Clear Sky Science · en
ISGylation prevents autophagic degradation of STING and promotes antitumor immunity in lung cancer
Why Protecting a Cell Alarm Matters for Lung Cancer
Our immune system carries tiny alarm switches that can warn the body when something is wrong, such as a virus infection or a growing tumor. One of these alarms, called STING, helps turn quiet tumors into targets the immune system can attack. But in many lung cancers, the amount of STING inside tumor cells drops over time, which can blunt the impact of new cancer drugs that try to turn this alarm on. This study uncovers why STING disappears and shows a way to keep it stable, opening a path to make immunotherapy work better in patients whose tumors currently respond poorly.
How Cells Sense Danger Inside
When pieces of DNA appear where they do not belong inside a cell, it is often a sign of viral infection or damaged cancer cells. The cell uses a detection system known as the cGAS–STING pathway to sense this misplaced DNA. Once activated, STING triggers a wave of signals that leads to the production of type I interferons, powerful immune messengers. These messengers rally several types of defenders, including natural killer cells and killer T cells, to attack infected or malignant cells. However, in many tumors, especially advanced lung cancers, STING levels are unusually low, weakening this internal alarm system and helping cancer cells hide.
A Protective Tag That Saves STING
Proteins inside cells are constantly being marked, moved, and destroyed. The authors focused on a specific kind of molecular tag called ISGylation, in which a small protein, ISG15, is attached to other proteins. They discovered that attaching ISG15 to STING at four particular positions acts like a protective shield. This shield prevents STING from being swallowed by the cell’s recycling machinery, a self-cleaning process called autophagy that normally helps dispose of used or excess components. In lung cancer cells, when STING cannot receive these ISG15 tags, it is cleared away faster, its alarm signal fades, and the downstream immune response — including interferon production — drops sharply. 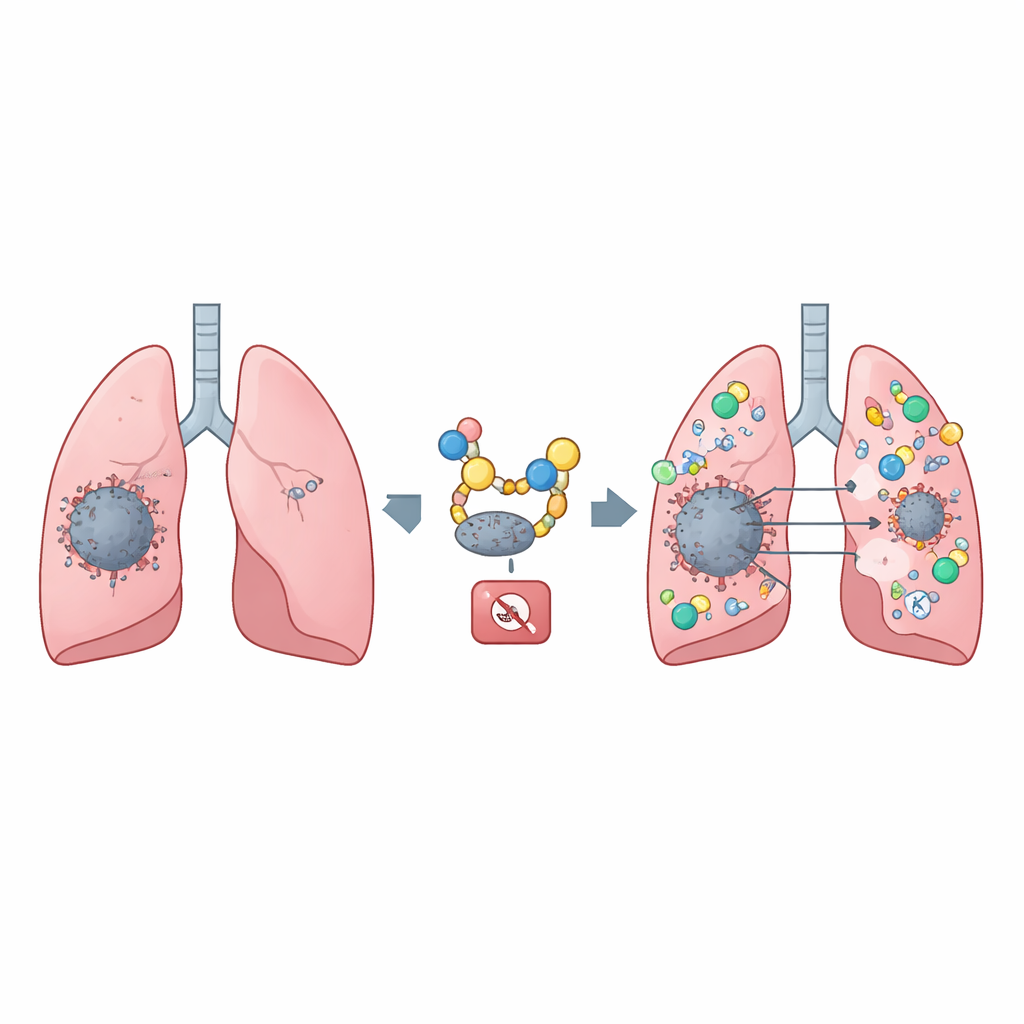
Blocking the Cellular Shredder
The team then asked which molecules remove this protective tag. They identified an enzyme, USP18, that clips ISG15 off STING, exposing it to autophagic breakdown. In cell experiments and mouse lung tumor models, higher USP18 levels meant less STING, reduced immune cell entry into tumors, and faster cancer growth. Human lung cancer samples told a similar story: USP18 was often elevated in tumors, while STING was reduced, and patients with higher USP18 tended to have poorer survival. These patterns suggest that USP18 acts like a cellular shredder switch, dialing down the STING alarm at the wrong time in cancer.
