Clear Sky Science · en
A Lamp2a-linked RNA secreted by ADSCs prevents ENO1–lactylation–glycolysis feedback and cell malignant behavior in triple-negative breast cancer
Why this matters for patients
Triple-negative breast cancer is one of the most aggressive forms of breast cancer and lacks the hormone or growth-factor targets that make other subtypes more treatable. This study uncovers how these tumors rewire their energy use to survive in low-oxygen environments and describes an inventive way to attack that wiring. For readers, it offers a glimpse of how understanding cancer metabolism can lead to new, highly targeted therapies that differ sharply from traditional chemotherapy.
How these tumors fuel themselves
Many cancers rely heavily on a fast but inefficient way of making energy called glycolysis, which turns sugar into the by-product lactate. By examining patient samples, public gene databases, and multiple breast cancer cell lines, the researchers show that triple-negative tumors lean on glycolysis even more than other breast cancers. A key enzyme in this pathway, ENO1, is present at especially high levels in these tumors and is linked to worse survival. When ENO1 levels were reduced in lab-grown triple-negative cells, the cells shifted away from glycolysis toward more normal energy production in mitochondria, grew more slowly, and were less able to invade or form colonies.
A dangerous feedback loop inside cancer cells
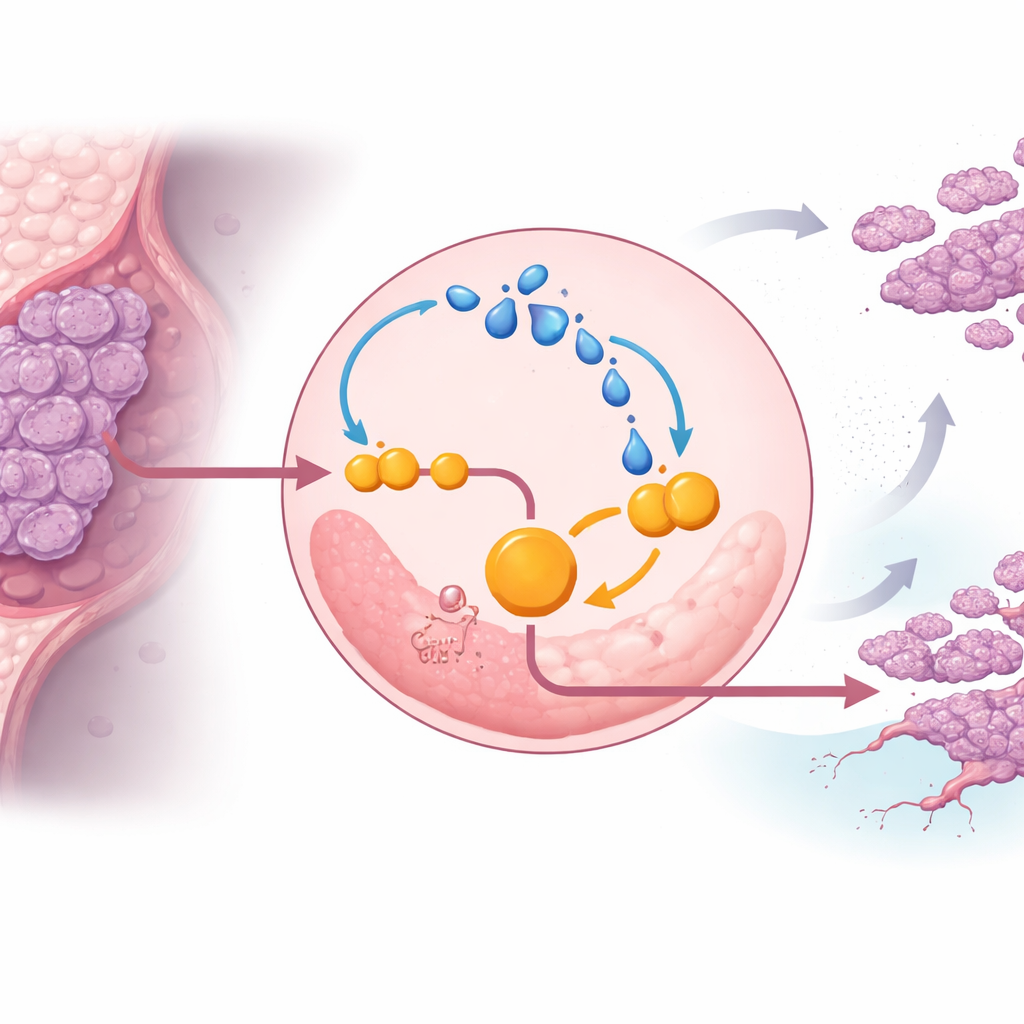
Digging deeper, the team discovered that lactate itself strengthens ENO1 and locks cancer cells into a vicious cycle. Lactate can chemically modify proteins in a process called lactylation. In triple-negative cells, extra lactate increased lactylation of ENO1, which in turn boosted the enzyme’s activity and stability, further accelerating glycolysis and