Clear Sky Science · en
CRMP2 inhibits metastasis formation by impairing ILF3-dependent stabilization of CXCL10 mRNA in breast cancer
Why stopping cancer spread matters
Most deaths from breast cancer happen not because of the original tumor, but because cancer cells travel to vital organs like the lungs and form new growths. This study uncovers a built‑in protective protein inside breast cancer cells that can slow or even block this spread by changing the “welcome mat” that distant organs lay out for wandering cancer cells. Understanding this hidden safety switch could open the door to new treatments that keep cancer from taking root elsewhere in the body.
A quiet defender inside cancer cells
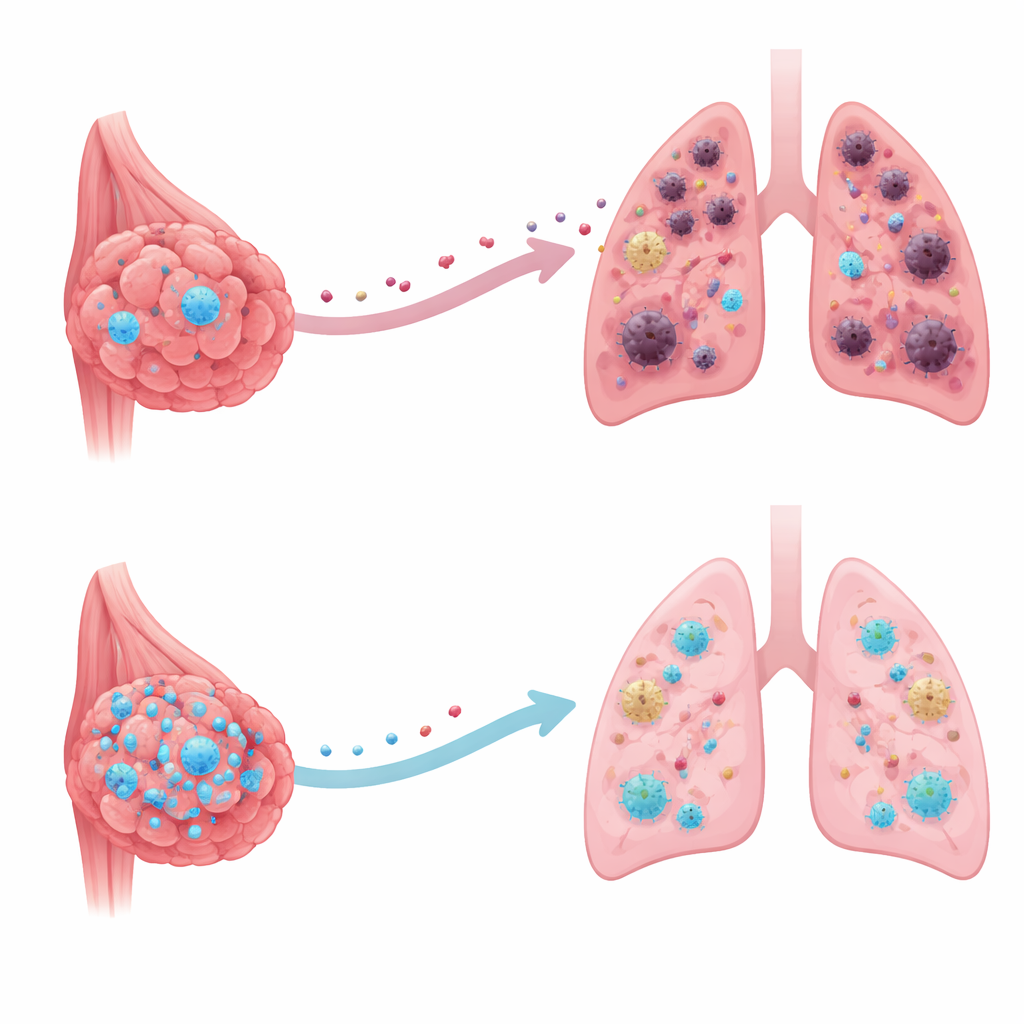
The researchers focused on a protein called CRMP2, best known for helping nerve cells grow and maintain their internal skeleton. When they examined large public cancer databases and patient tumor samples, they found that CRMP2 levels were unusually low in breast tumors, especially in cancers that had already spread. Patients whose tumors still had higher amounts of CRMP2 tended to live longer and were less likely to develop distant metastases. This pattern suggested that CRMP2 behaves more like a brake than an accelerator in breast cancer, acting as an internal defender rather than a driver of disease.
