Clear Sky Science · en
Histone 3 lysine 36 trimethylation by SETD2 shapes an epigenetic landscape in intestinal stem cells to orchestrate lipid metabolism and attenuate cell senescence
Why Gut Stem Cells Matter as We Grow Older
Deep inside the lining of our intestines, tiny stem cells work nonstop to renew the tissue that helps us digest food and absorb nutrients. As we age, these stem cells tire out, making the gut more vulnerable to disease and slower to heal. This study explores how subtle chemical tags on DNA-packaging proteins help keep intestinal stem cells youthful, how their loss disrupts fat burning inside cells, and how carefully chosen drugs might one day slow this decline.
Aging Cells in a Demanding Environment
The inner surface of the intestine is one of the fastest renewing tissues in the body. Stem cells nestled at the base of small pockets, called crypts, divide to replace billions of cells every few days. This constant work requires a steady energy supply, much of it from breaking down fats. At the same time, these stem cells are controlled by an “epigenetic” code—chemical marks on histone proteins that help determine which genes are active or silent. The authors focused on one such mark, known as H3K36 trimethylation, created by an enzyme called SETD2, and asked whether changes in this mark might help explain why intestinal stem cells lose their vigor with age.
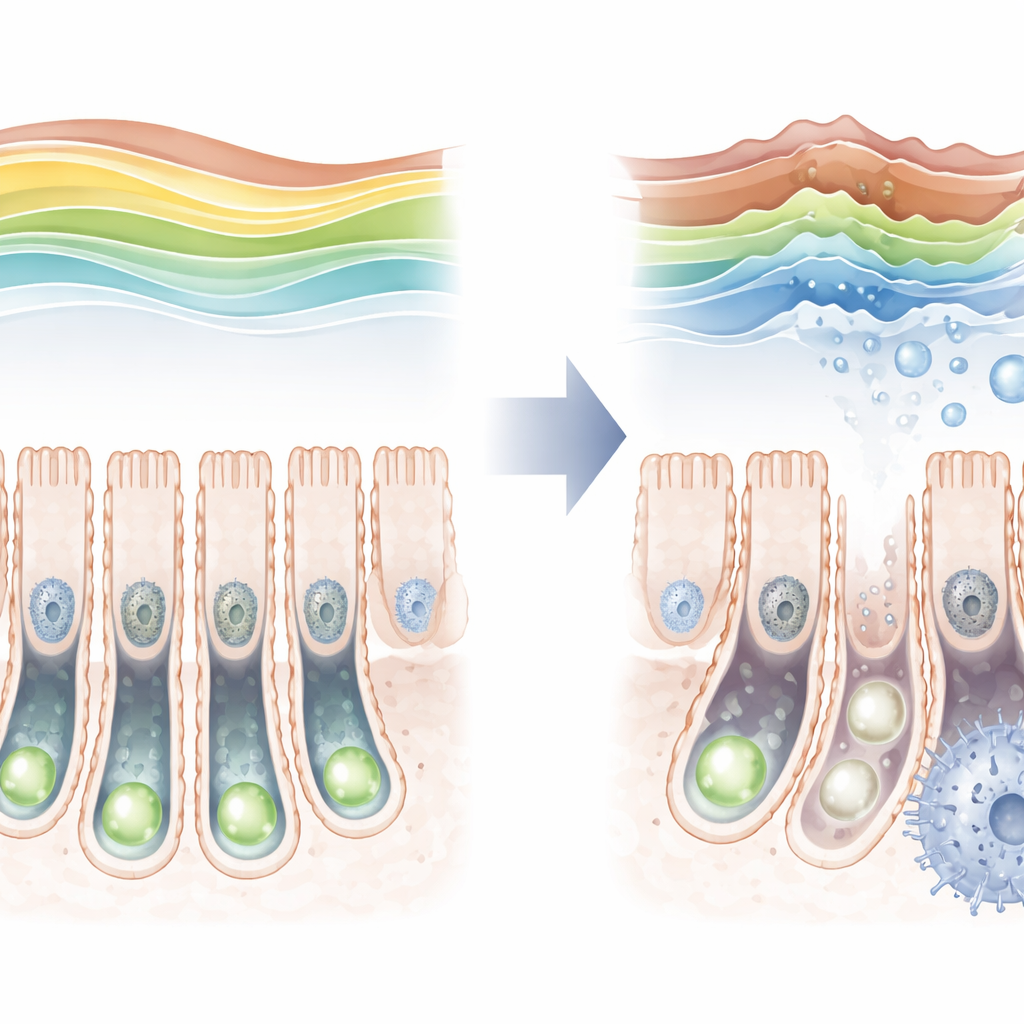
A Protective Mark That Fades With Age
By examining mouse intestines across the lifespan, the researchers found that the H3K36 trimethyl mark is not static. It is relatively low just after birth, then rises to a peak in young adults before falling again in old age. As this mark declined in older animals, markers of cellular aging increased in stem cells, hinting at a link between the two. To test this more directly, the team engineered mice in which SETD2 could be removed only in intestinal stem cells. When this enzyme was deleted, the protective mark almost vanished, stem cells divided less frequently, and they struggled to produce the full range of specialized intestinal cell types.
