Clear Sky Science · en
Repression of EGFR by new biguanide 4C potentiated ovarian cancer to PARP inhibitors through down-regulation of BRCA2 and Rad51
Why this research matters
For many people diagnosed with ovarian cancer, today’s targeted drugs work only for a small minority whose tumors carry specific genetic flaws. This study explores a way to extend the benefits of an important class of medicines, called PARP inhibitors, to the much larger group of patients whose tumors lack those mutations. By pairing a new experimental compound with existing drugs, the researchers show a strategy to push cancer cells into a lethal dead end while sparing healthy tissue.
A roadblock in current ovarian cancer treatment
Ovarian cancer is often discovered late and remains one of the deadliest cancers affecting women. PARP inhibitors, such as olaparib, can be remarkably effective, but mainly in patients whose tumors already have defects in a DNA repair pathway tied to the BRCA1 and BRCA2 genes. Most tumors, however, still have intact repair machinery and can fix the DNA damage caused by these drugs, allowing cancer cells to survive. A key part of this repair crew involves two proteins, BRCA2 and Rad51, which help mend broken DNA strands through a process called homologous recombination. Finding ways to selectively weaken this repair pathway in cancer cells could make PARP inhibitors useful for far more patients.
The role of a well-known growth switch
The team focused on a familiar cancer-linked molecule, the epidermal growth factor receptor (EGFR), which sits on the surface of many tumor cells and drives their growth. They found that EGFR does more than send growth signals: in ovarian cancer cells with normal BRCA genes, high levels of EGFR were tied to poor patient survival and resistance to PARP inhibitors. When EGFR was reduced or shut down in cell cultures and in mice, tumors became much more vulnerable to olaparib. The