Clear Sky Science · en
Arginine dependency in omental metastasis of epithelial ovarian cancer reveals a therapeutic vulnerability
Why this matters for women’s health
Epithelial ovarian cancer is one of the deadliest cancers affecting women because it is usually discovered only after it has quietly spread through the abdomen. A favorite landing spot for these wandering cancer cells is a fatty apron of tissue called the omentum. This study uncovers a hidden weakness in those omental metastases: they are unusually dependent on the dietary amino acid arginine. By tracing how metastatic cells use arginine to repair their damaged DNA and survive stress, the researchers point to new treatment strategies that might starve metastases or block their escape routes.
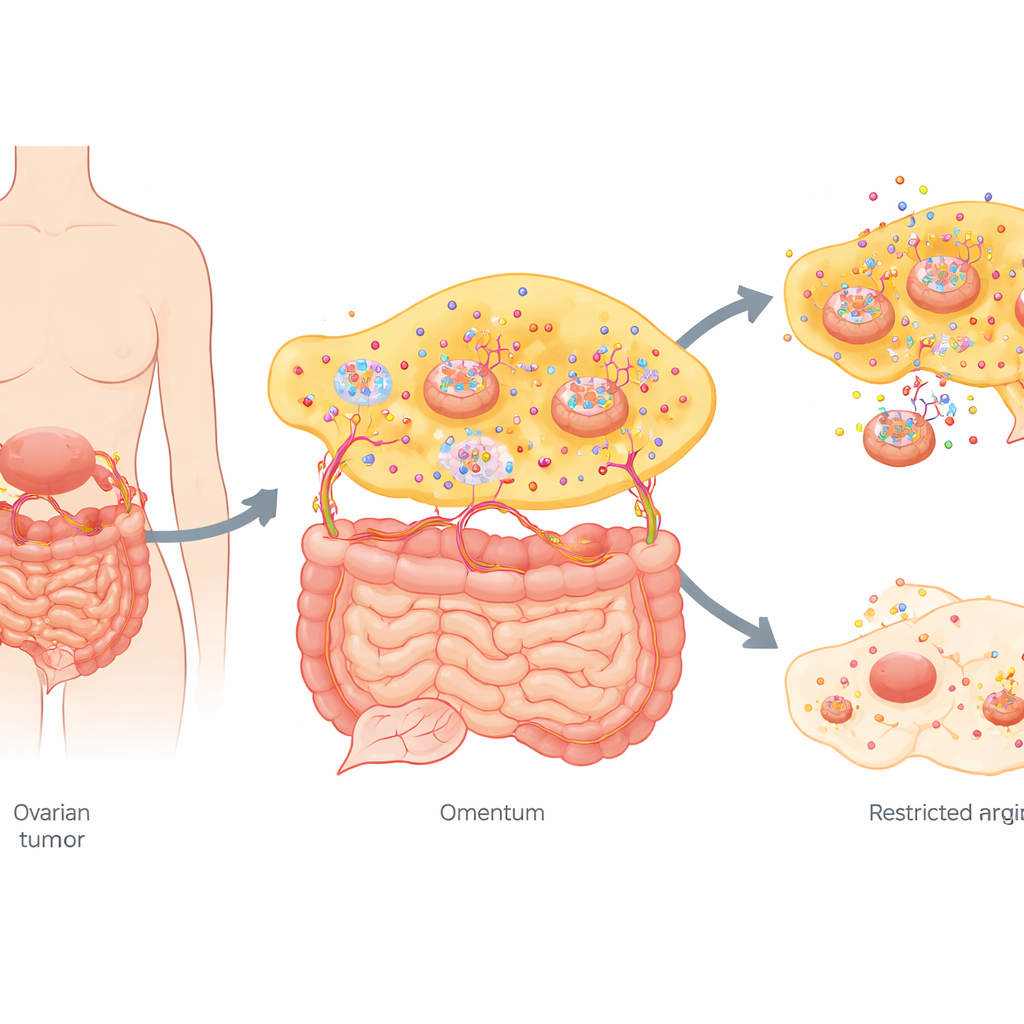
A nutrient-hungry cancer in a special niche
Ovarian tumors at an advanced stage often seed tiny colonies across the abdominal cavity, especially in the omentum, a fat-rich tissue draped over the intestines. The team compared primary ovarian tumors with their matching omental metastases from women who had not yet received treatment. A broad survey of hundreds of small molecules showed that the metabolic “fingerprint” of metastases was strikingly different. Among many changes, one stood out: omental metastases accumulated far more arginine than the original ovarian tumors. At the same time, the enzymes needed to make arginine inside the cell were dialed down, while transport proteins that import arginine from outside were turned up, indicating that metastatic cells had become reliant on arginine from their surroundings.