How Epigenetic Changes Derail Fat Burning
The loss of this single histone mark set off a cascade of changes inside stem cells. Gene-activity measurements showed that many genes involved in the cell cycle and DNA copying were turned down, while genes tied to stress and aging were turned up. Strikingly, the strongest signal pointed to disturbed lipid handling: genes that normally support fatty acid oxidation—the controlled burning of fats for energy—were dampened, and fats began to build up inside the crypts. Metabolite profiling confirmed that long-chain fatty acids accumulated, a sign that the usual fat-processing pathways were faltering. At the same time, the physical packing of DNA in the nucleus shifted, with many regions becoming more exposed or more tightly closed, reshaping which genes the cell could easily use.
Chromatin Remodelers and the March Toward Senescence
To understand how these DNA-packing changes arise, the authors mapped both open chromatin regions and a suite of other histone marks across the genome. When H3K36 trimethylation was lost, large stretches of DNA gained “active” marks and became more accessible, especially near genes linked to metabolism and aging. A key player in this transition was the SWI/SNF chromatin remodeling complex, powered by a core subunit called SMARCA4. In stem cells lacking SETD2, SMARCA4 levels rose, and the complex appeared to help pry open regions that drive senescence programs. When the researchers reduced SMARCA4 activity genetically or with a targeted inhibitor, stem cells regained some of their ability to form healthy mini-intestines in a dish, and aging markers declined—suggesting that excessive remodeling pushes these cells toward a tired, senescent state.
Reawakening Energy Pathways to Rescue Stem Cells
Because disrupted fat burning emerged as a central problem, the team tested whether boosting this pathway could counteract stem cell aging. They used a drug that activates PPARα, a master regulator of fatty acid oxidation. In organoids grown from SETD2-deficient stem cells, this treatment increased growth, restored the budding structures characteristic of vigorous stem cells, and reduced fat accumulation. In live mice, the same intervention partially replenished the stem cell pool, lowered markers of senescence, and normalized lipid buildup in the intestinal crypts. These findings place fatty acid oxidation as a crucial link between epigenetic control and stem cell health.
What This Means for Healthy Aging
Taken together, the work shows that a specific histone mark, deposited by SETD2, helps maintain a balanced epigenetic landscape in intestinal stem cells. When this mark fades, chromatin becomes abnormally remodeled, fat-burning pathways falter, lipids accumulate, and cells drift into senescence, weakening the gut’s repair system. By either dialing down the overactive remodeling machinery or restoring fat metabolism with targeted drugs, it may be possible to preserve stem cell function and intestinal health later in life. While these findings come from mice, they echo patterns seen in human gut disorders, raising the prospect that carefully tuned metabolic therapies could one day help counteract age-related decline in our own intestinal stem cells.
Citation: Xu, Y., Wang, Z., Feng, W. et al. Histone 3 lysine 36 trimethylation by SETD2 shapes an epigenetic landscape in intestinal stem cells to orchestrate lipid metabolism and attenuate cell senescence. Cell Death Dis 17, 273 (2026). https://doi.org/10.1038/s41419-026-08518-2
Keywords: intestinal stem cells, epigenetics, fatty acid oxidation, cellular senescence, SETD2