How the lungs are prepared for invasion
Cancer cells rarely arrive in a distant organ by surprise. Long before they get there, the original tumor releases molecules that reshape that organ into what scientists call a “pre‑metastatic niche” – a microenvironment that favors cancer survival. Using a well‑established mouse model that closely mimics human breast cancer, the team showed that when breast tumor cells were engineered to produce extra CRMP2, the primary tumors grew just as fast, but the lungs became far less hospitable. Markers of inflammation and tissue‑remodeling in the lungs dropped, and fewer of the immune cells that normally suppress anti‑tumor defenses were recruited. As a result, after the main tumor was removed, far fewer metastatic nodules formed in the lungs.
A signaling chain that stirs up trouble
Diving deeper, the scientists asked which tumor‑secreted signals were responsible for preparing the lungs. They homed in on CXCL10, a small messenger protein (chemokine) known to attract certain immune cells and to awaken dormant cancer cells. In both lab‑grown human and mouse breast cancer cells, boosting CRMP2 sharply reduced the amount of CXCL10 made and released. In mice, lungs exposed to tumor material rich in CRMP2 showed much lower CXCL10 and fewer incoming immune cells that normally dampen protective T‑cell activity. When the researchers blocked CXCL10 with an antibody, lung colonization by cancer cells decreased; when they added extra CXCL10 back, it erased the protective effect of CRMP2. This placed CXCL10 squarely downstream of CRMP2 as a key instigator of a metastasis‑friendly niche.
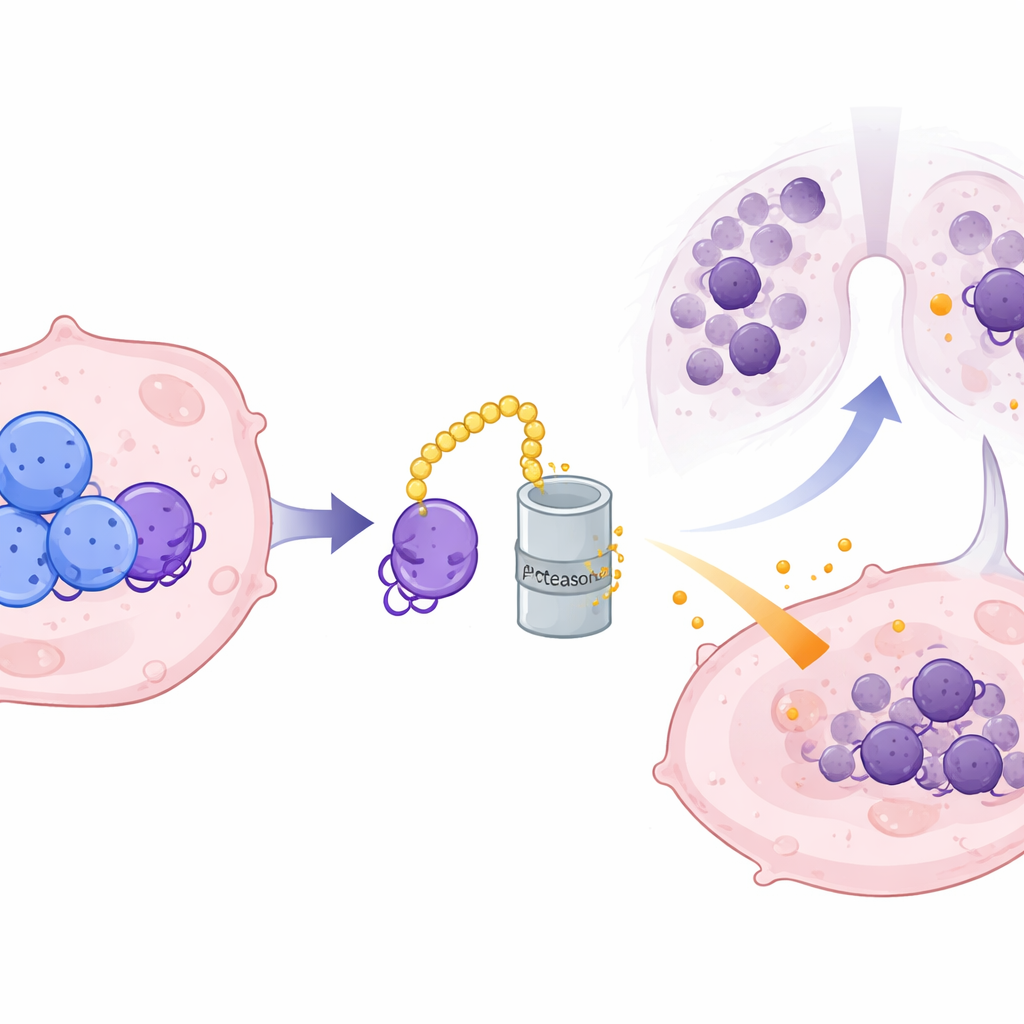
The hidden middleman that stabilizes bad signals
Because CRMP2 lives inside cells while CXCL10 is secreted, the team suspected a molecular middleman. They identified ILF3, an RNA‑binding protein, as a direct partner of CRMP2. ILF3 latches onto the RNA blueprint for CXCL10 and normally protects it from being broken down, allowing more CXCL10 protein to be produced. CRMP2 binds ILF3 at specific contact points and tags it for destruction through the cell’s protein‑recycling machinery, shortening