Clear Sky Science · en
The research advances of crosstalk between cancer-associated fibroblasts and tumor cells using co-culture organoids
Why the Tumor Neighborhood Matters
Cancer is more than just a mass of rogue cells; it grows inside a bustling neighborhood of support cells, scaffolds, blood vessels, and immune defenders. Among these neighbors, a group called cancer-associated fibroblasts, or CAFs, quietly shape how tumors start, spread, and respond to treatment. This review article explains how scientists are using tiny 3D mini-tumors called organoids, grown together with CAFs, to uncover this hidden conversation and to design smarter, more precise therapies.
Tiny Tumors in a Dish
Traditional cancer research has often relied on flat layers of cells in plastic dishes or on animal models, both of which capture only part of the real disease. Organoids change this picture. They are three-dimensional clusters of patient-derived tumor cells that preserve much of the original tumor’s structure, genetic makeup, and behavior. When these organoids are grown together with CAFs, they begin to mimic actual solid tumors, including how they respond to chemotherapy and radiation. Different culture setups have been developed: simple gel-based systems where tumor cells and CAFs share the same matrix, mixed "hanging drop" methods that first form compact spheroids, and sophisticated microfluidic chips that control fluid flow and organoid size for high-throughput drug testing. Each approach balances realism, cost, and technical difficulty.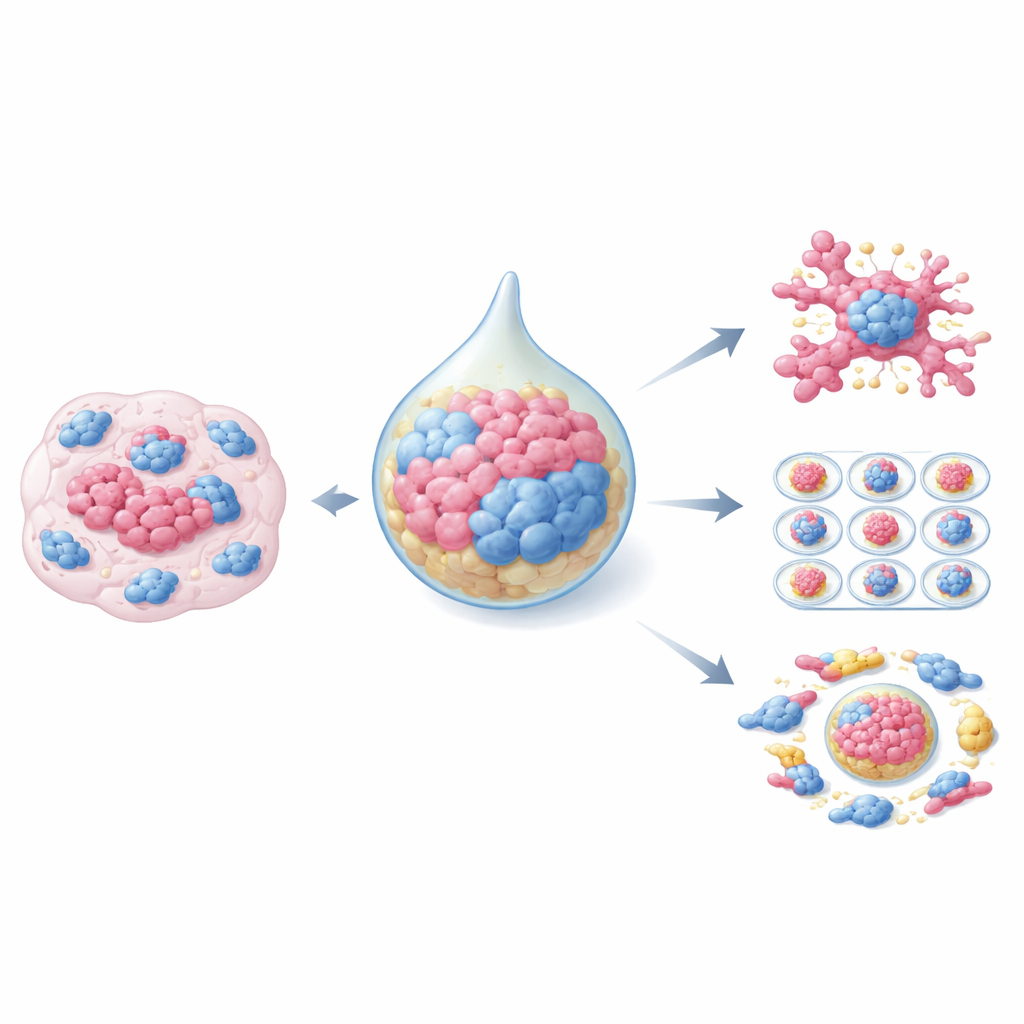
How Support Cells Help Tumors Thrive
CAFs are not a single uniform cell type; they come in many flavors, each with its own job. In co-culture organoids, CAFs help build and remodel the fibrous framework around cancer cells, changing stiffness and structure in ways that favor tumor growth and invasion. They release a cocktail of signals that spur cancer cells to multiply, move, and invade nearby tissues, and they can even coax ordinary cells to acquire stem cell–like traits that seed new tumors. CAFs also reshape