Clear Sky Science · en
A feedback mechanism from prostate cancer cells to macrophages, reinforced by STAT1, regulates tumor progression and resistance to radiotherapy
Why this research matters
Radiotherapy is a cornerstone treatment for prostate cancer, yet many tumors gradually stop responding, allowing the disease to return or spread. This study uncovers a hidden chemical “conversation” between prostate cancer cells and nearby immune cells called macrophages that helps tumors survive radiation. By decoding this loop, the researchers highlight new drug targets that could make radiotherapy more effective for men with advanced prostate cancer.
Cancer cells that burn sugar differently
Prostate cancer cells, like many tumors, rely on an altered form of sugar burning known as aerobic glycolysis. Instead of fully breaking down sugar with oxygen, they choose a faster but less efficient path that produces large amounts of lactic acid. The team identified a protein inside tumor cells, STAT1, as a key switch that turns this sugar-hungry state on. STAT1 boosts the activity of three core enzymes that drive glycolysis, increasing both sugar uptake and lactic acid production. In cell cultures and mouse models, tumors with higher STAT1 grew faster and were more resistant to radiation, whereas blocking glycolysis blunted these aggressive traits. 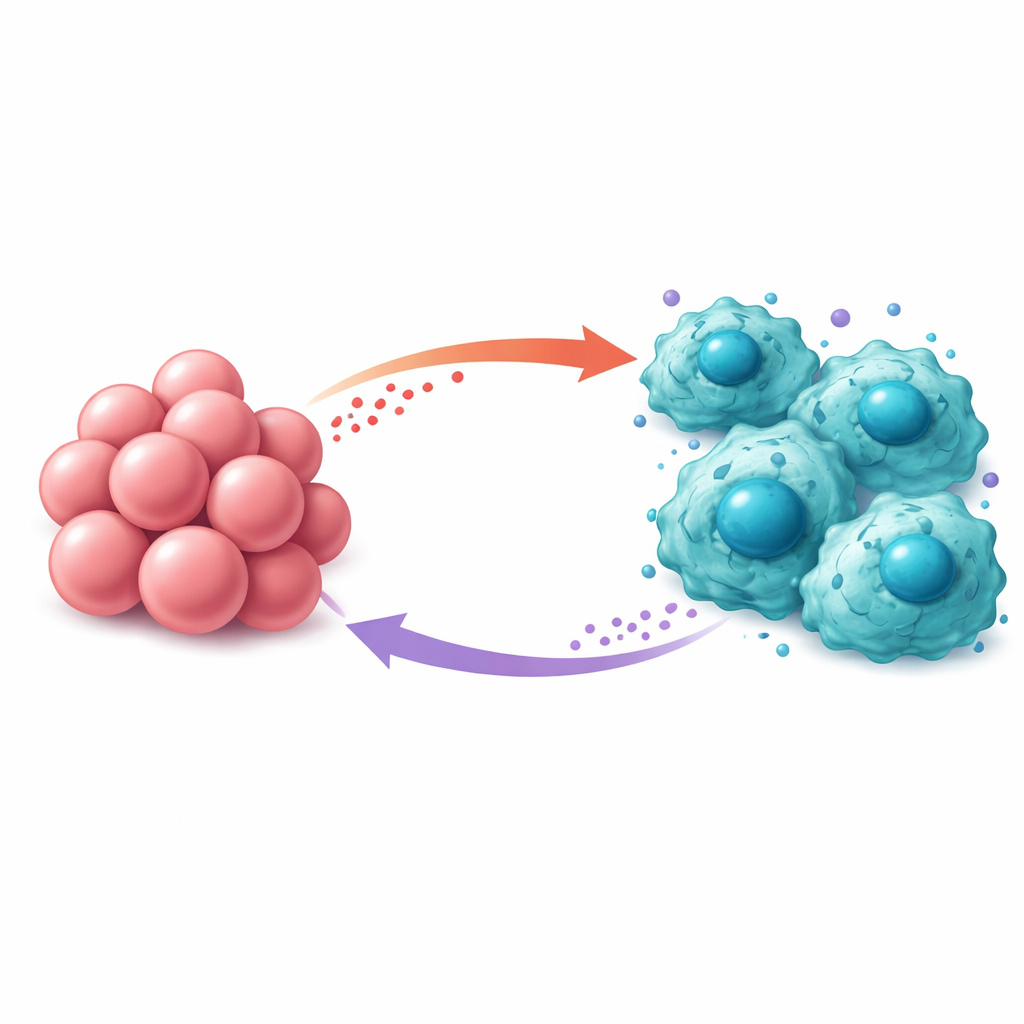
How waste from tumors reshapes immune cells
Lactic acid is often thought of as metabolic “waste,” but in the tumor’s neighborhood it acts more like a signal. The researchers showed that lactic acid released by prostate cancer cells is taken up by macrophages through a membrane transporter called MCT1. Once inside, this acid activates an internal alarm system known as the NFκB pathway, which nudges macrophages into a helper state called M2. Instead of attacking cancer, M2 macrophages tend to dampen immune responses and support tumor growth, for example by promoting blood vessel formation. When the scientists blocked glycolysis in cancer cells, inhibited lactic acid production, or stopped its entry into macrophages, these immune cells shifted away from the tumor-supporting M2 state and toward a more defensive profile.
