Clear Sky Science · en
O-GlcNAcylation of YAP1 promotes lung transplant ischemia-reperfusion injury via binding to HIF1α transcription factor and activating autophagy and mitophagy
Why this matters for lung transplant patients
Lung transplantation can give people with severe lung disease a second chance at life, but the newly transplanted lung often suffers a wave of damage as blood flow is cut off and then restored. This so‑called ischemia–reperfusion injury can leave patients with fragile lungs, long stays in intensive care, and poorer long‑term outcomes. The study behind this article uncovers a hidden molecular chain reaction inside lung cells that helps drive this early injury, pointing to new drug targets that might one day make lung transplants safer and more successful.
A chain reaction sparked by lost oxygen
During a transplant, the donor lung is temporarily deprived of blood and oxygen, then suddenly re-exposed when it is connected to the recipient’s circulation. The researchers modeled this stop–start in oxygen supply in lung and blood-vessel lining cells in the lab, and in a rat lung transplant system. They saw that this stress strongly switched on a growth-and-survival control system inside cells called the Hippo–YAP pathway. When activated, the YAP1 protein moves into the nucleus and teams up with other factors to turn many genes on or off. In this study, hypoxia followed by reoxygenation made YAP1 and its partner genes much more active, and this surge was tightly linked to cell death and the release of inflammatory molecules that can inflame and weaken the transplanted lung.
Self-cleaning gone too far
Cells rely on an internal “self-cleaning” system known as autophagy to break down and recycle worn-out components, and on a more focused version, mitophagy, to cull damaged mitochondria, the tiny power plants of the cell. In moderate amounts, these processes help cells cope with stress. But here, the team found that after oxygen was cut off and restored, both autophagy and mitophagy were turned up sharply in lung cells and in transplanted lungs. Under the microscope, they saw more recycling vesicles and signs of mitochondria being engulfed and digested. At the same time, animals showed swelling and structural damage in lung tissue, along with more cells undergoing programmed death. This suggested that, in the transplant setting, the self-cleaning machinery is pushed into overdrive and starts to contribute to injury instead of protection.
A sugar tag that changes protein behavior
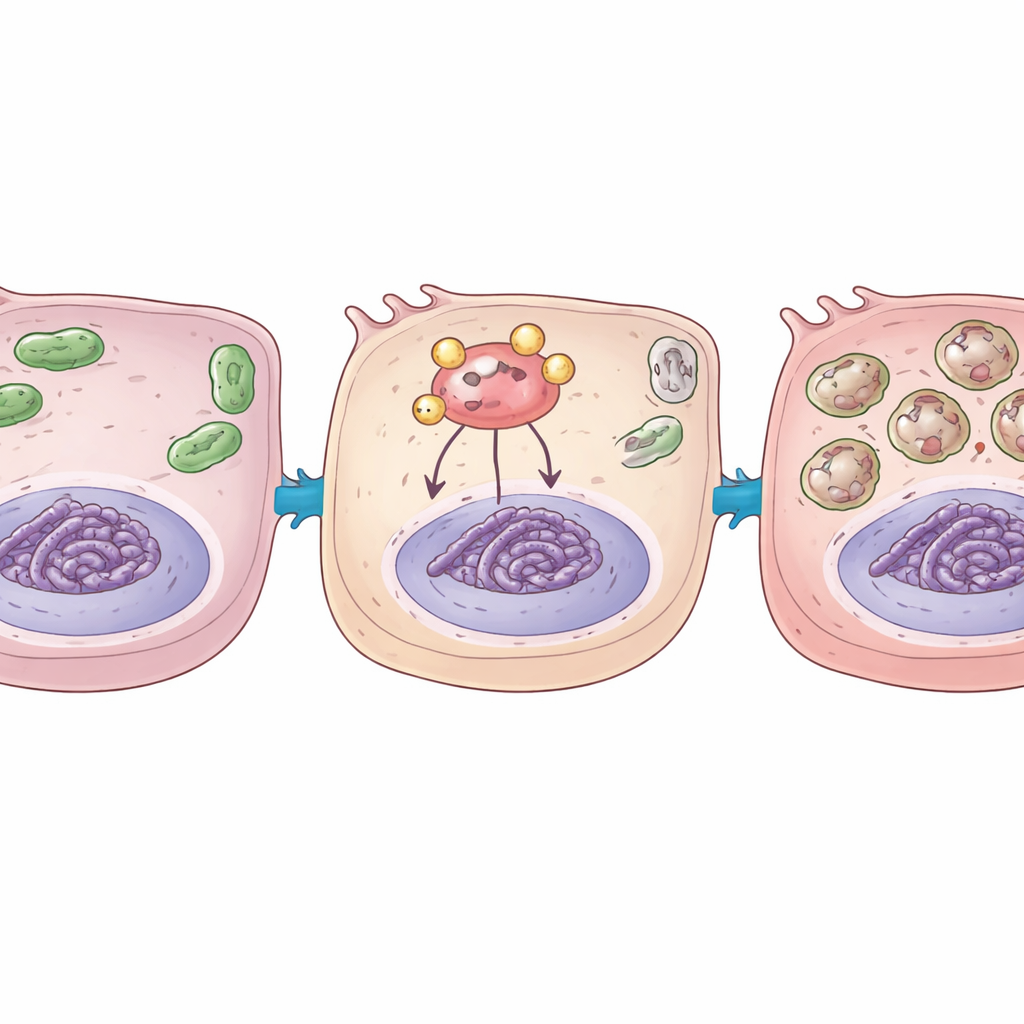
