Clear Sky Science · en
Metabolic orchestration driven by GGCT: diverting glutamine to glutathione biosynthesis while enhancing glucose anaplerosis for tumor proliferation
Why this matters for cancer treatment
Cancer cells grow fast, and to do so they burn through huge amounts of fuel and must continually protect themselves from toxic by‑products of their own metabolism. This study uncovers how a little‑known enzyme, GGCT, helps liver and prostate tumors cleverly reroute two common nutrients—glutamine and glucose—so they can both power growth and keep damaging molecules called reactive oxygen species under control. Understanding this metabolic juggling act could open up fresh ways to starve tumors or overwhelm their defenses.
How tumor cells juggle fuel and protection
Tumor cells depend heavily on glutamine, an amino acid that plays a double role: it can be broken down to feed the cell’s main energy‑producing cycle, and it also supplies building blocks for glutathione, a powerful antioxidant that neutralizes reactive oxygen species. The authors first confirmed that glutamine levels are higher in liver tumors than in nearby healthy tissue, and that cancer cells grown with more glutamine divide more rapidly both in dishes and in mice. When glutamine is scarce, cells slow down, key cell‑cycle proteins drop, and tumors in animals shrink—partly because antioxidant defenses weaken and reactive oxygen species accumulate.
A metabolic switch hiding in plain sight
Looking across patient samples and large cancer databases, the team found that the enzyme GGCT is consistently more abundant in liver and prostate tumors than in normal tissues, and higher GGCT levels predict worse outcomes. In both tumor samples and cultured cells, GGCT levels rise in step with glutamine concentration, hinting that tumors “read” glutamine availability through this protein. The researchers worked out a control chain: glutamine keeps the pro‑growth factor c‑Myc active, which suppresses a small regulatory RNA called miR‑29b‑3p; when miR‑29b‑3p is low, GGCT is released from its restraint and accumulates. Under glutamine shortage, this chain reverses, miR‑29b‑3p rises, GGCT messages are broken down more quickly, and the enzyme level falls.
Redirecting glutamine and pulling in more glucose
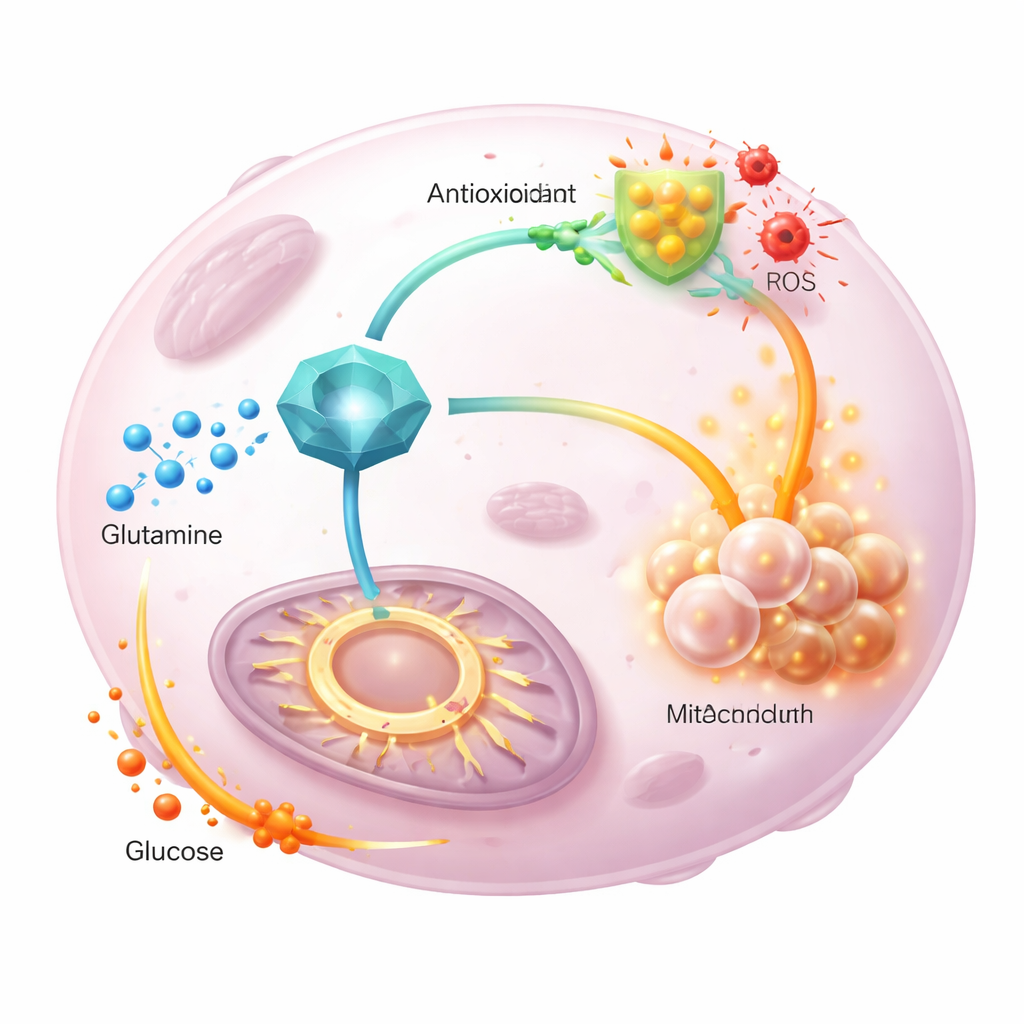
