Clear Sky Science · en
Vaccinia-related kinase 2 inhibition elicits vulnerability of glutathione metabolism in pancreatic cancer
Why This Matters for Pancreatic Cancer
Pancreatic cancer is among the deadliest cancers, in part because standard treatments often fail. This study uncovers a hidden weakness in certain pancreatic tumors: a dependence on a cellular “shield” against toxic oxygen byproducts. By understanding when that shield is fragile, and how a cancer gene called VRK2 strengthens it, the researchers suggest new ways to selectively poison cancer cells while sparing healthy tissue.
The Cell’s Rust Problem
Every cell constantly battles “rusting from within” caused by reactive oxygen species—highly reactive forms of oxygen that can damage DNA, fats, and proteins. To survive, cells use chemical defenses, with glutathione acting as one of the most important shields. Pancreatic cancer cells, which live under harsh conditions and grow rapidly, lean heavily on metabolic tricks to keep enough glutathione on hand. Disrupting this balance between damaging oxidants and protective antioxidants can push cancer cells over the edge into cell death.
Finding a Hidden Weak Spot
The researchers began by turning off VRK2, a cancer-promoting gene previously linked to poor outcomes in pancreatic cancer, and then testing 281 metabolism-targeting compounds. They discovered that cells lacking VRK2 were strikingly sensitive to drugs that block glutathione production. In these VRK2-deficient cells, glutathione levels dropped while reactive oxygen shot up, killing the cells. Supplying extra antioxidants or glutathione-like molecules rescued them, confirming that the key vulnerability lay squarely in their impaired ability to detoxify oxidative stress.
How VRK2 Helps Cancer Cells Armor Up
Next, the team asked why VRK2 changes glutathione metabolism. They homed in on a transporter protein called SLC7A11, which sits in the cell’s outer membrane and ferries in cystine, a building block required to make glutathione. Surprisingly, VRK2 did not alter how much SLC7A11 the cell produced overall; instead, it controlled whether that transporter successfully reached the cell surface. In healthy VRK2-active cells, SLC7A11 travels from the endoplasmic reticulum to the Golgi and then to the membrane. VRK2 boosts this trafficking by chemically modifying a cargo-sorting protein called Sec24C, which helps load SLC7A11 into transport vesicles. When VRK2 is missing or blocked, SLC7A11 gets stuck inside the cell, fewer transporters reach the surface, and the glutathione shield weakens.
