Clear Sky Science · en
Androgen receptor-dependent DRAM1 activation drives autophagic resistance to BRAF inhibitors in BRAFV600-mutant melanoma
Why some skin cancers outsmart powerful drugs
Targeted drugs have transformed treatment for many patients with advanced melanoma, an aggressive form of skin cancer. Yet these therapies often stop working after months, as tumors adapt and begin to grow again. This study uncovers a hidden survival trick used by melanoma cells: they switch on a hormone sensor more common in male biology and use the cell’s own recycling system to dodge the effects of treatment. Understanding this escape route could open the door to smarter combinations of medicines that keep the cancer in check for longer.
A closer look at drug-resistant melanoma
About half of melanomas carry a specific DNA change in a gene called BRAF, which makes tumor cells grow faster. Drugs that block BRAF, often paired with related MEK blockers, can shrink these tumors dramatically. Unfortunately, many patients see their disease return within two years. Doctors have also noticed that men tend to respond less well to these drugs than women, raising questions about the role of sex-related hormones and their receptors in driving resistance.
A male hormone sensor steps into the spotlight
The researchers focused on the androgen receptor, a protein inside cells that normally responds to male sex hormones such as testosterone. Even though melanoma is not a classic “hormone-driven” cancer, earlier work showed that this receptor can make melanoma cells more aggressive. In this study, the team exposed multiple melanoma cell lines to BRAF and MEK inhibitors and found that the drugs themselves pushed cells to produce more androgen receptor, regardless of whether the original tumor came from a male or female patient. Cells engineered to carry extra copies of this receptor survived BRAF-blocking drugs much better and formed more colonies, while real patient tumor samples taken after treatment showed higher receptor levels than samples from the same patients before therapy.
Turning on the cell’s recycling and clean-up system
The team then asked how the androgen receptor helps melanoma cells withstand treatment. They focused on autophagy, the cell’s built‑in clean‑up and recycling process, which can either help cells die or help them survive stress, depending on context. Using microscopes, fluorescent reporters, and protein markers, they showed that boosting the androgen receptor in melanoma cells strongly increased the formation and turnover of the tiny sacs and digestion chambers that carry out autophagy. Blocking this recycling pathway with a chemical inhibitor made receptor‑rich cells much more sensitive to BRAF-blocking drugs, while activating autophagy could partially rescue cells in which the receptor had been silenced. Together, these experiments showed that the receptor supports drug resistance by dialing up a protective form of cellular self‑cleaning.
A key switch called DRAM1 links hormones to recycling
To pinpoint which genes connect the androgen receptor to the recycling machinery, the researchers mined several large gene‑expression datasets. One gene, DRAM1, stood out as both strongly induced by the receptor and consistently elevated in cells and tumors exposed to BRAF inhibitors. DRAM1 encodes a protein embedded in the membranes of recycling and digestion compartments inside the cell. When the team forced melanoma cells to make more DRAM1, autophagy increased; when they reduced DRAM1 levels, autophagy and drug resistance both fell. They also showed that the androgen receptor binds directly to specific control sequences in the DRAM1 gene and switches it on, establishing a clear chain from drug exposure to receptor increase to DRAM1 activation and heightened recycling.
