Clear Sky Science · en
Long noncoding RNA ADEI/miR-93-3p/STAT3 axis promotes Epstein–Barr virus-positive diffuse large B-cell lymphoma progression and immune evasion through regulating the PD-1/PD-L1 checkpoint
Why this matters for our immune defenses
Some lymphomas linked to the common Epstein–Barr virus (EBV) are unusually hard to treat and often escape the body’s immune defenses. This study uncovers a hidden messaging system inside these cancers: tiny bubbles called exosomes carrying a long RNA molecule that helps tumors grow and hide from immune cells. Understanding this covert conversation could open new ways to diagnose and treat these stubborn blood cancers.
A virus that tilts the balance
Diffuse large B-cell lymphoma (DLBCL) is the most common aggressive lymphoma. When tumor cells carry EBV, patients tend to respond worse to standard chemo‑immunotherapy. The researchers first compared EBV‑positive and EBV‑negative lymphoma cells in the lab. They found that EBV infection made lymphoma cells divide faster, form more colonies, and grow larger tumors in mice. EBV‑positive cells also displayed more of a surface molecule called PD‑L1, which interacts with PD‑1 on immune cells to switch off their attack. When the team mixed lymphoma cells with killer CD8 T cells, EBV‑positive tumors shrank the number and potency of these T cells, an effect that could be reversed with antibodies that block the PD‑1/PD‑L1 brake.
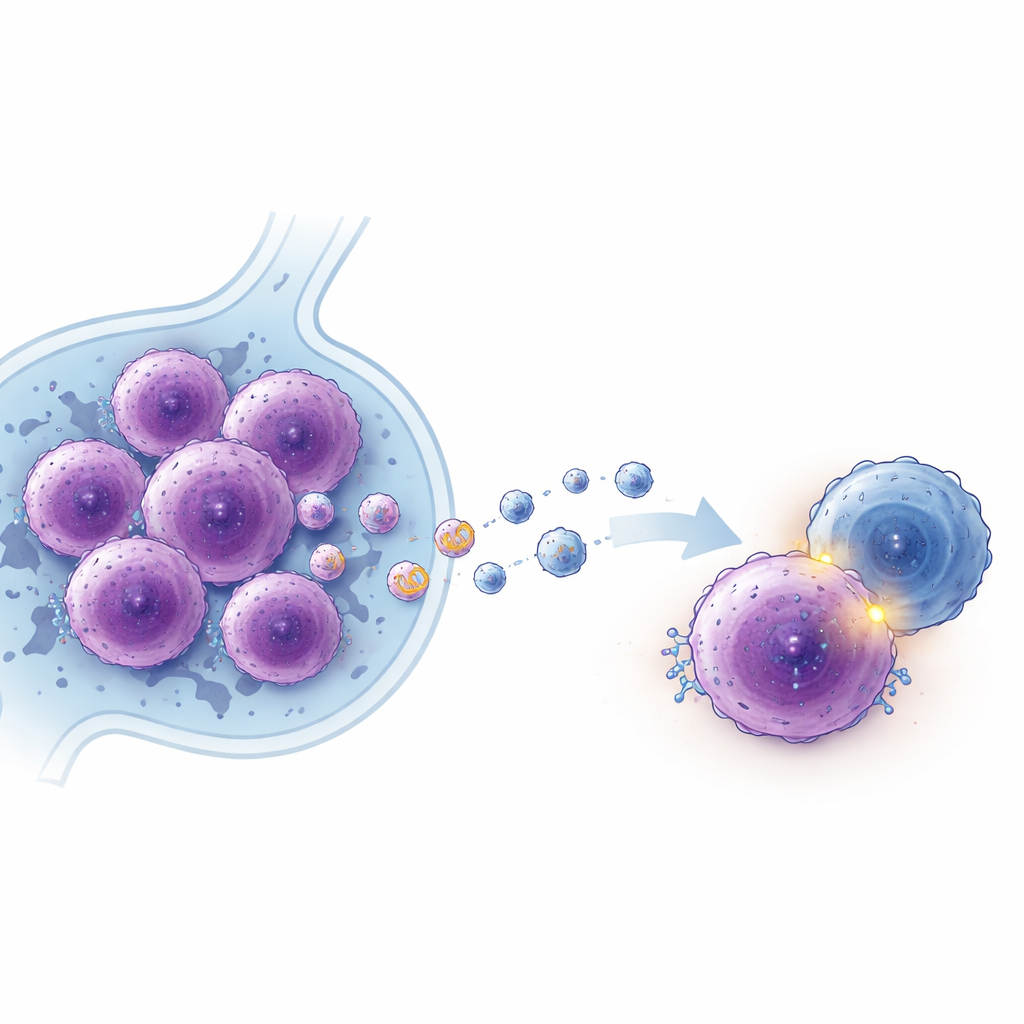
A long RNA that helps tumors grow and hide
To understand how EBV reshapes tumor behavior, the team looked at long noncoding RNAs—stretches of RNA that do not make proteins but can regulate many cell processes. By scanning the RNA content of exosomes released from EBV‑positive and EBV‑negative DLBCL cells, they discovered a previously