Clear Sky Science · en
The DAG/PKC/CREB1/TGF-β1 axis drives shear-wave elastography stiffness and malignant progression in triple-negative breast cancer via lipid metabolic reprogramming
Why Tumor Stiffness Matters
Doctors have long noticed that some breast tumors feel harder than others, and that this stiffness often signals a more dangerous cancer. In triple-negative breast cancer, an aggressive form that lacks common drug targets, obesity appears to make tumors stiffer and more likely to spread. This study explains how changes in fat metabolism in obese patients harden tumors and shows that a specialized ultrasound technique, shear-wave elastography, can read this stiffness from the outside as a window into tumor behavior.
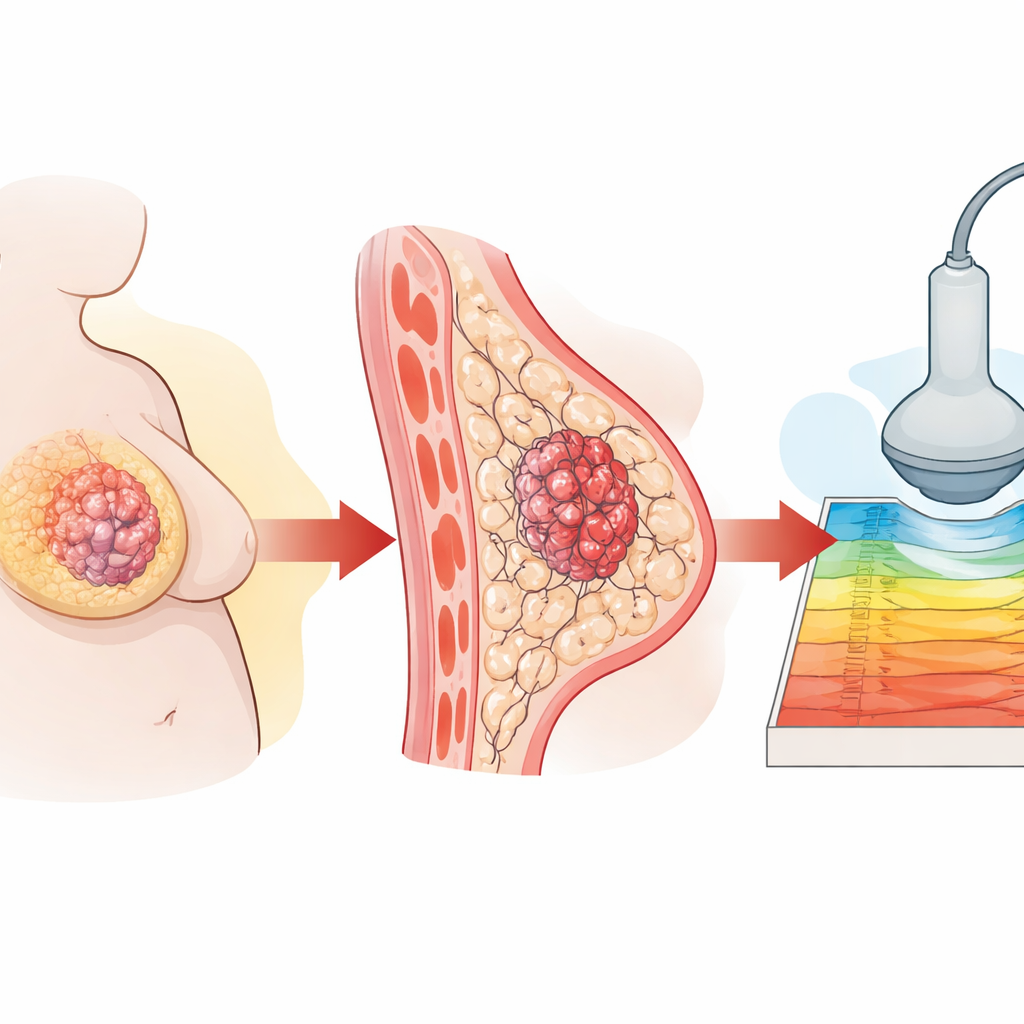
Body Weight and Tumor Risk
The researchers started by following 147 women with triple-negative breast cancer at a major cancer center in Shanghai. They found that women with higher body mass index (BMI) had worse long-term outcomes, including more recurrences and deaths. When they examined ultrasound scans, they saw that tumors in heavier patients were consistently stiffer on shear-wave elastography, even though standard gray-scale ultrasound images looked similar. Tissue samples confirmed that tumors from heavier patients contained more collagen and more actively dividing cells, both signs of a more hostile environment.
From Diet to Stiffer Tumors
To probe cause and effect, the team turned to mouse models, feeding some mice a high-fat diet and others a normal diet before implanting triple-negative breast tumors. Mice on the high-fat diet gained more weight, grew larger tumors, and, crucially, showed steadily rising stiffness on shear-wave elastography over time. Microscopic examination revealed denser collagen networks, more activated support cells called fibroblasts, and higher levels of an enzyme that cross-links collagen, all of which make tissue more rigid. At the same time, tumor cells from obese mice showed signs of revved-up fat metabolism and accumulated more lipid droplets inside.
