Clear Sky Science · en
Nuclear Myosin 1 links genomic architecture to adipose tissue remodeling, metabolic inflammation and obesity in mice
Why the Cell’s Skeleton Matters for Body Fat
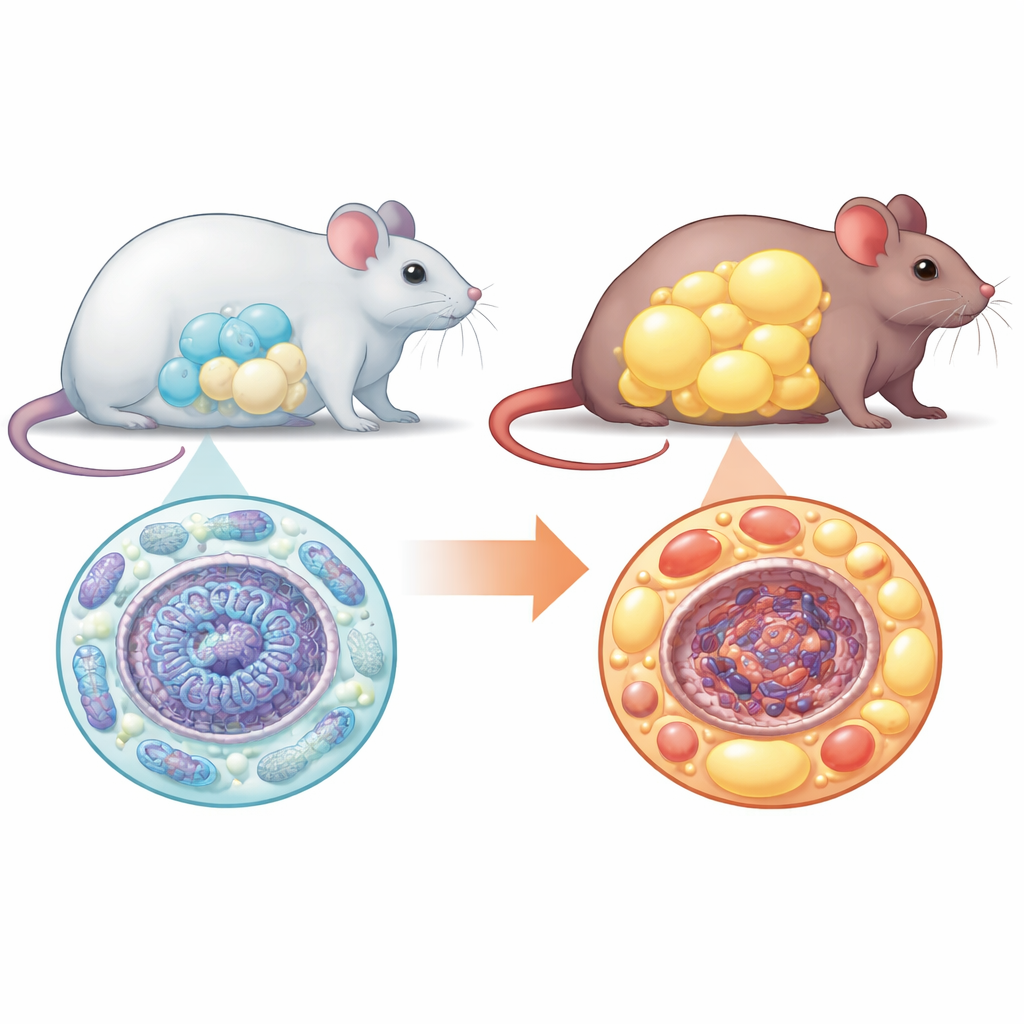
Obesity is often blamed on what we eat and how much we move, but deep inside our cells, the way DNA is packed and read can also tip the scales. This study reveals that a tiny motor protein, nuclear myosin 1 (NM1), which helps organize DNA inside the cell nucleus, plays a surprising role in how fat cells form, store energy, and trigger inflammation. By studying mice lacking this protein, the researchers uncovered a direct link between the physical architecture of our genes and the development of unhealthy visceral fat and metabolic problems.