To see what GGCT actually does inside the cell, the authors reduced or boosted its levels and measured both cell behavior and hundreds of metabolites. Silencing GGCT stalled cells in the division cycle, shrank their colonies, and sharply lowered glutathione while raising reactive oxygen species; adding a chemical antioxidant partly rescued growth. Under the microscope, mitochondria became abnormally elongated and less efficient at consuming oxygen, even as cells tried to compensate by ramping up glycolysis, the first step of sugar breakdown. Detailed metabolic profiling showed that intermediates of the central energy cycle dropped when GGCT was lost and rose when it was overproduced.
Tracing the path of carbon atoms
The team then followed labeled glutamine and glucose atoms as they flowed through tumor metabolism. When GGCT was overexpressed, less labeled glutamine ended up in the energy cycle, and more was channeled into newly made glutathione, supporting antioxidant defenses. At the same time, labeled glucose contributed more strongly to the cycle, compensating for the diverted glutamine. A mutant form of GGCT that lacked normal enzyme activity could not drive these shifts or promote proliferation, showing that GGCT’s catalytic function is crucial. Importantly, supplying extra pyruvate or tweaking entry into the energy cycle restored cellular energy levels but did not lower reactive oxygen species, indicating that GGCT mainly controls redox balance through glutathione, not by simple changes in energy supply.
What this means for future therapies
Taken together, the findings position GGCT as a central coordinator that lets tumors split glutamine’s duties: it steers more of this nutrient toward antioxidant production while pulling glucose into the energy cycle to keep cell division humming. In animal models, knocking down GGCT slowed tumor growth, reduced glutathione, and raised oxidative stress, and these effects could be partly reversed with an antioxidant drug. For a lay observer, the message is that some cancers survive by using GGCT as a metabolic switchboard; drugs that disable this switch could simultaneously weaken the tumor’s defenses against oxidative damage and disrupt its flexible use of nutrients, making standard treatments more effective.
Citation: Yang, L., Sun, H., Wang, R. et al. Metabolic orchestration driven by GGCT: diverting glutamine to glutathione biosynthesis while enhancing glucose anaplerosis for tumor proliferation. Cell Death Dis 17, 358 (2026). https://doi.org/10.1038/s41419-026-08619-y
Keywords: cancer metabolism, glutamine, glutathione, oxidative stress, GGCT enzyme