The scientists then asked why YAP1 became so damaging under these conditions. They focused on a small sugar-based tag called O-GlcNAc that can be attached to many proteins and alter how they behave. This tag is added by an enzyme called OGT. They discovered that oxygen deprivation and restoration increased both overall O-GlcNAc tagging in cells and, more specifically, the tagging of YAP1. When YAP1 carried this sugar tag, it bound more tightly to another key oxygen-sensing protein, HIF1α, which normally helps cells adapt to low oxygen by switching on protective genes. Using biochemical tests, the team showed that tagged YAP1 pulled HIF1α to stretches of DNA controlling genes that drive autophagy and mitophagy, boosting their activity and further amplifying the cell’s recycling and stress responses.
Switching off the damaging loop
To test whether this pathway could be tamed, the researchers used genetic tools to dial down either YAP1 itself or OGT, the enzyme that adds the sugar tag. In cell models, reducing YAP1 weakened activation of the stress pathway, cut back excessive autophagy and mitophagy, and lessened cell death after oxygen was restored. In transplanted rat lungs, knocking down YAP1 or OGT reduced tissue swelling, lowered markers of self-digestion and mitochondrial breakdown, and left fewer cells undergoing programmed death. Importantly, blocking OGT also reduced the sugar tagging of YAP1 and its ability to recruit HIF1α to autophagy-related genes, directly weakening the harmful loop the team had uncovered.
What this means for future transplants
Taken together, the study shows that a small chemical tag on the YAP1 protein can turn a normal stress-response network into a driver of lung damage after transplantation. By helping YAP1 cling to the oxygen sensor HIF1α and crank up cellular self-digestion beyond healthy levels, this tag promotes swelling, structural damage, and cell loss in the new lung. The findings suggest that drugs aimed at dampening YAP1 activity, blocking its sugar tagging by OGT, or carefully modulating autophagy and mitophagy could offer new ways to protect transplanted lungs from early injury, improving both survival and quality of life for recipients.
Citation: Dai, S., Wan, X., Xia, L. et al. O-GlcNAcylation of YAP1 promotes lung transplant ischemia-reperfusion injury via binding to HIF1α transcription factor and activating autophagy and mitophagy. Cell Death Dis 17, 311 (2026). https://doi.org/10.1038/s41419-026-08548-w
Keywords: lung transplantation, ischemia reperfusion injury, YAP1, autophagy, mitophagy