From Stem Cells to Fat Cells
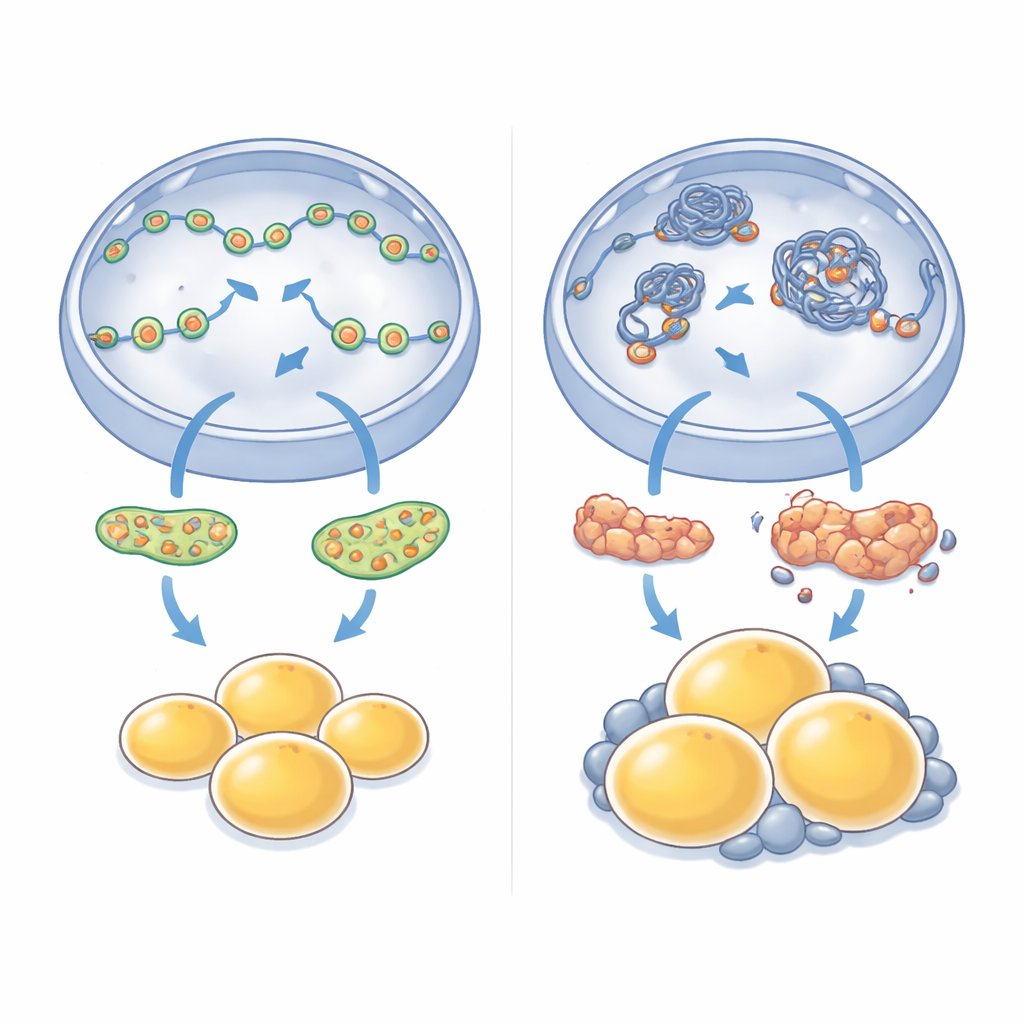
Fat cells develop from versatile precursors called mesenchymal stem cells. Normally, these precursors multiply, switch their energy use, and mature into many small, well-functioning fat cells that safely store excess calories. The team found that without NM1, this orderly process breaks down. When they grew stem cells from normal and NM1-deficient mice in the lab and pushed them to become fat cells, far fewer cells completed the transition in the absence of NM1. The cells that did mature were noticeably larger than normal, packed with fat but missing the usual signature of healthy fat cells at the gene and protein level. This pattern – fewer cells but each one enlarged – is a hallmark of unhealthy fat tissue.
