Clear Sky Science · en
AATF supports proliferation of glioblastoma cells by sustaining mitochondrial respiration through an NRF-1-dependent mechanism
Why brain tumor energy use matters
Glioblastoma is the most aggressive form of adult brain cancer, and despite surgery, radiation and chemotherapy, most patients survive little more than a year. This study asks a deceptively simple question with far-reaching implications: how do glioblastoma cells fuel their relentless growth, and can that fuel line be cut? The authors focus on a little-known regulatory protein called AATF and show that it helps tumor cells keep their energy factories—mitochondria—running at full speed. Understanding this hidden support system may open new ways to slow or stop these deadly tumors.
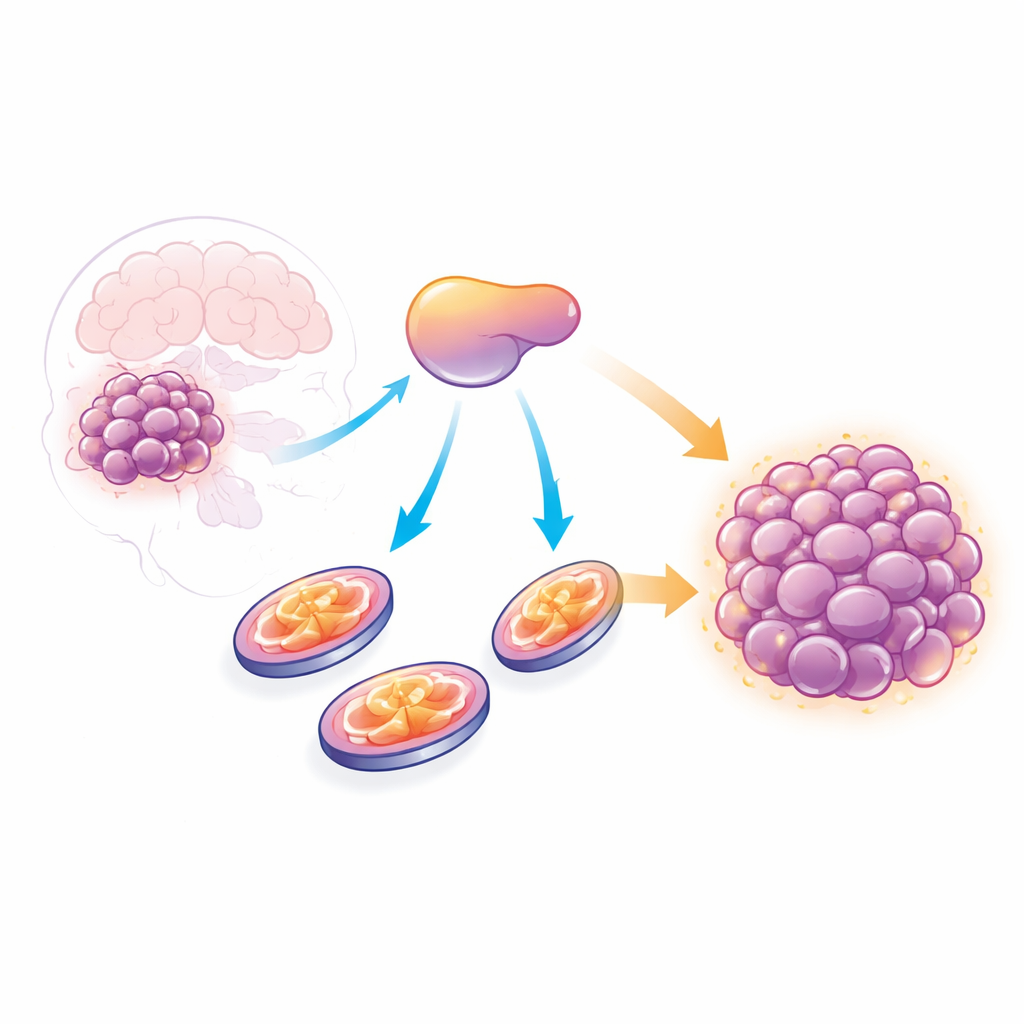
A hidden helper in tumor growth
Cancer cells often rely on a sugar-burning strategy called glycolysis, long considered their trademark energy pathway. Yet recent work has revealed that many tumors, including glioblastoma, also depend heavily on mitochondria, which produce energy through oxidative phosphorylation, or OXPHOS. By mining large patient datasets, the researchers found that AATF levels are higher in glioblastoma tissue than in normal brain and rise with tumor grade. Patients whose tumors expressed more AATF tended to have poorer survival, suggesting that this protein is linked to more aggressive disease.
Shutting down AATF slows cancer cells
To test whether AATF is merely a marker or an active driver of growth, the team reduced its levels in several glioblastoma cell lines using RNA-based tools and CRISPR interference. Cells lacking AATF multiplied far more slowly, accumulated in the resting phase of the cell cycle, and formed many fewer colonies over the long term. Importantly, this slowdown happened without triggering widespread cell death or senescence, meaning the cells were alive but stuck. When AATF-depleted cells were implanted into mice, the resulting tumors were much smaller and showed fewer dividing cells, confirming that AATF is required for robust tumor growth in living tissue.
Rewiring the tumor power grid
Because AATF is known to influence gene activity, the authors examined how its loss reshapes the cell’s genetic program. RNA sequencing revealed that hundreds of genes changed their activity when AATF was silenced. Among the most consistently reduced were genes encoding key components of the mitochondrial OXPHOS machinery. Protein measurements confirmed that multiple parts of the energy-producing complexes inside mitochondria were diminished. When the researchers directly measured oxygen consumption and energy output, AATF-depleted cells showed lower respiration and reduced ATP levels. At the same time, their mitochondria produced more reactive oxygen species, adopted a fragmented, ring-like shape associated with dysfunction, and upregulated genes that promote mitochondrial fission, all signs of a stressed and compromised power grid.