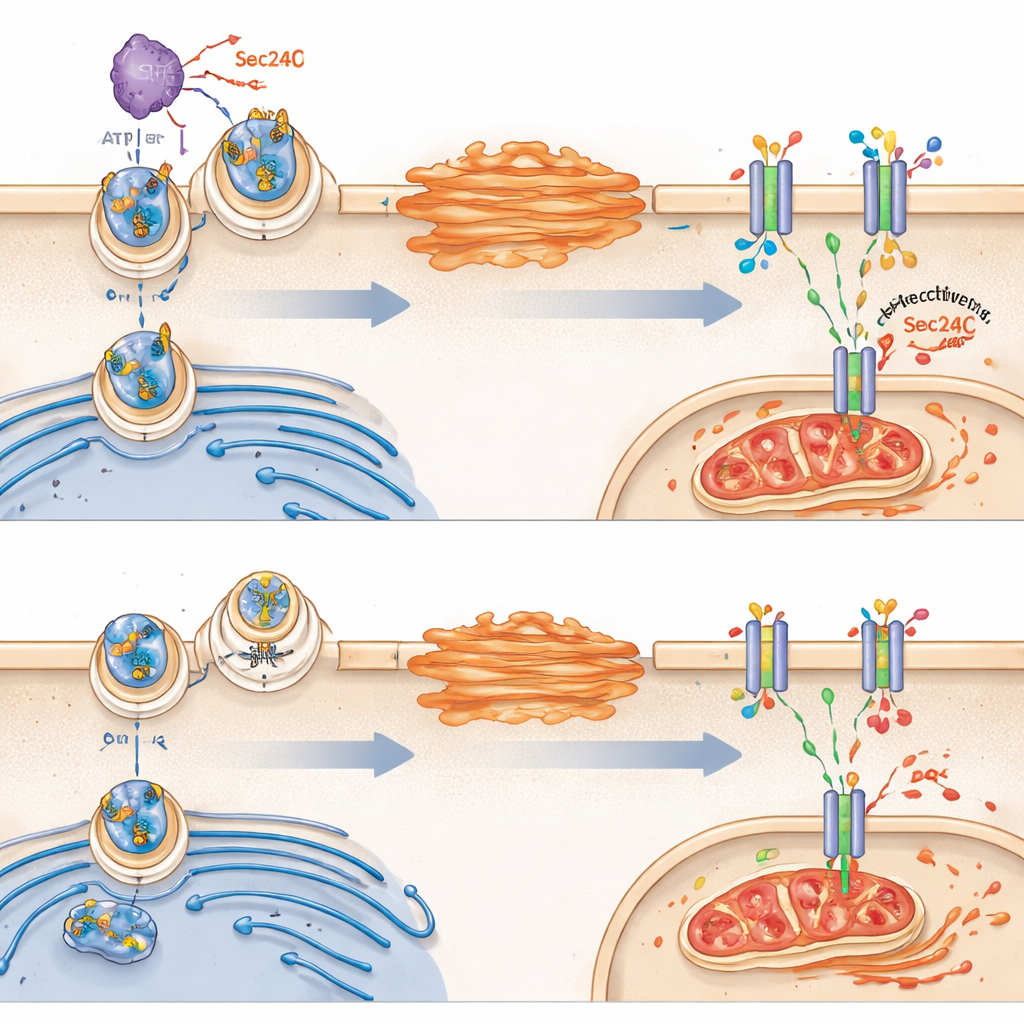
From Broken Shield to Ferroptosis
Without enough membrane SLC7A11, VRK2-deficient cells struggle to import cystine and maintain glutathione. This leaves them especially vulnerable to a form of iron-dependent cell death called ferroptosis, characterized by runaway oxidation of cell membrane fats and mitochondrial damage. The study showed that drugs cutting off glutathione synthesis triggered hallmarks of ferroptosis in VRK2-deficient cells, including distorted mitochondria and buildup of lipid oxidation products. Blocking ferroptosis, but not other types of cell death, protected these cells, underscoring that their Achilles’ heel is an inability to contain oxidative damage.
Guiding Personalized Treatment Choices
Finally, the researchers moved into animal models and cells freshly derived from patients. Tumors with low VRK2 levels shrank when treated with a glutathione-targeting drug, while tumors with high VRK2 expression largely resisted it. However, when VRK2-high tumors were also given a VRK2-blocking compound, they regained sensitivity to glutathione inhibition. This suggests VRK2 levels can sort patients into groups: those whose tumors are naturally vulnerable to glutathione-targeted therapy, and those who might benefit from a two-pronged strategy that first disables VRK2, then attacks glutathione metabolism.
What This Means for Patients
Put simply, this work shows that some pancreatic cancers survive by using VRK2 to keep their antioxidant shield strong. Take away VRK2, and that shield thins; take away glutathione on top of that, and the cancer cells collapse under oxidative stress. Measuring VRK2 in tumors could therefore help doctors decide who is most likely to benefit from drugs that disrupt glutathione metabolism, and who might first need a VRK2-targeting step. While more testing in humans is needed, the study outlines a clear path toward more tailored, metabolism-based therapies for a cancer that desperately needs better options.
Citation: Chen, S., Fu, X., Zhang, T. et al. Vaccinia-related kinase 2 inhibition elicits vulnerability of glutathione metabolism in pancreatic cancer. Cell Death Dis 17, 325 (2026). https://doi.org/10.1038/s41419-026-08573-9
Keywords: pancreatic cancer, glutathione metabolism, VRK2, ferroptosis, SLC7A11