How Gene Packing Shapes Fat Cell Behavior
NM1 works in the nucleus, where it helps keep stretches of DNA accessible so that important genes can be turned on. To understand how its loss changes cell behavior, the researchers overlaid maps of open DNA regions with measurements of gene activity in cells with and without NM1. They saw that many genes needed for fat cell formation, fat droplet handling, and mitochondrial upkeep lost both accessibility and activity when NM1 was missing. At the same time, other genes tied to growth and signaling pathways became more open and more active. This pointed to NM1 as a key traffic controller: it helps maintain a balanced landscape of open and closed DNA regions so that the right networks of genes drive healthy fat cell development and metabolism.
When Local Changes Become Whole-Body Weight Gain
The consequences of losing this single nuclear motor did not stay confined to cells in a dish. Mice lacking NM1 gradually became heavier than their normal littermates, even though they did not eat more food. Scans and tissue analysis showed a clear buildup of fat, especially around internal organs in the abdomen, and the individual fat cells in these areas were strikingly enlarged. When the researchers measured which genes were turned up or down in this visceral fat, they found widespread changes: pathways involved in energy use, insulin signaling, and tissue remodeling were reprogrammed. Genes associated with fat cell growth and expansion were often boosted, while several central controllers of healthy fat cell formation were dampened, mirroring the defects seen in cultured stem cells.
Inflamed Fat and Disturbed Energy Engines
Beyond sheer size, the fat tissue in NM1-deficient mice showed signs of being inflamed and metabolically stressed. A detailed pathway analysis highlighted strong activation of immune signals driven by molecules such as interferon-gamma and tumor necrosis factor, which are known to contribute to insulin resistance and metabolic disease. At the same time, regulators that support calm, energy-efficient fat tissue were suppressed. The study also connected NM1 loss to disturbed mitochondria – the cell’s energy engines. Genes that normally oversee mitochondrial quality control and the shift between efficient energy production and quick, glycolytic energy were misregulated, suggesting that when NM1 is absent, fat cells adopt a less efficient, more stress-prone energy strategy.
Shared Signals Between Mice and Humans
To ask whether this nuclear motor pathway might matter in people, the authors examined large genetic datasets from human visceral fat. They focused on a human relative of NM1 called MYO1C and looked at networks of variants that tune the activity of nearby genes. They found that many of the same themes seen in mice – cytoskeletal control, immune signaling, and metabolic regulation – appeared in human gene networks centered on MYO1C. Several genes altered in NM1-deficient mouse fat were also embedded in these human networks and have been linked to fat tissue remodeling and metabolism. This cross-species overlap suggests that similar nuclear mechanisms could influence obesity risk in humans.
What This Means for Obesity and Metabolic Health
Taken together, the work shows that NM1 is much more than a nuclear helper; it is a central hub tying DNA organization to how fat cells grow, store energy, and communicate with the immune system. When NM1 is missing, gene switches in fat precursors are mis-set, mitochondria function poorly, fat cells enlarge instead of multiplying, and visceral fat becomes inflamed – a combination that resembles the unhealthy fat seen in obesity and insulin resistance. For a lay reader, the message is that the physical arrangement of DNA and its helpers inside the nucleus can predispose tissues toward healthy or harmful fat expansion. Proteins like NM1 and its human counterpart MYO1C may one day become targets for therapies aimed at reshaping fat tissue from the inside out, offering a more precise way to tackle obesity-related diseases.
Citation: Khalaji, S., Venit, T., Lukáčová, Z. et al. Nuclear Myosin 1 links genomic architecture to adipose tissue remodeling, metabolic inflammation and obesity in mice. Cell Death Dis 17, 270 (2026). https://doi.org/10.1038/s41419-026-08525-3
Keywords: adipogenesis, chromatin, mitochondria, visceral fat, metabolic inflammation