Clear Sky Science · en
Deubiquitinase UCH-L1 confers paclitaxel resistance via stabilizing PKM2 to promote glycolysis in triple-negative breast cancer
Why this research matters for patients
Chemotherapy is still one of the few options for people with triple‑negative breast cancer, an aggressive form of the disease that lacks common drug targets. Yet many tumors eventually learn to shrug off powerful medicines like paclitaxel, allowing the cancer to return and spread. This study uncovers a hidden wiring inside cancer cells that helps them resist paclitaxel by rewiring how they burn sugar, and it points to a new weak spot that future treatments could hit to keep chemotherapy working longer.
A tough-to-treat breast cancer
Triple‑negative breast cancer is defined by what it lacks: it does not carry receptors for estrogen, progesterone, or the HER2 growth factor that many modern drugs target. As a result, standard chemotherapy drugs remain the mainstay of treatment. Paclitaxel is one such workhorse, used around the world. However, a substantial fraction of patients either do not respond well from the start or become resistant over time, leading to tumor relapse and poorer survival. Doctors have long seen this pattern in the clinic but have lacked clear markers that could predict which tumors will resist paclitaxel and which molecular switches drive that resistance.
A nerve protein turns up in breast tumors
The researchers focused on a protein called UCH‑L1, best known for its role in nerve cells and brain disorders. By mining public cancer databases and examining tumor samples from triple‑negative breast cancer patients, they found that UCH‑L1 levels were much higher in these tumors than in normal breast tissue. Patients whose tumors had more UCH‑L1 tended to have shorter survival and were less likely to benefit from chemotherapy, especially from paclitaxel or related drugs. In lab-grown cancer cell lines, boosting UCH‑L1 made cells harder to kill with paclitaxel, while lowering UCH‑L1 had the opposite effect, both in dishes and in mouse tumor models. 
Cancer cells’ sugar craving and drug resistance
Cancer cells often favor a fast but inefficient way of burning sugar, known as aerobic glycolysis or the Warburg effect. This sugar rush supplies quick energy and building blocks for growth and has been linked to resistance to many treatments. When the team compared cancer cells with and without UCH‑L1, they saw clear changes in this metabolic behavior. Cells with less UCH‑L1 consumed less glucose, made less lactate, and produced less cellular energy, while cells with extra UCH‑L1 showed the opposite pattern and a stronger acidification signal tied to glycolysis. Importantly, when they blocked glycolysis with a compound called 2‑DG, the protective effect of UCH‑L1 against paclitaxel largely disappeared. This showed that UCH‑L1 helps cancer cells survive chemotherapy in large part by powering up their sugar‑burning machinery.