uncharacterized RNA, which they named lncADEI, that was strongly increased in EBV‑positive cells and their exosomes. When they forced lymphoma cells to make more lncADEI, the cells proliferated faster, formed more colonies, and resisted programmed cell death. Reducing lncADEI had the opposite effect, slowing growth and increasing cell death both in dishes and in mouse tumor models.
How lncADEI rewires immune escape
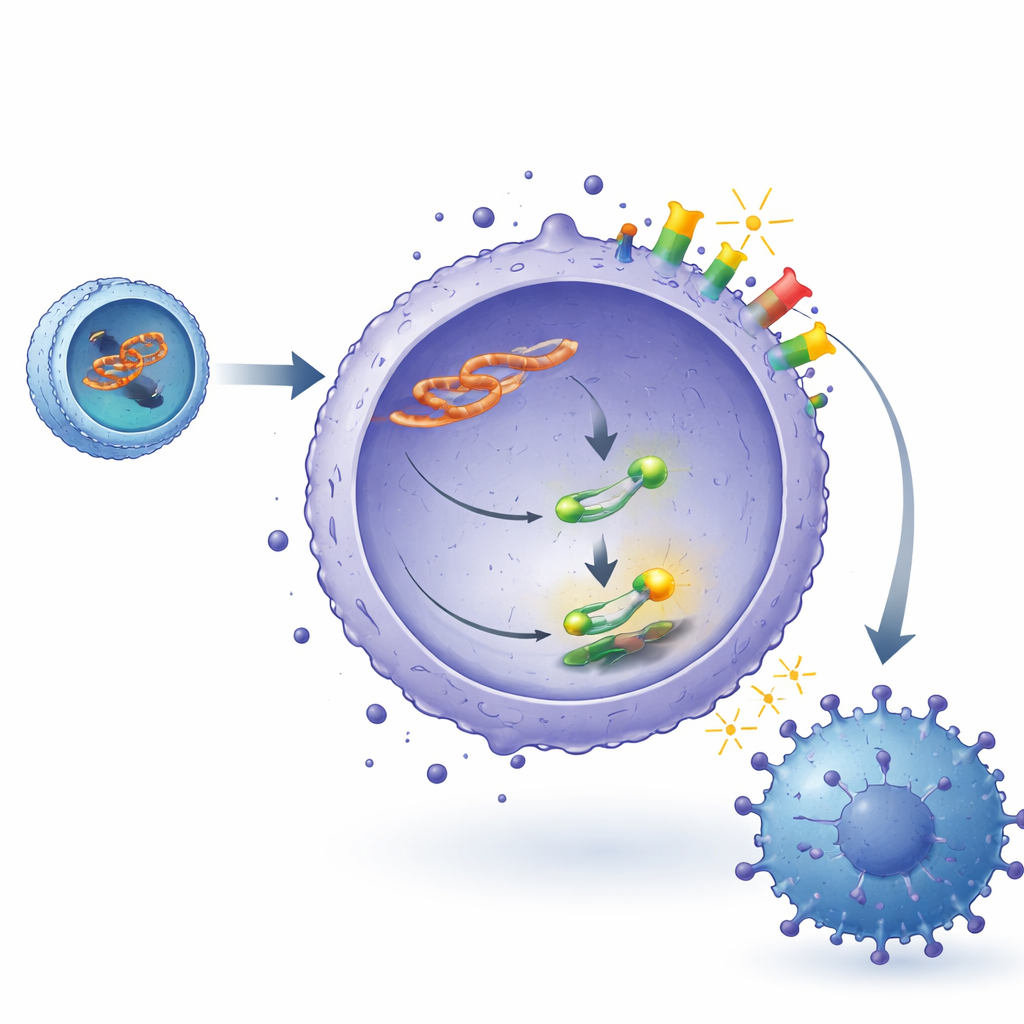
The researchers then traced how lncADEI connects to the PD‑1/PD‑L1 immune checkpoint. Inside lymphoma cells, lncADEI sits mainly in the cytoplasm, where it acts like a sponge for a small regulatory RNA called miR‑93‑3p. Under normal conditions, miR‑93‑3p helps keep in check a key signaling protein, STAT3, by binding to its message and limiting its production. When lncADEI soaks up miR‑93‑3p, more STAT3 is made and activated. STAT3, in turn, binds directly to the control region of the PD‑L1 gene in lymphoma cells and boosts PD‑L1 production. This chain—lncADEI blocking miR‑93‑3p, freeing STAT3, and driving PD‑L1—results in higher PD‑L1 on tumor cells, stronger shutdown signals to CD8 T cells, and reduced T‑cell activity.
Exosome “mail” that spreads bad instructions
EBV‑positive lymphoma cells do not keep lncADEI to themselves. They secrete many more exosomes than EBV‑negative cells, and these exosomes are packed with lncADEI. When EBV‑negative lymphoma cells were exposed to exosomes from EBV‑positive cells, they took them up, their internal lncADEI levels rose, and they began to behave more aggressively—dividing faster and forming more colonies. In mixed cultures that included T cells, exosomes enriched in lncADEI reduced the number and killing power of CD8 T cells and increased PD‑1 on these immune cells, further weakening the immune response. This suggests that EBV‑positive tumors can “educate” neighboring tumor cells and remodel their surroundings through lncADEI‑rich exosomes.
