Clear Sky Science · en
Shp1 phosphatase regulates CXCR2 protein stability and IL8-mediated invasiveness in breast cancer
Why this research matters for breast cancer
Breast cancer remains one of the leading causes of cancer death in women, largely because some tumors become invasive and spread to distant organs. This study unpacks how a common inflammatory signal in tumors, called IL8, talks to breast cancer cells and how a molecular "brake" named Shp1 helps decide whether those cells stay put or become more aggressive. Understanding this molecular tug-of-war could open new ways to slow or stop the spread of difficult-to-treat breast cancers, especially triple-negative disease.
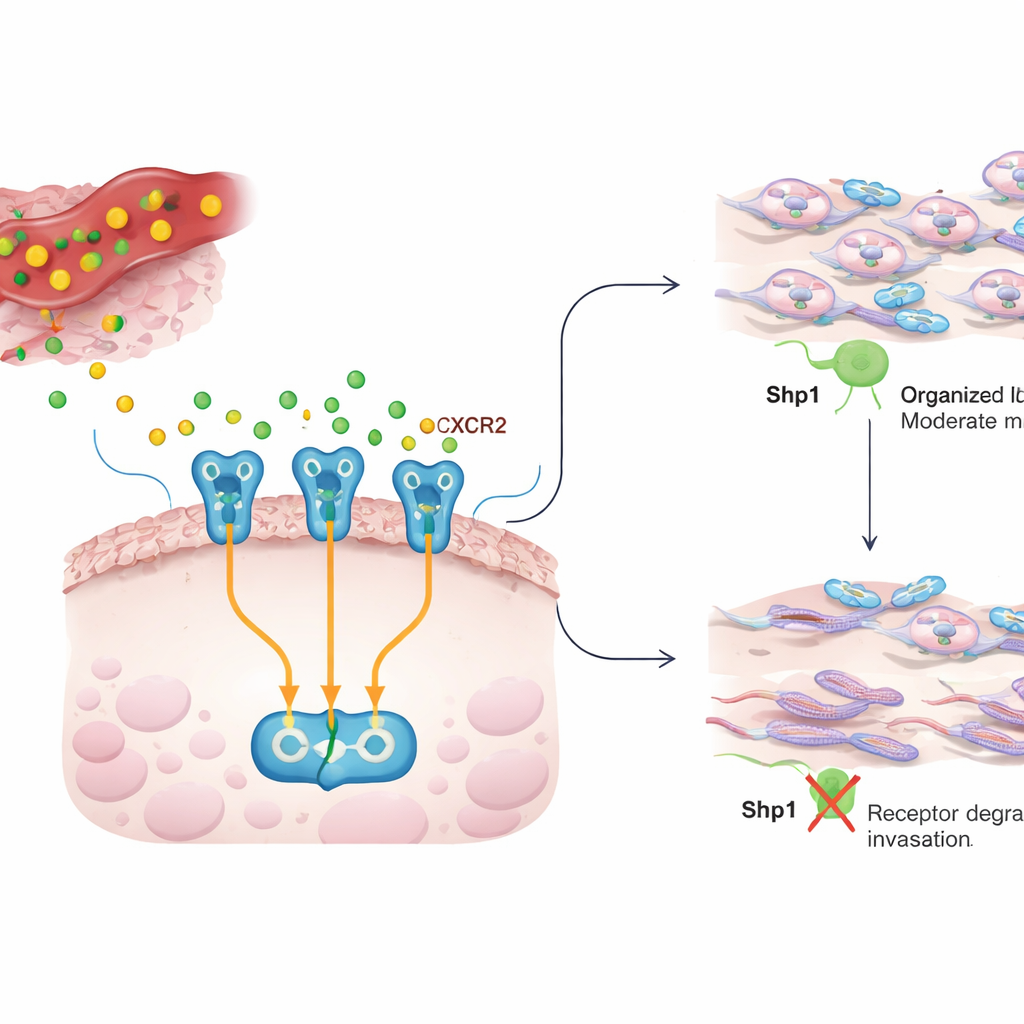
A conversation between inflammation and cancer cells
Inside a tumor, cancer cells are surrounded by immune and support cells that release chemical messengers, or cytokines. IL8 is one of these messengers and is often found at high levels in many cancers, where it fuels growth, blood vessel formation, and the ability of cancer cells to move and invade. IL8 signals through a receptor on the cancer cell surface called CXCR2. When IL8 binds to CXCR2, it switches on several internal circuits that reorganize the cell’s skeleton, helping cells crawl through tissue and spread. The current work asks how this IL8–CXCR2 conversation is kept under control, and whether Shp1, an enzyme usually seen as a tumor-suppressing brake on growth signals, also shapes this invasive behavior.
The hidden role of Shp1 in cell movement
The researchers first removed or chemically blocked Shp1 in a widely used luminal breast cancer cell line (MCF7) and measured how well the cells could migrate and invade through a gel that mimics tissue. Without Shp1, cancer cells closed wounds faster and squeezed through the gel two to three times more efficiently, revealing that Shp1 normally restrains movement. Using patient data, they also found that women whose tumors expressed less Shp1 had worse survival and shorter time before relapse. Together, these results suggest that Shp1 limits the aggressive behavior of breast cancer cells and that losing this brake is linked to poorer outcomes.