Macrophages send growth signals back to tumors
The story does not end with macrophages simply changing character. Once lactic acid activates NFκB inside them, macrophages begin to make and secrete a powerful signaling molecule called MCP-1. This molecule diffuses back to prostate cancer cells and binds to a surface receptor named CCR2. That binding switches on yet another internal pathway in the cancer cells, involving JAK proteins and STAT1. In other words, the same STAT1 that first drove excess sugar burning is switched on again by a signal that ultimately traces back to the lactic acid it helped generate. When the team added MCP-1 to prostate cancer cells, glycolysis increased, cells grew and moved more readily, and they repaired radiation-induced DNA damage more efficiently. Blocking CCR2 or JAK prevented these effects.
A self-reinforcing survival loop
Taken together, the results reveal a self-reinforcing loop between cancer cells and macrophages. STAT1 inside prostate cancer cells boosts sugar breakdown and lactic acid release. That lactic acid is imported into macrophages, where it activates NFκB and pushes them into a tumor-supporting M2 state while triggering MCP-1 production. MCP-1 then travels back to cancer cells, activates CCR2 and the JAK/STAT1 pathway, and further strengthens glycolysis and resistance to radiotherapy. Animal experiments showed that drugs blocking CCR2 or JAK, especially when combined with radiation, slowed tumor growth and reduced the presence of M2 macrophages, underscoring the clinical promise of targeting this circuit. 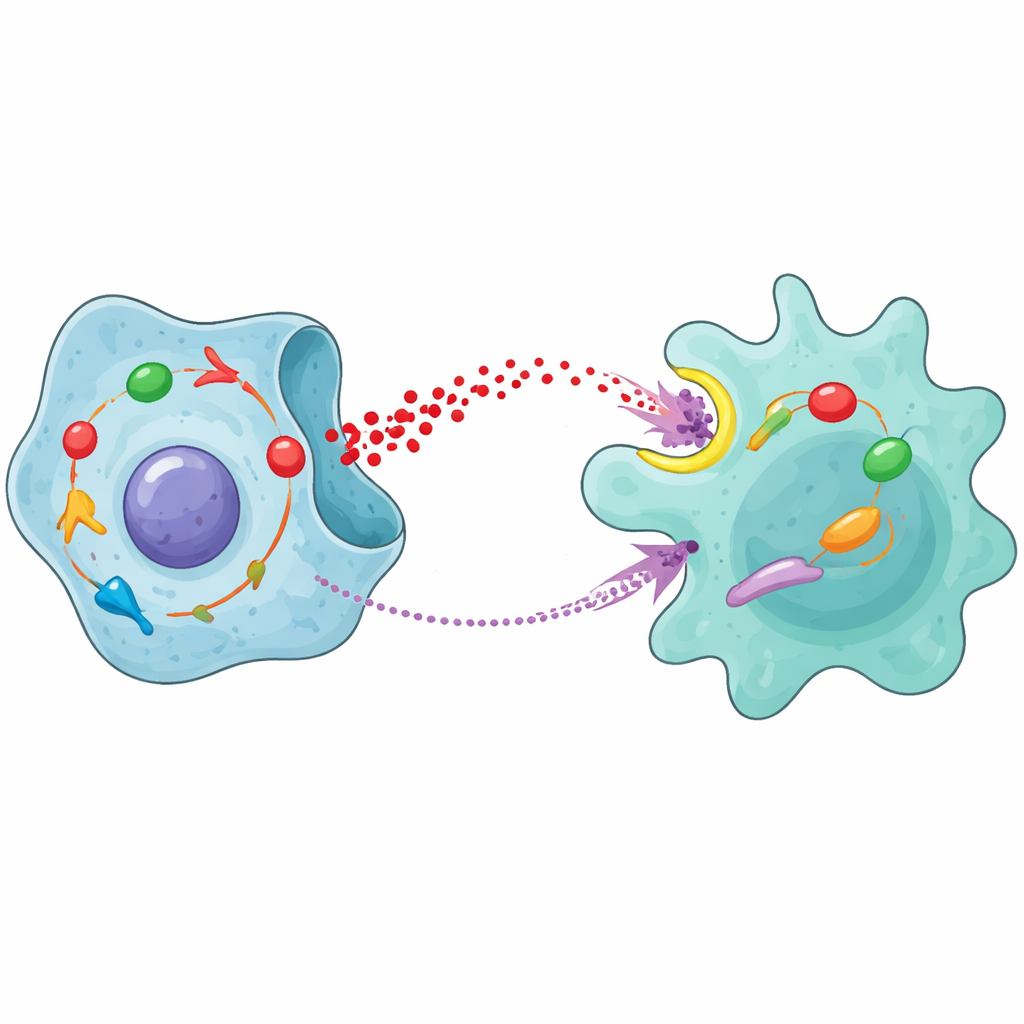
What this means for patients
For a layperson, the key idea is that prostate tumors and certain immune cells form a chemical partnership that helps the cancer endure radiation treatment. Tumor cells change how they use sugar, producing lactic acid that reprograms nearby macrophages into allies instead of enemies. Those reprogrammed cells then send signals back that make tumors tougher and harder to kill. By interrupting this loop—at the level of lactic acid handling, the MCP-1 signal, or the downstream JAK/STAT1 pathway—future therapies could weaken the tumor’s defenses and allow radiotherapy to work better, offering new hope for men with difficult-to-treat prostate cancer.
Citation: Chen, JY., Xue, YT., Lin, B. et al. A feedback mechanism from prostate cancer cells to macrophages, reinforced by STAT1, regulates tumor progression and resistance to radiotherapy. Cell Death Dis 17, 282 (2026). https://doi.org/10.1038/s41419-026-08577-5
Keywords: prostate cancer, radiotherapy resistance, tumor microenvironment, macrophages, lactate metabolism