Clues from patient blood samples
To link these lab findings to real disease, the team measured lncADEI in blood exosomes from 47 patients with DLBCL. Those whose tumors were EBV‑positive had significantly higher levels of exosomal lncADEI than EBV‑negative patients. High lncADEI in blood was also associated with more advanced disease stage, more aggressive tumor subtype, higher standard risk scores, and elevated markers of tissue damage. These patterns suggest that exosomal lncADEI in blood could serve as a minimally invasive indicator of EBV‑driven lymphoma and its severity.
What this means for future care
In everyday terms, this work reveals a three‑step trick used by EBV‑linked lymphomas: they overproduce a long RNA (lncADEI), pack it into traveling bubbles, and use it both inside the tumor and in neighboring cells to crank up a well‑known immune brake, PD‑L1. The result is faster tumor growth and a muted T‑cell attack. Because each step in this chain—lncADEI itself, its interaction with miR‑93‑3p and STAT3, and the release of lncADEI‑rich exosomes—offers a possible point of intervention or measurement, the study points toward new strategies to improve immunotherapy and to monitor EBV‑associated lymphomas using a simple blood test.
Citation: Zheng, W., Lai, G., Liao, Z. et al. Long noncoding RNA ADEI/miR-93-3p/STAT3 axis promotes Epstein–Barr virus-positive diffuse large B-cell lymphoma progression and immune evasion through regulating the PD-1/PD-L1 checkpoint. Cell Death Dis 17, 280 (2026). https://doi.org/10.1038/s41419-026-08532-4
Keywords: Epstein–Barr virus lymphoma, immune checkpoint, long noncoding RNA, exosomes, STAT3 signaling