how cancer cells make and use energy, trading nutrients back and forth to keep both partners alive under stress. In many models, CAFs make tumors more resistant to chemotherapy and targeted drugs, for example by shielding cancer cells within dense collagen, switching on survival pathways, or sending small vesicles packed with RNA molecules that strengthen DNA repair.
Immune Escape and Blood Supply
Beyond feeding and sheltering cancer cells, CAFs strongly influence the body’s defenses. Co-culture organoids and organoid-on-a-chip systems show that CAFs can repel killer immune cells, attract suppressive immune partners, and drive immune cells into a state where they can no longer attack. Some CAF subsets overload natural killer cells with iron and push them into a form of cell death, while others call in tumor-supporting macrophages. At the same time, CAFs encourage the growth of new blood vessels by secreting factors that prompt vessel-forming cells to sprout and build channels, ensuring tumors receive a steady flow of oxygen and nutrients. Yet the story is not entirely one-sided: certain rare CAF subtypes can actually support anti-tumor immunity by helping activate T cells or reprogramming other immune cells into a more helpful state.
A Two-Way Conversation with Tumor Cells
The relationship between CAFs and cancer cells is a feedback loop. Tumor cells send out signals that turn nearby normal fibroblasts into CAFs and then further sculpt them into different subgroups, such as contractile, matrix-producing cells or inflammation-driven cells that secrete many cytokines. Through soluble factors and tiny vesicles, cancer cells can push CAFs toward states that stiffen the tissue, promote invasion, and drive drug resistance. Co-culture organoids have revealed how specific signal routes act like dials that shift CAF identity, allowing tumors to adapt to changing conditions or treatments. Understanding this back-and-forth is crucial because blunt approaches that simply try to eliminate all CAFs have, in some animal studies and clinical trials, unexpectedly accelerated tumor growth.
