular "on switch" that helps gastric (stomach) cancer cells change how they use fuel and become more mobile. Even more importantly, it points to an existing, FDA‑approved drug that might be repurposed to turn this switch off and slow or prevent spread.
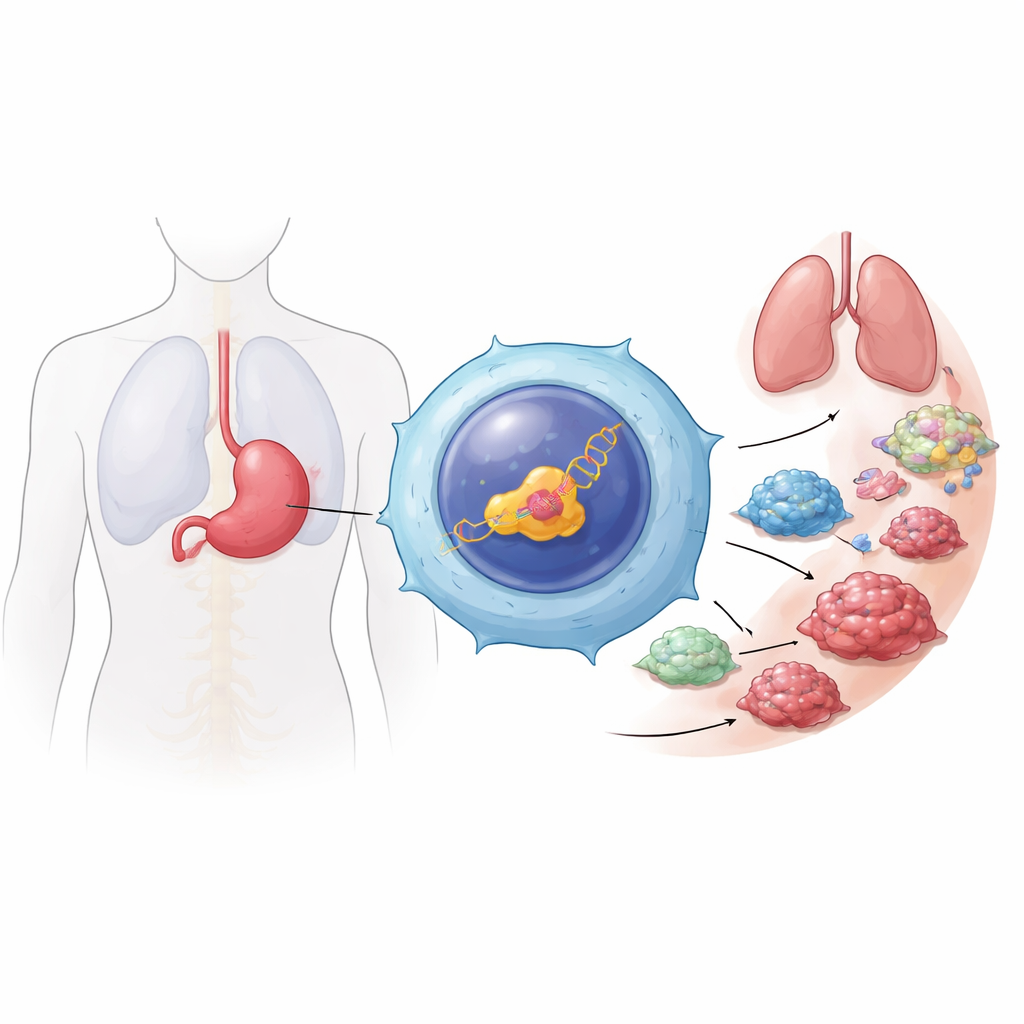
A dangerous shift in stomach cancer
Gastric cancer is the fifth most common cancer worldwide and is often detected late, after it has already spread. The authors focused on metastasis—the step when tumor cells break away, move through the abdomen or bloodstream, and colonize new sites. By mining large public cancer databases and examining tissue samples from patients, they found that a specific version of a gene regulator called HNF4α, driven by its P2 promoter (P2‑HNF4α), is especially abundant in distant metastatic lesions compared with primary tumors and nearby non‑cancerous tissue. This version was found mainly inside tumor cells, not in surrounding normal cells, suggesting it is tightly linked to the cancer’s invasive behavior.
A master switch that boosts cell movement
To test cause and effect, the researchers altered P2‑HNF4α levels in a panel of human gastric cancer cell lines that represent major molecular subtypes of the disease. When they reduced HNF4α in cells that normally express it, the cells became far less able to migrate and invade through artificial membranes or close wounds in culture dishes. When they forced cells with low HNF4α to make more of a representative P2‑HNF4α isoform, those cells became markedly more mobile and invasive, including in three‑dimensional spheroid assays that mimic real tumors. In mice, cells engineered to overproduce P2‑HNF4α formed many more metastatic nodules throughout the abdominal cavity, showing that this factor actively drives spread in living organisms.
Fueling spread by rewiring energy use
Cancer cells often reshape how they process nutrients to support growth and movement. Using combined RNA sequencing and metabolite profiling, the team discovered that turning down HNF4α dampens a key energy‑producing pathway: glycolysis, the breakdown of glucose. Levels of several glycolytic enzymes and their intermediate products dropped, as did measures of energy output such as oxygen consumption, acid production, and ATP generation. Further analyses singled out one enzyme, HKDC1, as the most strongly and consistently linked to HNF4α. HKDC1 sits at the very first step of glucose processing and helps direct sugar toward both lactate production and mitochondrial respiration—two outputs that together provide energy and create a chemical environment favorable to cell movement.