producing even more lactate. Under low-oxygen conditions—typical of solid tumors—this loop became stronger, helping cancer cells resist cell death and continue proliferating. Blocking ENO1’s activity with a small-molecule inhibitor, or reducing lactate production, weakened this loop, curbed glycolysis, and slowed tumor growth in mice.
The molecular switch that protects ENO1
The study identified a specific control point on ENO1 that makes this loop possible. A protein known as EP300 adds lactate-based tags to several lysine sites on ENO1, with one site, termed K262, turning out to be crucial. When this site was mutated so it could no longer be lactylated, ENO1 was rapidly shuttled to the cell’s recycling centers, the lysosomes, and broken down. Without this protective tag, triple-negative cells lost much of their glycolytic drive and ability to form tumors and metastases in animal models. This singles out ENO1 lactylation at K262 as a molecular switch that shields the enzyme from destruction and sustains the cancer’s altered metabolism.
A new way to mark ENO1 for destruction
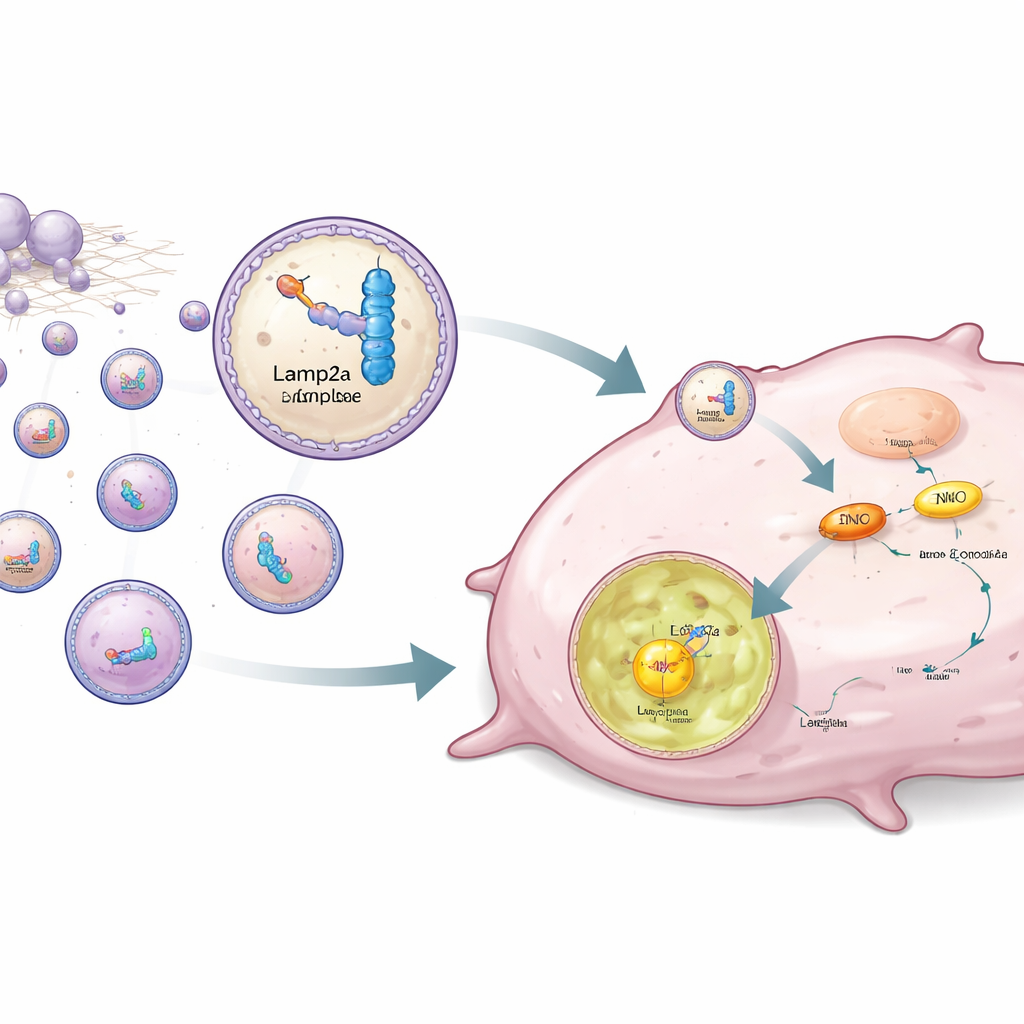
Rather than only blocking ENO1, the authors designed a strategy to selectively remove it from cancer cells. ENO1 also acts as an RNA-binding protein, recognizing specific short RNA sequences. The team engineered synthetic RNA pieces that bind ENO1 strongly and then linked them to Lamp2a, a lysosomal membrane protein involved in targeted protein degradation. Human fat-derived stem cells were genetically modified to produce this RNA–Lamp2a pair and to package it into tiny vesicles called exosomes. When these engineered exosomes were added to triple-negative cells, the RNA portion latched onto ENO1, while the Lamp2a part guided the whole complex into lysosomes, where ENO1 was degraded even if it carried the stabilizing lactylation mark.