New Ways to Target the Tumor Neighborhood
Armed with organoid models that preserve CAF–tumor crosstalk, researchers are exploring more refined treatment strategies. Instead of wiping out all fibroblasts, some approaches aim to block harmful signals between CAFs and cancer cells, disrupt their shared metabolism, or gently “re-educate” CAFs into more benign or even tumor-restraining states. Experimental tactics include small molecules that alter CAF signaling pathways, drugs that soften or thin the tumor’s fibrous shell, and advanced cell therapies such as engineered immune cells designed to recognize and attack specific CAF subsets. Organoid-based platforms make it possible to test these ideas on patient-derived tissue in the lab, offering a path toward personalized combinations that target both cancer cells and their supportive neighborhood.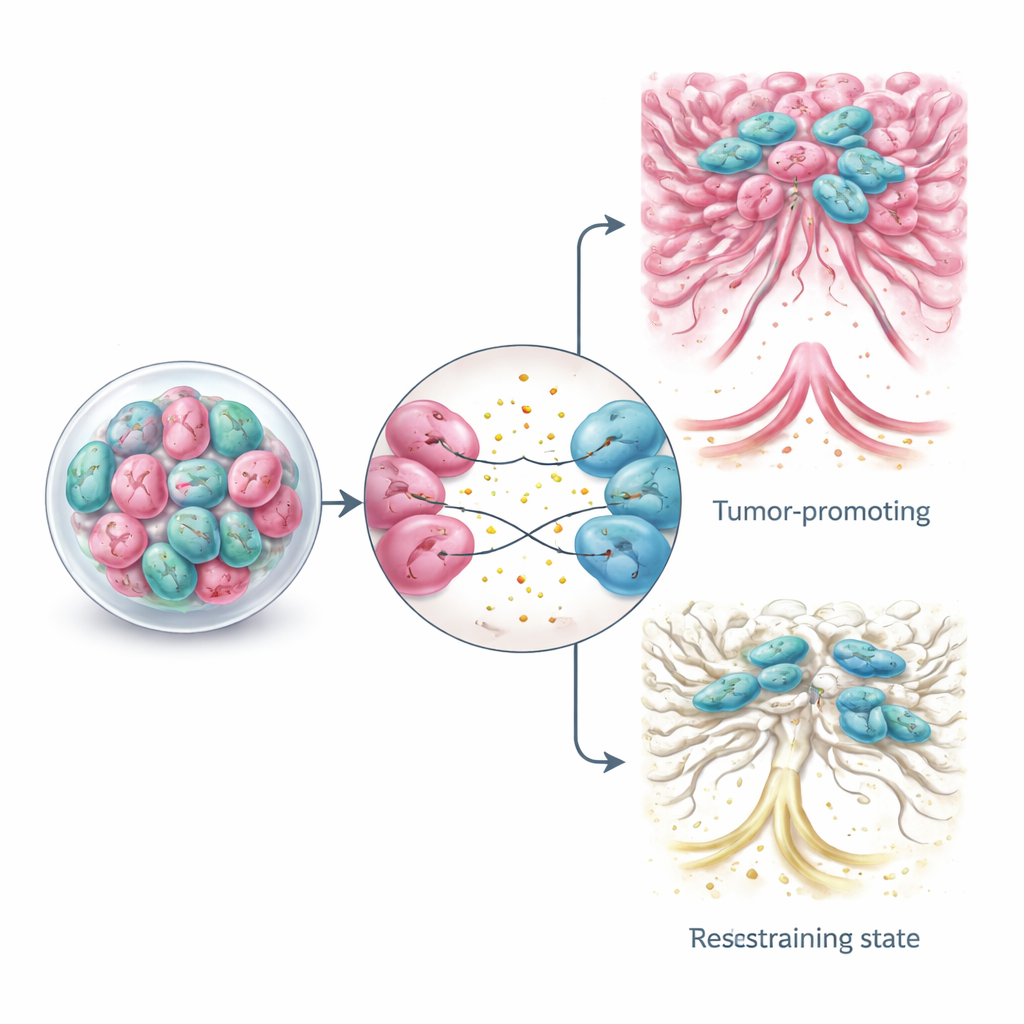
What This Means for Future Cancer Care
This review concludes that CAFs are both powerful helpers and, at times, quiet opponents of cancer. Their diversity and flexibility explain why simple “CAF-killing” treatments have often failed and sometimes caused harm. By using co-culture organoids as realistic testbeds, scientists can now sort good from bad CAF behaviors, map the signals that link them to tumor cells, and design therapies that selectively block tumor-promoting roles while preserving or boosting protective ones. For patients, this work lays the groundwork for more accurate drug testing and combination treatments that do not just attack the tumor, but also reshape the neighborhood that lets it flourish.
Citation: Wang, M., Ding, X., Chen, L. et al. The research advances of crosstalk between cancer-associated fibroblasts and tumor cells using co-culture organoids. Cell Death Dis 17, 267 (2026). https://doi.org/10.1038/s41419-026-08512-8
Keywords: cancer-associated fibroblasts, tumor organoids, tumor microenvironment, therapy resistance, 3D co-culture models