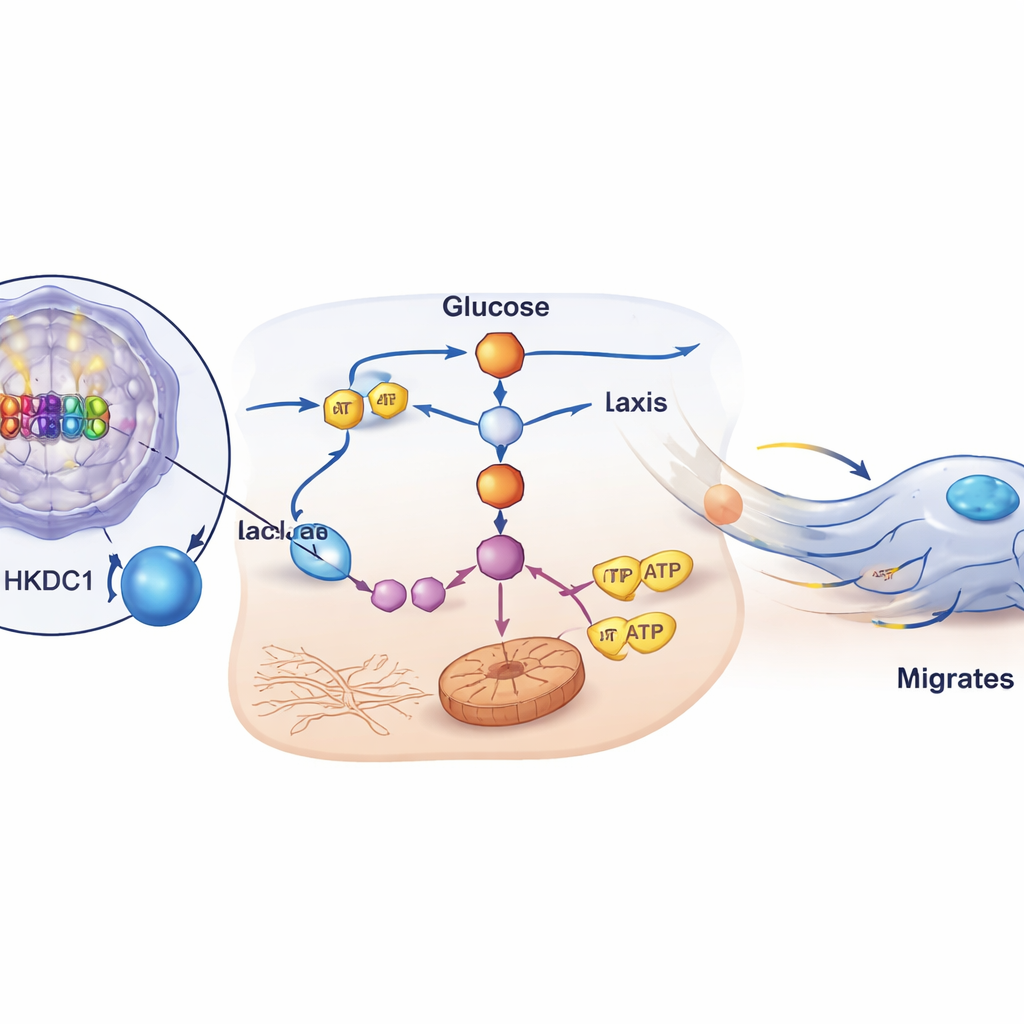
A direct molecular link and a key accomplice
The scientists showed that P2‑HNF4α does not just correlate with HKDC1; it directly turns this gene on. Using genome‑wide binding data, DNA reporter assays, and protein–DNA binding tests, they mapped specific sites in an enhancer region of the HKDC1 gene where HNF4α sits and activates transcription. When HKDC1 was reduced in cells, their ability to migrate and form peritoneal metastases in mice plummeted, even though their short‑term survival was not affected. Critically, restoring HKDC1 in cells where HNF4α had been knocked down rescued glycolytic activity, energy production, and migratory behavior, and adding metabolic end products such as lactate or α‑ketoglutarate also restored movement. Together, these results argue that HKDC1 is a central downstream enabler through which P2‑HNF4α reprograms metabolism to power metastasis.
Turning the switch off with an existing drug
Because HNF4α belongs to a family of proteins that are often drug targets, the team tested whether an approved medication could block this pro‑metastatic axis. They had previously identified mycophenolic acid, the active form of the transplant drug mycophenolate mofetil (MMF), as an HNF4α antagonist in lung cancer. Here, MMF treatment reduced HKDC1 levels and cell migration in gastric cancer lines that express high HNF4α, but had little effect in cells with low HNF4α. In mouse models, MMF significantly curtailed abdominal metastases and lowered HKDC1 expression—but only when tumors had high P2‑HNF4α. This suggests that HNF4α could serve both as a therapeutic target and as a biomarker to select patients most likely to benefit from MMF‑based strategies.
What this means for future care
In plain terms, this study identifies a molecular fuel‑control system—P2‑HNF4α working through HKDC1—that helps stomach cancer cells rev up their energy machinery and become better at spreading. By showing that an existing drug can interfere with this system and reduce metastasis in preclinical models, it opens a realistic path toward new treatments for advanced gastric cancer. If future clinical studies confirm these findings, testing tumors for HNF4α activity could help guide the use of MMF or related agents to slow or prevent the deadly spread of this disease.
Citation: Xu, X., Wu, H., Shang, J. et al. HNF4α-HKDC1 axis orchestrates a metabolic rewiring to promote migration and metastasis in advanced gastric cancer. Cell Death Dis 17, 347 (2026). https://doi.org/10.1038/s41419-026-08627-y
Keywords: gastric cancer metastasis, HNF4A, HKDC1, cancer metabolism, mycophenolate mofetil