researchers showed that EGFR helps usher BRCA2 and Rad51 into the cell nucleus after DNA damage, where they can repair breaks and blunt the impact of treatment. Simply blocking EGFR’s enzyme activity was not enough; the total amount of EGFR protein had to be lowered to significantly disrupt this repair pathway.
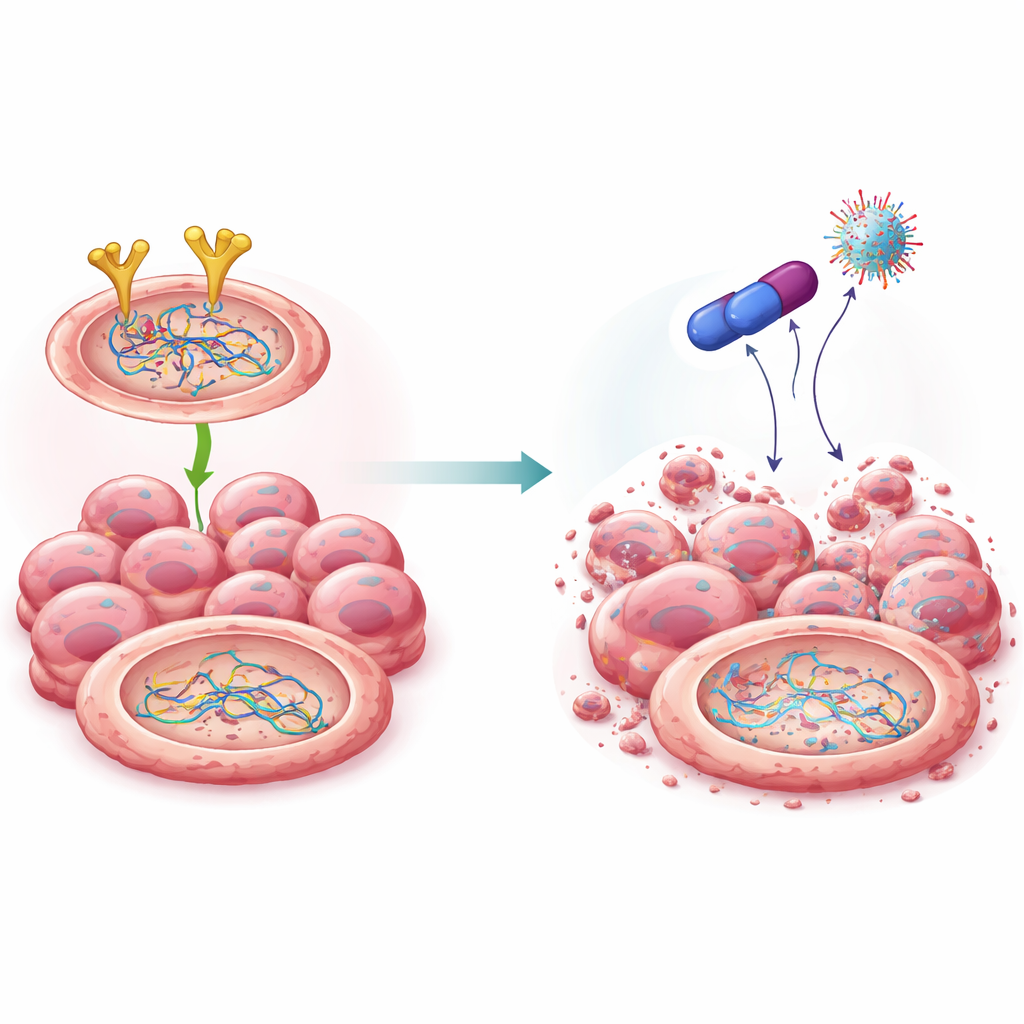
A new compound that disarms the repair crew
Building on earlier work with a drug family known as biguanides, the scientists synthesized several chemical cousins and identified one standout molecule, called 4C. This compound was strongly toxic to ovarian cancer cells yet relatively gentle on normal cells. Computer modeling and laboratory tests indicated that 4C binds directly to EGFR and marks it for destruction in the cell’s protein-shredding machinery. Unlike some existing EGFR drugs, 4C reduced the overall amount of EGFR rather than just silencing its activity. As EGFR levels fell, the stability of BRCA2 and Rad51 also dropped: they were tagged for disposal, broken down, and no longer able to support efficient DNA repair. Importantly, this pruning occurred at the protein level without altering the underlying genes.
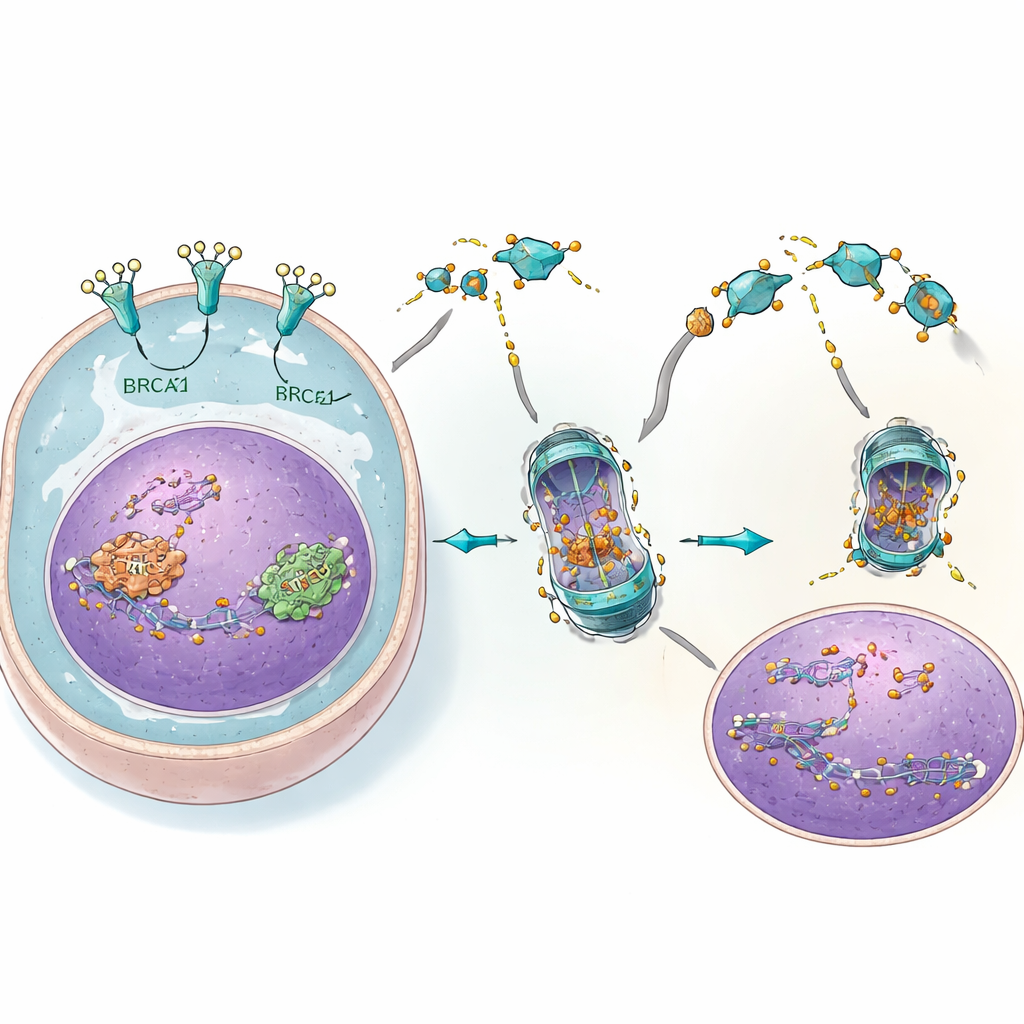
Blocking a rescue route inside the cell
The study uncovered a more detailed chain of events connecting DNA damage to tumor survival. After PARP inhibitors injure the DNA, another sensor protein called ATM sends a message from the nucleus out to the cell body. In response, EGFR teams up with BRCA2 and Rad51 and helps them move into the nucleus, where they repair the damage. The researchers discovered that a third protein, c-Cbl, normally acts as a kind of disposal tagger for BRCA2 and Rad51. EGFR competes with c-Cbl for access to these repair proteins, shielding them from being marked for destruction. When 4C lowers EGFR levels, c-Cbl can bind more easily to BRCA2 and Rad51, leading to their tagging, breakdown, and loss. With fewer repair proteins in the nucleus, DNA lesions accumulate and cancer cells become far more sensitive to PARP inhibitors.
