Clear Sky Science · en
Ubenimex synergizes with the PD-L1 blockade in gastric cancer by competitively binding LAP3 with UBE3A
Why this research matters
For many people with advanced stomach cancer, modern immunotherapy drugs that “take the brakes off” the immune system have brought new hope—but they still help only a minority of patients. This study asks a practical question with real-world impact: can an older immune-boosting medicine, Ubenimex, be repurposed to make these drugs work better, and why would that help? The researchers uncover a hidden molecular trick tumors use to escape immune attack and show how Ubenimex can undo it, opening the door to more effective combination treatments.
A new partner for cancer immunotherapy
The work focuses on gastric (stomach) cancer, a disease that remains common and deadly, especially in China. Drugs that block PD-1 or PD-L1—proteins that dampen immune responses—are now a backbone of treatment. Yet most patients either never respond or eventually relapse. The authors turned to Ubenimex, a long-approved drug used as an add-on to chemotherapy and radiation, known to stimulate immune cells. They wondered whether pairing Ubenimex with PD-L1–blocking antibodies could make tumors more vulnerable to attack by killer T cells, the immune cells that directly destroy cancer cells.
Putting the drug combo to the test
Using a mouse model of gastric cancer in which tumors were engineered to overproduce a protein called LAP3, the team treated animals with Ubenimex, anti–PD-L1, both, or neither. Each treatment alone shrank tumors, but the combination stood out: tumors regressed completely in all treated mice, and survival improved more than with PD-L1 blockade alone. Immune analyses showed that the combo greatly boosted the number and activity of CD8 “killer” T cells, which released more toxic molecules and inflammatory messengers. Importantly, the treatment did not cause obvious liver damage or weight loss, suggesting the approach could be powerful without being overly harsh.
A hidden switch that shields tumors
To understand why Ubenimex helped, the researchers homed in on LAP3, an enzyme it inhibits. They found that LAP3 levels were higher in human gastric tumors than in normal stomach tissue and that patients with more LAP3 tended to have worse survival. In both patient samples and mouse tumors, high LAP3 went hand-in-hand with fewer CD8 T cells inside the cancer, even though other immune cell types were unchanged. When LAP3 was experimentally reduced in tumor cells, tumors grew more slowly, attracted more CD8 T cells, and became easier for these cells to kill in lab tests. Together, the data suggest that LAP3 is not just a bystander but an active promoter of immune escape.
How tumors use LAP3 to keep the brakes on
Digging deeper, the team uncovered how LAP3 protects tumor cells from immune attack. They discovered that LAP3 helps stabilize PD-L1, the key “brake” protein on cancer cells that shuts down T cells. Normally, PD-L1 can be tagged with small protein markers that send it to the cell’s garbage disposal machinery. An enzyme called UBE3A attaches these tags, leading to PD-L1 breakdown. LAP3 physically binds UBE3A and, by doing so, prevents UBE3A from tagging PD-L1. As a result, PD-L1 accumulates on the tumor surface, forming a protective shield against CD8 T cells. When LAP3 levels are lowered, or when its action is blocked, PD-L1 is tagged more heavily, broken down faster, and its surface levels fall—allowing T cells to approach and attack.
