Clear Sky Science · en
M2 macrophages promote lymphatic metastasis by regulating PKM2 nuclear translocation in triple-negative breast cancer
Why this research matters
For people diagnosed with triple-negative breast cancer, one of the biggest worries is that the cancer will spread to nearby lymph nodes, a key step toward spreading throughout the body. This study digs into how certain immune cells that should protect us instead help this kind of breast cancer invade the lymph system, and identifies a molecular "switch" that could be targeted with drugs to slow or stop this spread.
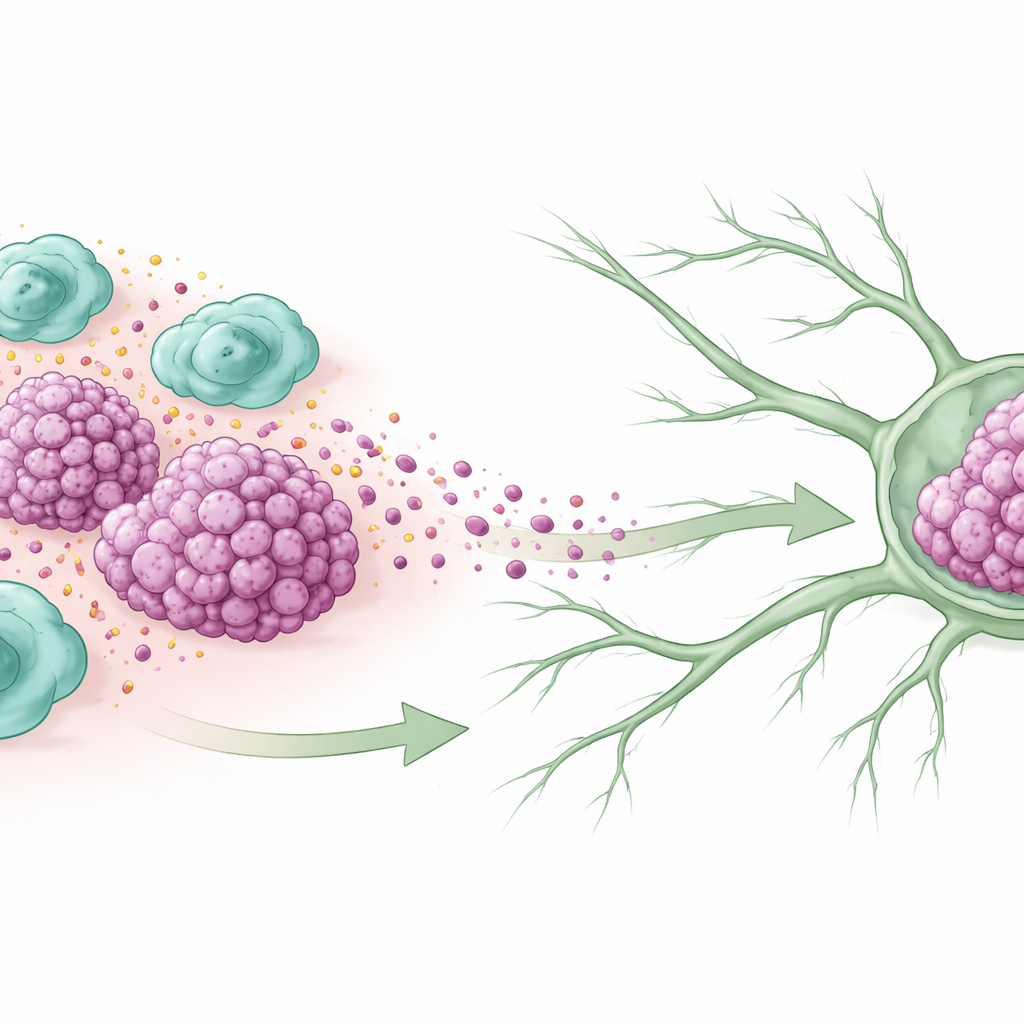
Immune helpers turned accomplices
Inside many tumors, immune cells called macrophages gather in large numbers. Some of them adopt a nurturing, wound-healing mode known as the M2 state, which in cancer often ends up helping tumors instead of fighting them. The researchers examined tissue from women with triple-negative breast cancer and found that tumors which had already spread to lymph nodes contained many more of these M2-type macrophages. Patients whose tumors were packed with these cells tended to have larger cancers, more aggressive features, and shorter survival, suggesting that these “friendly fire” immune cells are closely linked to cancer worsening.
Building new lymph “highways” for cancer cells
Cancer cells usually do not reach lymph nodes by chance; they often travel along newly formed lymphatic vessels, tiny channels that drain fluid from tissues. In this study, tumors rich in M2 macrophages also had more of these lymph vessels woven through them. In lab dishes, when breast cancer cells were grown in the presence of M2 macrophages, they released signals that made human lymphatic cells form tube-like networks and migrate more quickly, mimicking the growth of new lymph channels. In mice, cancer cells pre-exposed to M2 macrophages produced more lymph vessels in the tumor and more frequent metastases to nearby lymph nodes, confirming that these immune cells actively help construct escape routes.
The hidden signal chain inside cancer cells
The team then asked what chemical messages M2 macrophages send to push tumors toward lymphatic spread. They pinpointed a well-known molecule, TGF-β, released in higher amounts by M2 macrophages. This signal caused breast cancer cells to ramp up production of two growth factors, VEGFC and VEGFD, which are powerful drivers of lymph vessel growth. Crucially, the researchers showed that a metabolic enzyme inside cancer cells, PKM2, acts as a central switch in this process. Under TGF-β stimulation, PKM2 not only boosts the cell’s sugar-burning machinery but also becomes chemically modified and moves into the nucleus, where it helps turn on the genes for VEGFC and VEGFD. Without PKM2, or when its movement into the nucleus was blocked, cancer cells produced far less of these lymph-growing signals and were less able to stimulate lymph vessel formation.