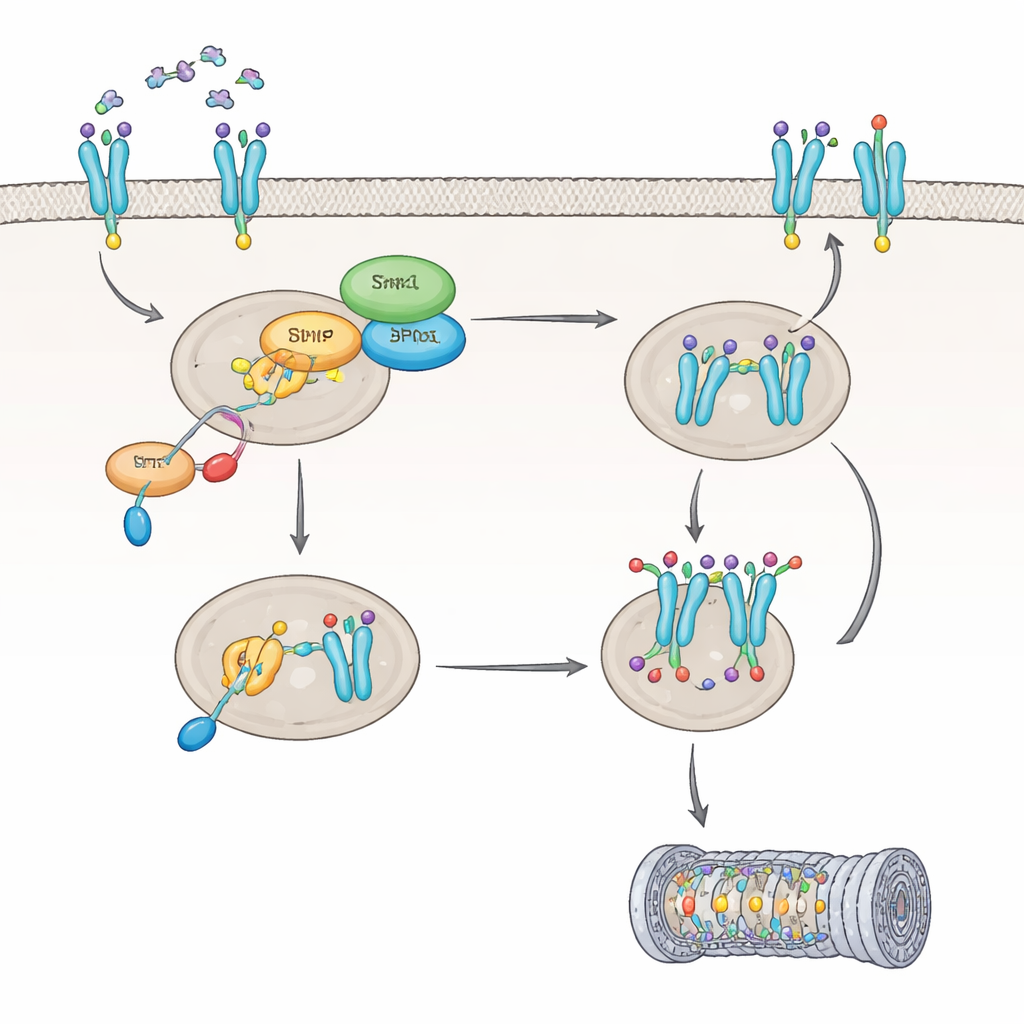
How IL8 flips Shp1’s switch and reprograms a key receptor
Next, the team explored how IL8 affects Shp1 itself. They discovered that IL8 activates a protein called PKC, which marks Shp1 at a specific site, turning down its enzymatic activity by about 60 percent. When Shp1 is dampened in this way, another enzyme, PP2A, becomes less active, and the CXCR2 receptor stays in a highly modified state that changes its fate inside the cell. Instead of being cleaned up and recycled back to the cell surface, CXCR2 becomes tagged for destruction, mainly via the cell’s protein-shredding machinery, the proteasome. A particular modification on CXCR2, at an amino acid named Ser347, proved crucial for attaching these degradation tags. In cells lacking Shp1, CXCR2 was more heavily tagged and broken down faster, leading to fewer receptors at the cell surface and a blunted response to IL8 over time.
Different breast cancer types, different wiring
Breast cancer is not a single disease; tumors are grouped into luminal, HER2-positive, and triple-negative subtypes based on their receptor patterns. The researchers tested the IL8–CXCR2–Shp1 circuit in several cell lines representing each subtype. In luminal and triple-negative cells, IL8 turned on the Shp1–PP2A–CXCR2 cascade, and blocking Shp1 both increased baseline invasiveness and erased IL8’s usual pro-invasive effect. In contrast, HER2-positive cells still responded to IL8, but Shp1 blockade hardly changed their behavior, suggesting that in these tumors Shp1 is tied up with other signals, such as HER2, and less available to regulate CXCR2. Large-scale gene expression analyses supported this picture: triple-negative lines tended to have lower Shp1, altered GPCR-related signaling, and higher IL8, consistent with a more aggressive, inflammation-driven profile.
What this means for future treatments
To a non-specialist, the core message is that the enzyme Shp1 acts as a traffic controller for the IL8 receptor CXCR2, determining whether the receptor is reused or destroyed and, in turn, how strongly breast cancer cells can respond to inflammatory cues. In luminal and triple-negative breast cancers, this control point appears especially important: when Shp1 levels are low or its activity is blocked, cells become more invasive and patient outcomes worsen. Because Shp1 can be boosted or modulated by certain existing drugs, targeting the Shp1–CXCR2 axis, possibly together with IL8 or CXCR2 blockers, could offer a new strategy to limit invasion and metastasis in aggressive breast cancer subtypes.
Citation: Monti, M., Amendola, P.G., Filograna, A. et al. Shp1 phosphatase regulates CXCR2 protein stability and IL8-mediated invasiveness in breast cancer. Cell Death Dis 17, 297 (2026). https://doi.org/10.1038/s41419-026-08516-4
Keywords: breast cancer invasion, IL8 signaling, CXCR2 receptor, Shp1 phosphatase, triple-negative breast cancer