From cells to animals: a potent pairing
In both cultured cells and mouse models, combining 4C with PARP inhibitors produced a powerful one-two punch. Tumors with normal BRCA genes that resisted either drug alone shrank dramatically or stopped growing when exposed to both together. Markers of DNA damage rose sharply, consistent with the idea that repair was being overwhelmed. At the same time, normal cells and key organs such as the liver and kidneys showed little evidence of harm, likely because they have much lower EGFR levels and rely less on this particular repair shortcut. The benefits of the combination extended to reducing the spread of ovarian cancer cells in the body.
What this could mean for patients
This work proposes a way to turn a cancer cell’s strength into a weakness. By targeting EGFR with the novel compound 4C, the researchers strip away the protection that BRCA2 and Rad51 provide, making tumors with otherwise normal DNA repair behave more like BRCA-mutant cancers in their response to PARP inhibitors. This enforced vulnerability, known as "synthetic lethality," could expand the reach of existing drugs to many more women with ovarian cancer, while keeping side effects manageable. Although 4C is still experimental and will require extensive testing in more advanced models and clinical trials, the study offers a clear blueprint for how dismantling specific repair helpers in tumor cells might improve treatment outcomes.
Citation: Xiao, D., Yao, J., Yang, X. et al. Repression of EGFR by new biguanide 4C potentiated ovarian cancer to PARP inhibitors through down-regulation of BRCA2 and Rad51. Cell Death Dis 17, 317 (2026). https://doi.org/10.1038/s41419-026-08556-w
Keywords: ovarian cancer, PARP inhibitors, EGFR, DNA repair, targeted therapy