A protein partnership that protects tumor fuel
Diving deeper, the investigators looked for direct partners of UCH‑L1 inside the cell and homed in on PKM2, a key enzyme that sits at a crucial junction in the glycolysis pathway. Normally, PKM2 levels are controlled by a cellular recycling tag called ubiquitin, which marks proteins for breakdown. UCH‑L1 is a “de‑tagging” enzyme that can remove ubiquitin. The study showed that UCH‑L1 physically binds to the tail region of PKM2 and specifically strips off a type of ubiquitin chain that sends PKM2 to the cell’s disposal system. By cutting these chains at a particular site on PKM2, UCH‑L1 prevents its destruction and stabilizes the enzyme. With more PKM2 around, glycolysis runs hotter, feeding the tumor and helping it weather the stress of paclitaxel exposure. 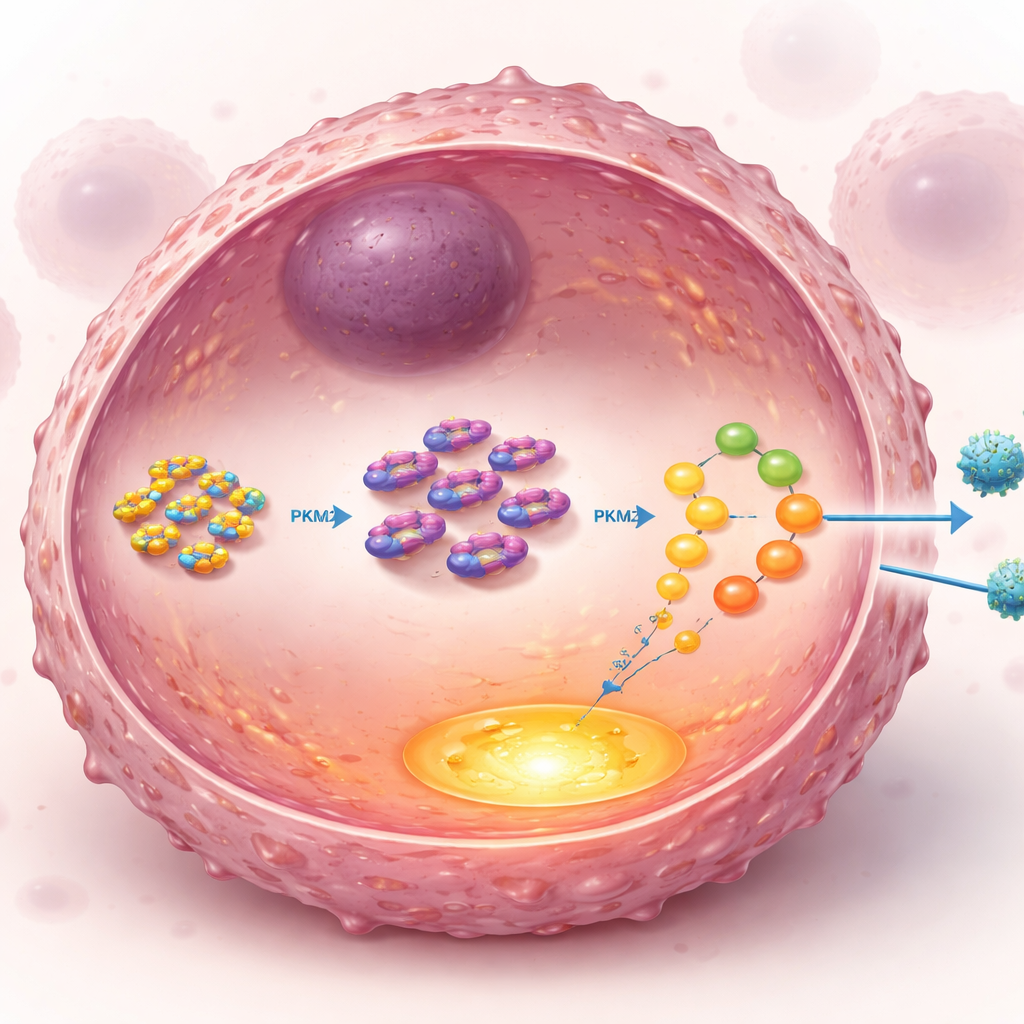
Reversing resistance in stubborn tumors
The team then turned to paclitaxel‑resistant triple‑negative breast cancer cells, both from patient‑derived datasets and from lab models. In these resistant cells, UCH‑L1 and PKM2 levels were both elevated, and glycolysis was heightened. Silencing UCH‑L1 or PKM2, or chemically blocking glycolysis, made these hardened cells once again vulnerable to paclitaxel, restoring cell death and shrinking tumors in mice. Re‑introducing PKM2 into UCH‑L1‑depleted tumors erased much of this benefit, underscoring that PKM2 is the key downstream player in this resistance circuit. In patient tumor samples, UCH‑L1 and PKM2 tended to be high together, and high PKM2 was also linked to poorer outcomes after chemotherapy.
What this means for future treatment
For non‑specialists, the message is that some triple‑negative breast cancers survive paclitaxel by turning up a sugar‑burning engine fueled by a partnership between UCH‑L1 and PKM2. UCH‑L1 acts like a protective mechanic that keeps PKM2 from being broken down, allowing cancer cells to generate more energy and resist chemotherapy‑induced damage. Measuring UCH‑L1 or PKM2 in tumors could help predict how well a patient will respond to paclitaxel, and drugs that block UCH‑L1, PKM2, or glycolysis may one day be combined with standard chemotherapy to overcome or prevent resistance and improve outcomes for people with this difficult cancer.
Citation: Chen, X., Zhou, X., Meng, Y. et al. Deubiquitinase UCH-L1 confers paclitaxel resistance via stabilizing PKM2 to promote glycolysis in triple-negative breast cancer. Cell Death Dis 17, 261 (2026). https://doi.org/10.1038/s41419-026-08521-7
Keywords: triple-negative breast cancer, paclitaxel resistance, tumor metabolism, glycolysis, PKM2