Starving tumors by cutting arginine supply
To test whether this arginine dependence truly mattered, the researchers used mouse models of ovarian cancer. When tumor-bearing mice were placed on an arginine-free diet, both the main ovarian tumors and the widespread abdominal metastases shrank markedly. Imaging showed weaker tumor signals, fewer and smaller metastatic spots, less fluid buildup in the abdomen, and better survival compared with mice on a normal diet. In dishes, human ovarian cancer cells grew slowly, migrated less, and invaded less when arginine was scarce. Raising arginine to levels similar to or higher than those seen in patients with metastases had the opposite effect, speeding growth and movement. Overexpressing an arginine transporter further boosted these aggressive behaviors, underscoring that the tumors thrive when arginine is plentiful.
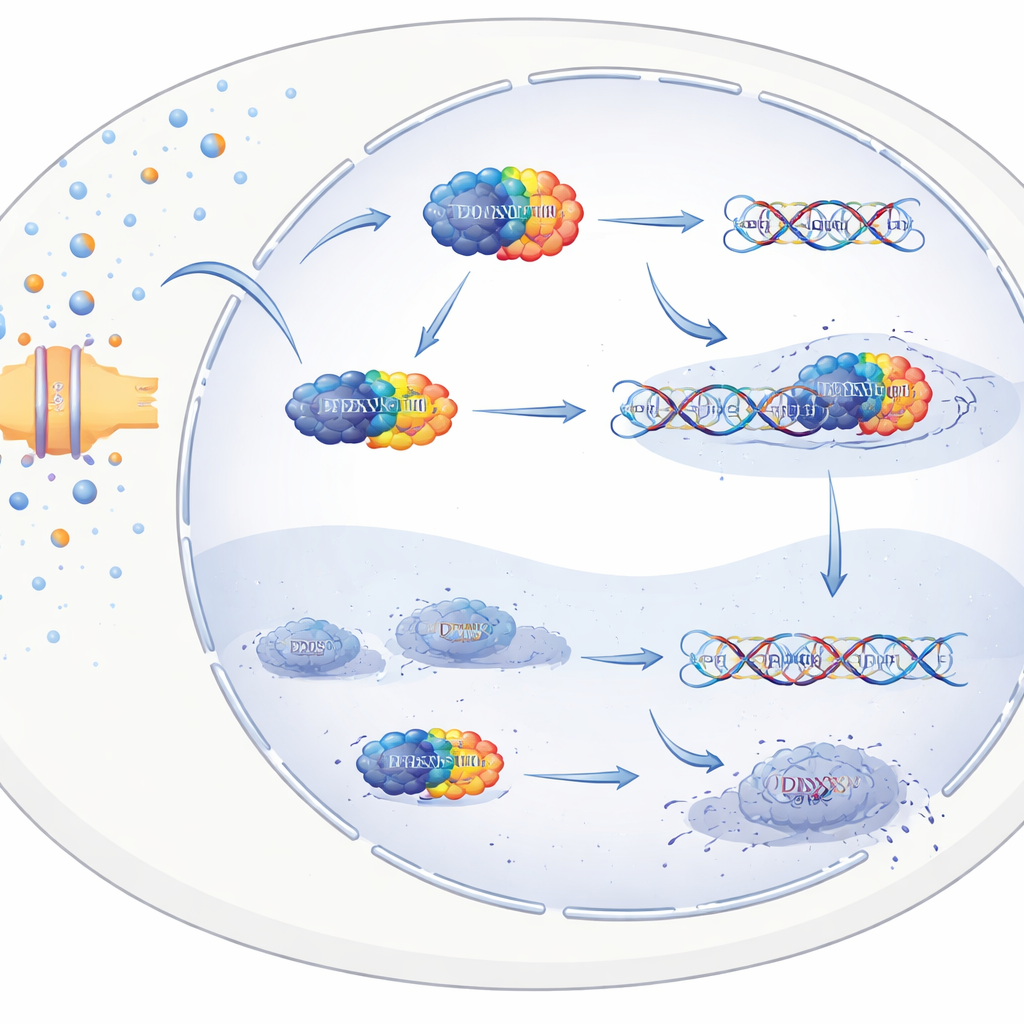
How arginine helps cancer cells fix their DNA
The scientists then asked how arginine fuels metastasis at the molecular level. They used chemical “bait” to fish out proteins that physically bind arginine and identified many RNA-handling proteins, focusing on one called DDX3X. This protein was more abundant in metastases than in primary tumors and was linked to poorer patient outcomes. Laboratory experiments and computer simulations showed that arginine docks onto a specific region of DDX3X and changes where it sits inside the cell. With high arginine, DDX3X builds up in the cell nucleus, because arginine interferes with the signal that normally exports DDX3X back to the cytoplasm. Once in the nucleus, DDX3X associates with DNA and boosts the activity of genes involved in DNA damage response pathways, particularly the ATM–CHK2–p53 axis that senses DNA breaks and coordinates their repair.
Surviving stress in a dangerous neighborhood