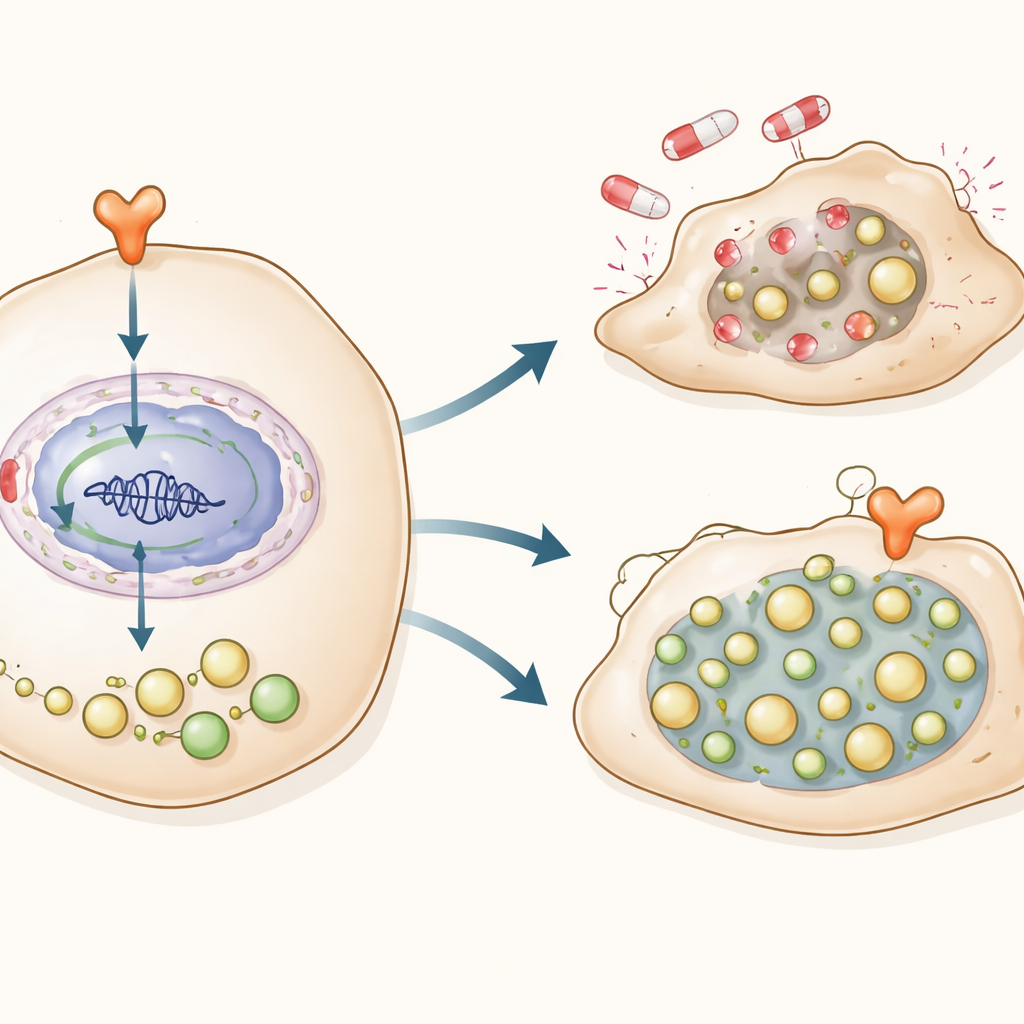
Combining two hits to block the cancer’s escape
Because this hormone‑recycling axis appeared central to resistance, the scientists tested whether blocking both arms at once would be especially effective. In cell cultures, they treated BRAF‑resistant melanoma cells with a modern degrader drug (ARV110) that removes the androgen receptor, together with an autophagy inhibitor. The combination sharply reduced the survival of resistant cells compared with either drug alone. In mice carrying resistant human melanoma tumors, the same dual treatment shrank tumors more and slowed their growth for longer, without obvious damage to major organs or loss of body weight. Tumors from treated mice showed reduced levels of the receptor, DRAM1, and recycling markers, confirming that the pathway had been successfully shut down.
What this means for people with melanoma
These findings reveal that BRAF‑targeted therapies can unintentionally activate a male‑hormone sensor in melanoma cells, which then flips on a recycling program through DRAM1 to help tumors survive. To a non‑specialist, this is like giving the cancer a powerful drug while the tumor quietly builds a better waste‑processing and repair system that lets it endure the attack. By cutting off both the hormone sensor and the recycling machinery at the same time, researchers were able to make resistant tumors vulnerable again in preclinical models. While more work is needed to confirm safety and benefit in patients, this study suggests that carefully designed combinations targeting hormone signaling and cell recycling could one day extend the power and duration of current melanoma treatments.
Citation: Zhi, D., Wu, B., Yang, J. et al. Androgen receptor-dependent DRAM1 activation drives autophagic resistance to BRAF inhibitors in BRAFV600-mutant melanoma. Cell Death Dis 17, 265 (2026). https://doi.org/10.1038/s41419-026-08547-x
Keywords: melanoma, drug resistance, androgen receptor, autophagy, BRAF inhibitors