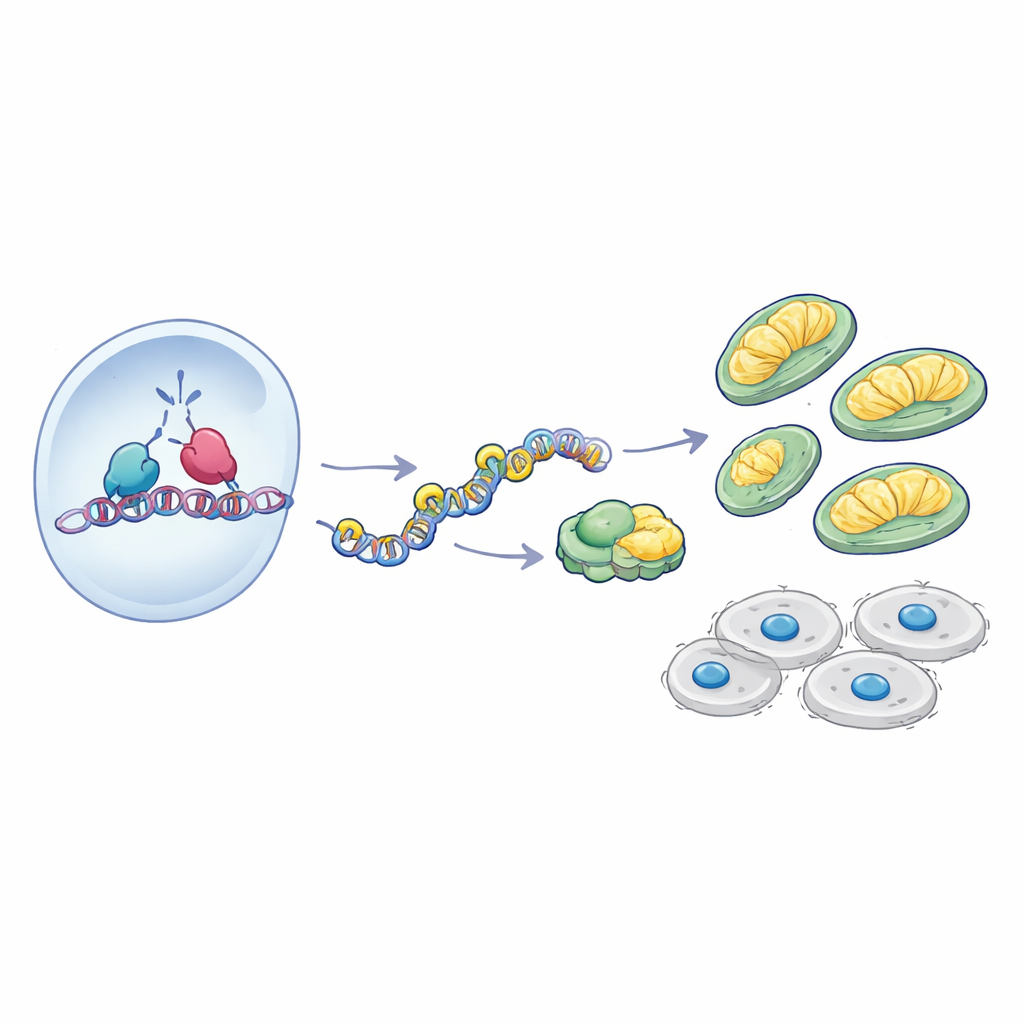
A partnership at the DNA level
How does AATF exert such control over mitochondrial genes? The study zeroed in on NRF-1, a transcription factor already known as a master regulator of mitochondrial biogenesis and OXPHOS. Patient data showed that AATF and NRF-1 levels rise and fall together in glioblastoma, and experiments in cell lines revealed that the two proteins physically interact. Using chromatin immunoprecipitation, the authors demonstrated that AATF is present at the promoter regions of several OXPHOS genes precisely where NRF-1 binds. When NRF-1 was knocked down, AATF binding to these promoters dropped sharply, indicating that NRF-1 helps recruit AATF to the DNA. Further, in cells lacking AATF, NRF-1 could still bind DNA, but the recruitment of the transcription enzyme RNA polymerase II was impaired and the surrounding chromatin became more tightly packed, with fewer activating histone marks and more repressive ones. In essence, without AATF, NRF-1 sits on the switches, but the lights do not turn on.
What this means for future treatments
To a non-specialist, the message is that glioblastoma cells use a specialized partnership between AATF and NRF-1 to keep their mitochondrial engines running hot, supplying the energy needed for rapid division and treatment resistance. AATF acts as a co-pilot at mitochondrial genes, shaping the local DNA environment so that NRF-1 can drive strong expression of OXPHOS components. Removing AATF stalls this system: mitochondria falter, damaging by-products build up, and tumor cells lose their proliferative edge both in dishes and in mice. Although there are currently no drugs that directly block AATF, this work identifies it as a promising target in glioblastoma and suggests that disrupting the AATF–NRF-1–mitochondria axis could weaken the tumor’s energy supply and improve the effectiveness of existing therapies.
Citation: Sorino, C., Di Giovenale, S., Falcone, I. et al. AATF supports proliferation of glioblastoma cells by sustaining mitochondrial respiration through an NRF-1-dependent mechanism. Cell Death Dis 17, 349 (2026). https://doi.org/10.1038/s41419-026-08617-0
Keywords: glioblastoma, mitochondrial respiration, oxidative phosphorylation, transcriptional regulation, cancer metabolism