Testing the targeted degradation approach in animals
To bring this idea closer to a practical therapy, the researchers grew the modified stem cells on a biodegradable scaffold and implanted this construct under the skin of mice, near triple-negative tumors. From this location, the stem cells continuously released exosomes carrying the ENO1-targeting complexes. Tumors in these mice showed markedly lower ENO1 protein levels, reduced glycolysis, fewer dividing cells, and more signs of cell death compared with control animals. The tumors grew more slowly, and their malignant behavior was clearly dampened, demonstrating that directing ENO1 into lysosomes can effectively starve the cancer of its preferred fuel pathway.
What this could mean for future treatment
For a non-specialist audience, the take-home message is that this work reveals both a weakness and a potential new treatment angle for triple-negative breast cancer. The weakness is the tumor’s dependence on a self-reinforcing energy loop built around ENO1 and lactate, especially in low-oxygen regions. The treatment angle is a targeted protein degradation system that uses custom RNA and stem-cell-derived exosomes to drag ENO1 into the cell’s disposal machinery. While far from clinical use, this approach illustrates how deeply understanding cancer’s metabolic tricks can inspire precise therapies that aim to disarm, rather than simply poison, malignant cells.
Citation: Cheng, S., Xia, B., Li, L. et al. A Lamp2a-linked RNA secreted by ADSCs prevents ENO1–lactylation–glycolysis feedback and cell malignant behavior in triple-negative breast cancer. Cell Death Dis 17, 288 (2026). https://doi.org/10.1038/s41419-026-08517-3
Keywords: triple-negative breast cancer, cancer metabolism, lactate signaling, targeted protein degradation, ENO1 enzyme