A Drug Combination that Supercharges Immune Attack
Because enzymes like USP18 can be blocked by small molecules, the researchers screened thousands of compounds and pinpointed Tanshinone IIA sulfonate (TST) as a direct USP18 inhibitor. In the lab, TST prevented USP18 from stripping ISG15 off STING, extended STING’s life inside cells, and boosted its signaling. In mice bearing lung tumors, TST slowed tumor growth. When the team combined TST with a separate drug that directly activates STING, called diABZi, the effect was striking: tumors shrank more, and immune cells — especially natural killer cells and killer T cells — flooded into the cancer. This combination created a stronger and more sustained immune assault than either treatment alone. 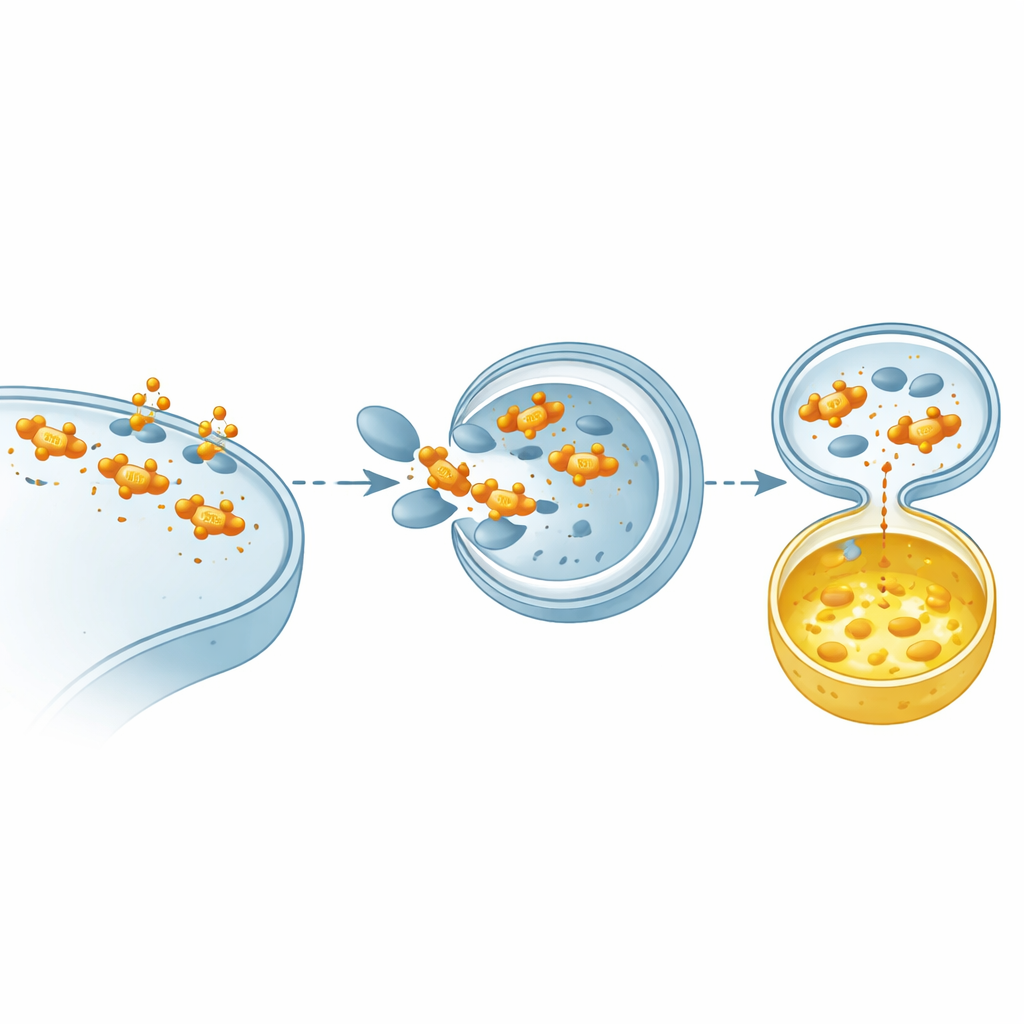
What This Means for Future Lung Cancer Treatment
Put simply, this work shows that lung tumors often silence a key internal alarm, STING, not only by making less of it, but also by speeding up its destruction. A natural protective tag, ISG15, keeps STING from being swept into the cell’s recycling system, while USP18 removes that tag and promotes STING loss. By identifying a drug that blocks USP18 and preserves STING, and by pairing it with a STING-activating drug, the study outlines a strategy to turn quiet, low-STING tumors into highly visible targets for the immune system. For patients whose cancers currently fail to respond to STING-based therapies, stabilizing STING itself could make the difference between a muted response and a potent, lasting antitumor attack.
Citation: Cao, D., Huang, B., Fu, X. et al. ISGylation prevents autophagic degradation of STING and promotes antitumor immunity in lung cancer. Cell Death Dis 17, 271 (2026). https://doi.org/10.1038/s41419-026-08527-1
Keywords: STING pathway, ISGylation, lung cancer immunotherapy, USP18 inhibition, autophagy