Metastatic cells in the omentum face intense oxidative stress and show more DNA damage than primary tumors, which should in principle threaten their survival. The study found that arginine-rich metastases also had higher levels of markers of active DNA repair. When arginine was added to cells exposed to chemotherapy or oxidative stress, DNA breaks were repaired more efficiently and fewer cells died. Removing DDX3X, or blocking the ATM or CHK2 proteins downstream of it, erased this protective effect. In other words, arginine allows metastatic ovarian cancer cells to sense their nutrient environment and, through DDX3X, switch on powerful DNA repair programs that help them withstand both the hostile omental niche and anti-cancer treatments.
Turning a nutrient craving into a treatment opportunity
Because long-term arginine restriction in patients could be difficult, the authors explored drug-based strategies. In mice, depleting arginine in the bloodstream with an enzyme or directly inhibiting DDX3X with a small molecule both slowed tumor growth, reduced metastatic spread, and extended survival. Combining arginine depletion with DDX3X inhibition worked even better, and blocking DDX3X also sensitized cisplatin-resistant ovarian cancer cells to chemotherapy, shrinking resistant tumors in mice. Together, the findings reveal that omental metastases in ovarian cancer are metabolically addicted to external arginine, which they channel through DDX3X to maintain DNA repair and survival. Targeting this arginine–DDX3X–DNA repair circuit, either by limiting arginine or by inhibiting DDX3X, could open new avenues to prevent or treat metastatic ovarian cancer and overcome drug resistance.
Citation: Tian, J., Lei, T., Du, Y. et al. Arginine dependency in omental metastasis of epithelial ovarian cancer reveals a therapeutic vulnerability. Cell Death Dis 17, 354 (2026). https://doi.org/10.1038/s41419-026-08606-3
Keywords: ovarian cancer, arginine metabolism, cancer metastasis, DNA damage repair, DDX3X