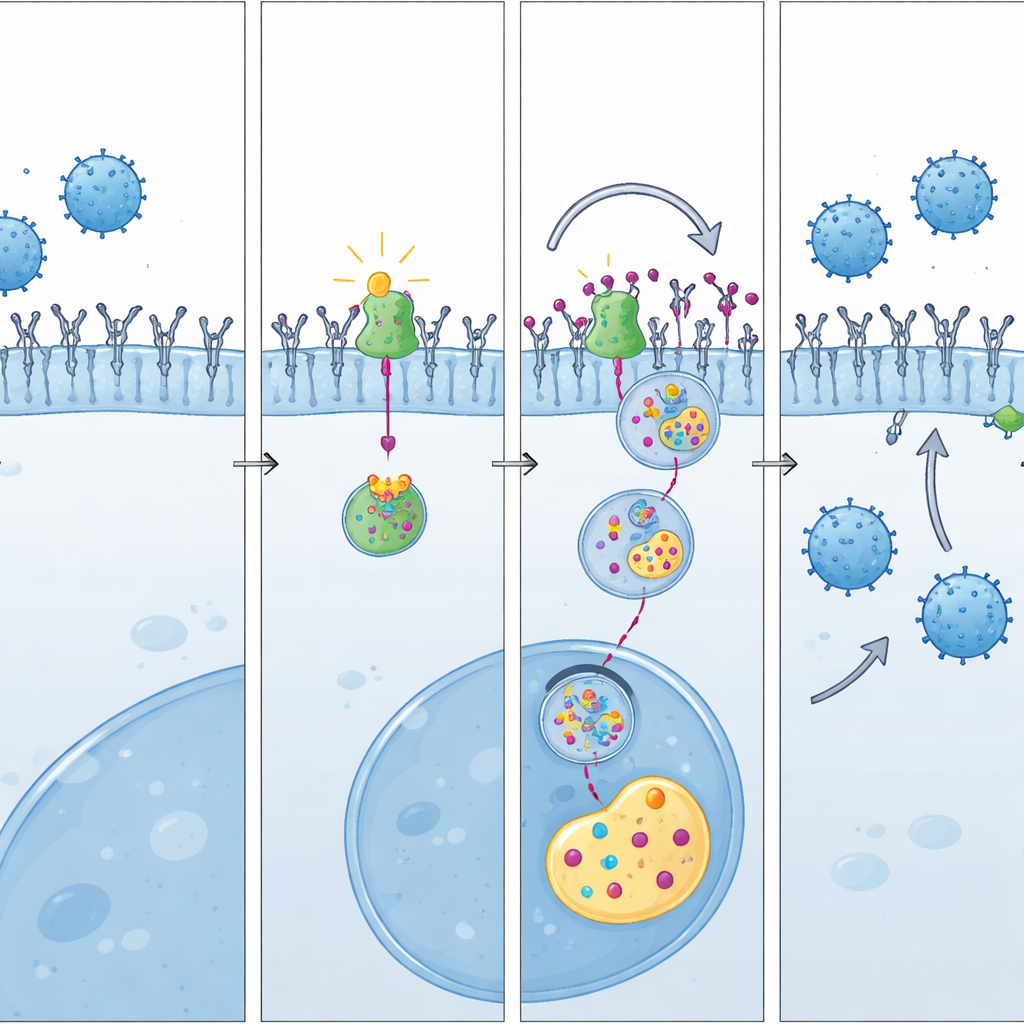
How Ubenimex tips the balance
The study then shows that Ubenimex works by interfering with the LAP3–UBE3A partnership. Computer modeling and biochemical experiments indicate that Ubenimex lodges in LAP3 at the same region where LAP3 normally interacts with UBE3A. This competitive binding pries UBE3A away from LAP3, freeing it to tag PD-L1 for destruction. In cell experiments, Ubenimex reduced PD-L1 on the tumor surface, increased the tagging of PD-L1 for disposal, and reversed the PD-L1-raising effects of excess LAP3. In living mice, that molecular shift translated into more active CD8 T cells and striking tumor control when Ubenimex was combined with PD-L1 blockade.
What this means for patients
Altogether, the study paints a clear picture: many gastric tumors overproduce LAP3, which helps them keep high levels of PD-L1 and thus hide from the immune system. Ubenimex breaks this protection by blocking LAP3’s grip on UBE3A, allowing PD-L1 to be cleared away. With fewer brakes on the surface of cancer cells, PD-L1–blocking drugs and killer T cells can work together far more effectively. Because Ubenimex is already in clinical use and LAP3 can be measured in tumor samples, this research provides a concrete, testable strategy for improving immunotherapy in stomach cancer and potentially other cancers with elevated LAP3.
Citation: Zhao, C., Li, J., Zheng, J. et al. Ubenimex synergizes with the PD-L1 blockade in gastric cancer by competitively binding LAP3 with UBE3A. Cell Death Dis 17, 241 (2026). https://doi.org/10.1038/s41419-026-08509-3
Keywords: gastric cancer, immunotherapy, PD-L1, Ubenimex, LAP3