ILF3’s lifespan and breaking its hold on CXCL10 RNA. When CRMP2 levels are high, ILF3 is degraded more quickly, CXCL10 RNA becomes unstable, and less of the inflammatory chemokine is secreted. When ILF3 was artificially restored, it could override CRMP2’s suppression of CXCL10, confirming its role as the crucial link in this chain.
Turning a plant compound into a potential ally
To test whether this pathway could be harnessed therapeutically, the researchers screened a library of natural compounds and found that psoralen – a plant‑derived molecule long used in skin treatments – binds directly to CRMP2 and makes it more stable. In cell experiments, psoralen raised CRMP2 levels, lowered ILF3 and CXCL10, and reduced the invasive behavior of breast cancer cells without harming their general viability. In multiple mouse models, psoralen did not shrink primary breast tumors, but it strongly reduced lung metastases and blunted formation of the pre‑metastatic niche, again in a manner that depended on CRMP2. Although psoralen itself can cause liver toxicity and will need refinement, these results show that small molecules can boost CRMP2’s protective role.
What this means for patients
Taken together, this work reveals a protective axis inside breast cancer cells in which CRMP2 destabilizes ILF3, which in turn lowers CXCL10 and keeps the lungs from becoming fertile ground for metastasis. Rather than attacking cancer cells directly, this strategy aims to disarm the distant niches they depend on, tipping the balance in favor of the body’s own defenses. While much remains to be done before such an approach reaches the clinic, targeting the CRMP2–ILF3–CXCL10 pathway – possibly with safer psoralen‑like drugs – offers a promising new angle for keeping breast cancer from spreading.
Citation: Lin, B., Luo, M., Zhou, Y. et al. CRMP2 inhibits metastasis formation by impairing ILF3-dependent stabilization of CXCL10 mRNA in breast cancer. Cell Death Dis 17, 255 (2026). https://doi.org/10.1038/s41419-026-08515-5
Keywords: breast cancer metastasis, pre-metastatic niche, chemokine signaling, RNA-binding proteins, natural product therapeutics