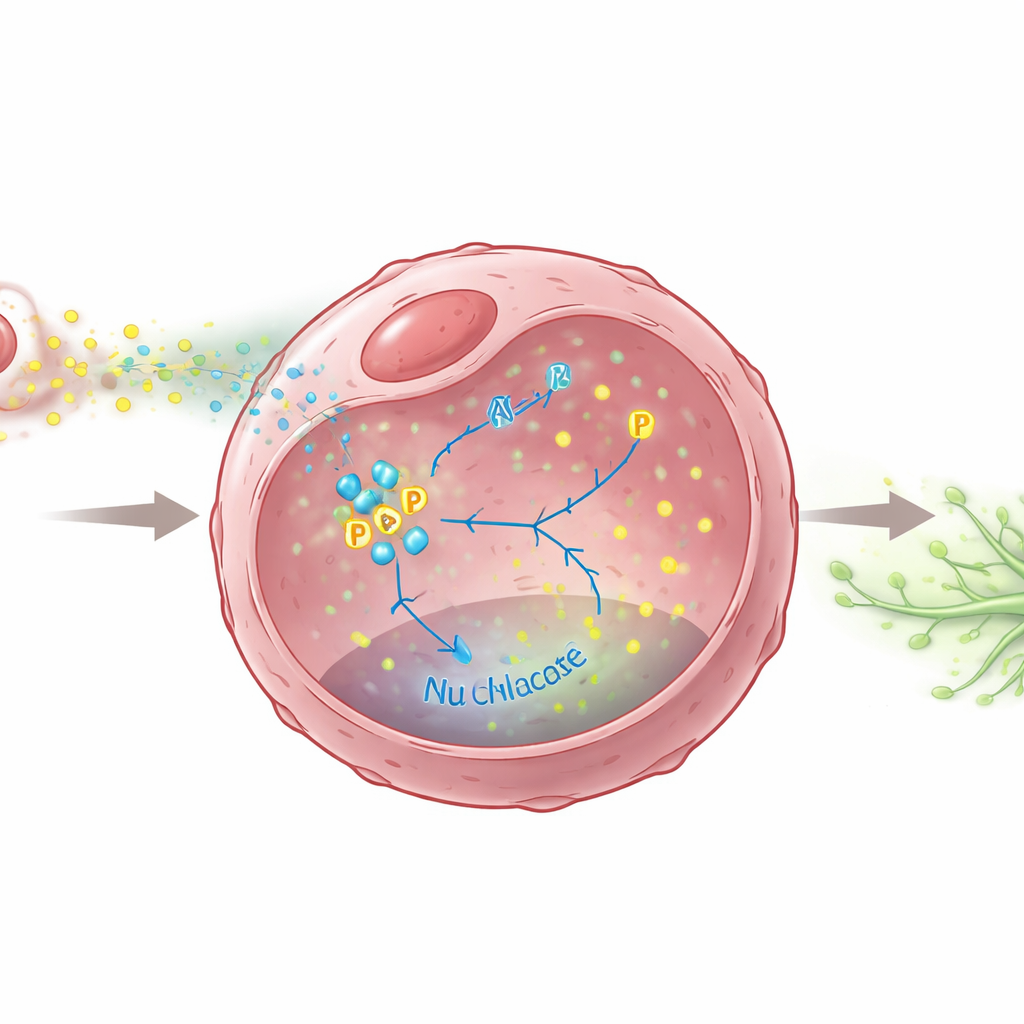
Testing a possible brake on spread
Because PKM2 sits at this crucial junction between altered metabolism and pro-metastatic signaling, the scientists tested whether shutting it down could slow lymphatic spread. They used shikonin, a compound that interferes with PKM2’s activation and nuclear entry. In cell experiments, shikonin sharply reduced VEGFC and VEGFD levels and weakened the ability of cancer-cell secretions to induce lymph vessel growth and movement. In mouse models, treatment with shikonin cut down the number and size of lymph node metastases and reduced the density of lymph vessels inside tumors, without obvious toxic effects on the animals. These findings suggest that blocking PKM2 may simultaneously starve cancer cells of some metabolic advantages and blunt their ability to remodel the lymph network in their favor.
What this means for patients
Altogether, the study proposes a clear chain of events: M2 macrophages in triple-negative breast tumors secrete TGF-β, which flips PKM2 inside cancer cells into a dual role—fueling high sugar use and acting in the nucleus to switch on VEGFC and VEGFD. These factors then stimulate the growth of lymph vessels that cancer cells use to reach lymph nodes and beyond. By identifying PKM2 as a critical hub in this chain, the work suggests new ways to predict and potentially treat lymphatic spread, especially in patients whose tumors show high levels of M2 macrophages, PKM2, or VEGFC/D. While more testing in humans is needed, targeting this pathway could help turn the lymphatic system from a cancer highway back into a barrier.
Citation: Yang, Y., Ye, H., Zhong, D. et al. M2 macrophages promote lymphatic metastasis by regulating PKM2 nuclear translocation in triple-negative breast cancer. Cell Death Dis 17, 262 (2026). https://doi.org/10.1038/s41419-026-08524-4
Keywords: triple-negative breast cancer, lymph node metastasis, tumor-associated macrophages, PKM2, lymphangiogenesis