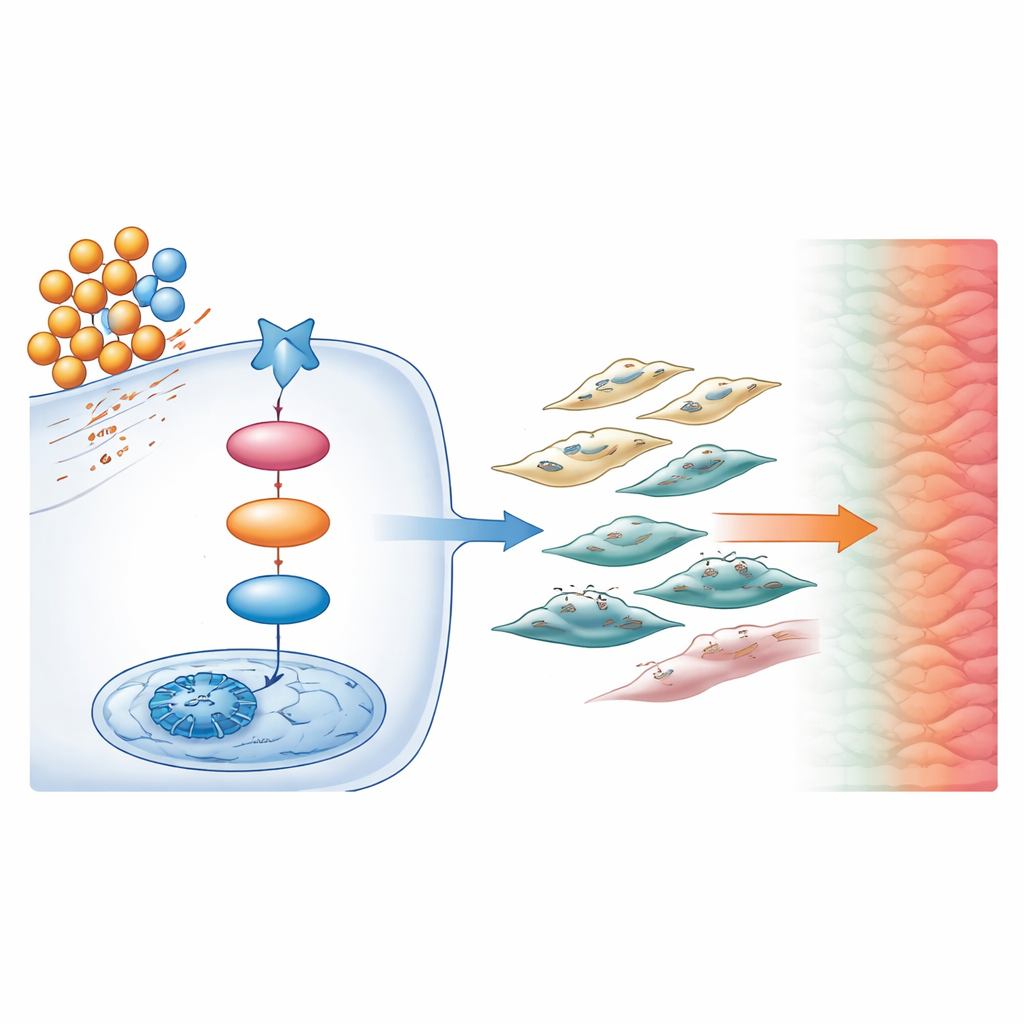
A Hidden Chemical Chain Reaction
Digging deeper, the scientists used lipid profiling and gene activity analysis to map the molecular chain reaction driving these changes. They found that a particular fat-based molecule called diacylglycerol (DAG) was elevated in tumors from obese mice. DAG acted like a biochemical switch, turning on a protein called PKC inside cancer cells. PKC in turn boosted the activity of a master control protein, CREB1, in the cell nucleus. Once activated, CREB1 drove the production of TGF-β1, a powerful signaling molecule known to promote scarring and cancer spread. Together, this DAG → PKC → CREB1 → TGF-β1 sequence formed a central pathway linking excess fat to a stiffer, more invasive tumor.
A Vicious Conversation Between Cells
TGF-β1 did not act on cancer cells alone. It also activated nearby fibroblasts, the structural cells that build and remodel the tissue framework around tumors. When exposed to signals from DAG-stimulated cancer cells, fibroblasts switched into a highly active state, laying down extra collagen and further tightening the tumor environment. These activated fibroblasts, in turn, sent signals back to the cancer cells that increased their fat metabolism, growth, and invasiveness. In mouse models, this back-and-forth conversation created a self-reinforcing loop that made tumors stiffer on shear-wave elastography, more fibrotic under the microscope, and more prone to forming distant metastases.
Turning Images Into Treatment Clues
By combining patient data, animal experiments, and detailed molecular studies, the authors show that shear-wave elastography stiffness is more than a structural oddity: it reflects the activation of a specific fat-driven signaling loop that worsens triple-negative breast cancer. For a layperson, this means that a noninvasive ultrasound measurement of how hard a tumor feels can reveal how strongly this harmful pathway is switched on. In the future, patients with high BMI and very stiff tumors on elastography might be flagged as high risk and considered for drugs that block PKC, CREB1, TGF-β1, or related steps in the loop, turning a simple imaging readout into a guide for more precise, biology-based treatment.
Citation: Wang, S., Zheng, D., Wang, Z. et al. The DAG/PKC/CREB1/TGF-β1 axis drives shear-wave elastography stiffness and malignant progression in triple-negative breast cancer via lipid metabolic reprogramming. Cell Death Dis 17, 327 (2026). https://doi.org/10.1038/s41419-026-08625-0
Keywords: triple-negative breast cancer, tumor stiffness, obesity, lipid metabolism, shear-wave elastography