Clear Sky Science · en
CCL11 promotes hepatocellular carcinoma recurrence after surgery by potentiating immunosuppressive CCR5 + CD206 + M2-like macrophages and promoting tumor invasiveness
Why liver cancer can return after surgery
Liver surgery is often the best hope for people with early-stage liver cancer, yet many patients see their cancer come back within a few years. This study asks a simple but crucial question: does the body’s own inflammatory response to surgery unintentionally help leftover cancer cells grow and spread? The researchers focus on a signaling molecule called CCL11 and show how a surge of this molecule after surgery can both weaken anti-cancer immunity and make surviving tumor cells more aggressive.
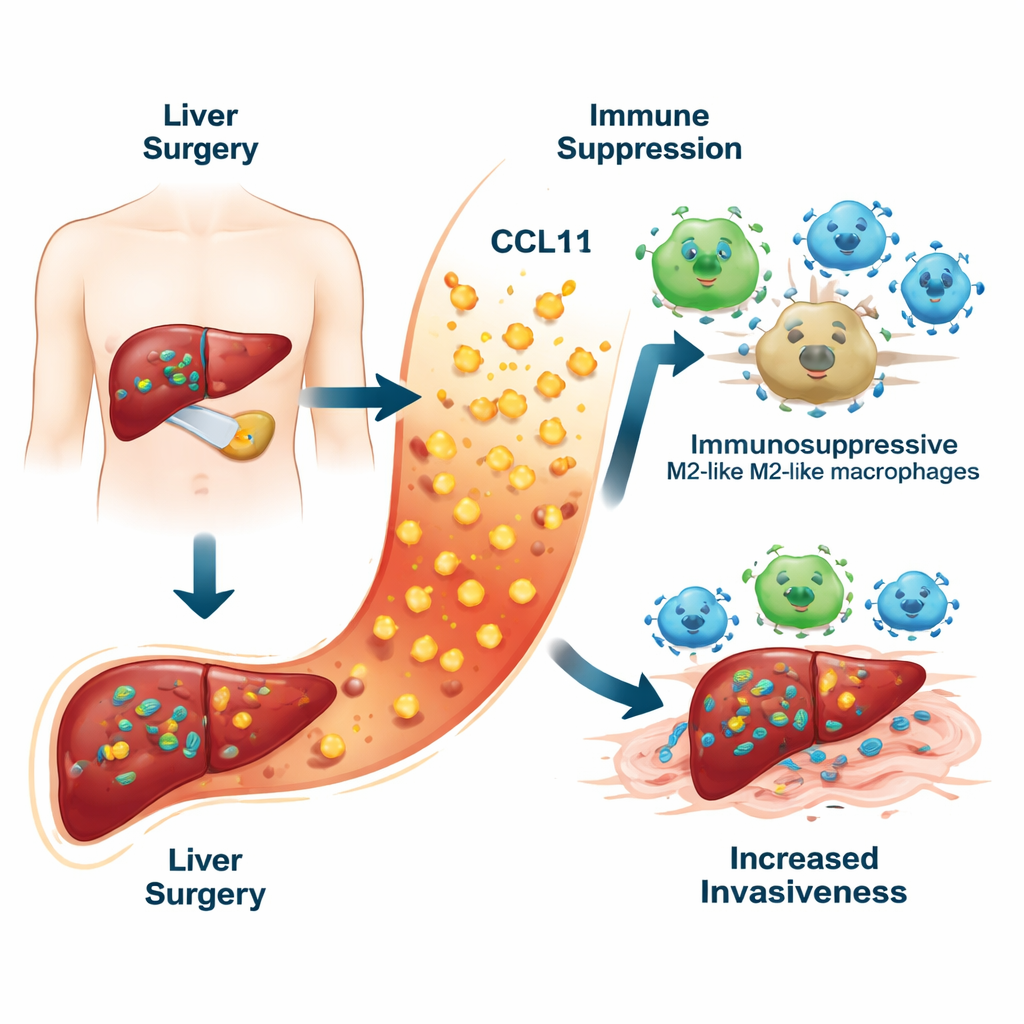
A hidden danger in the healing response
When surgeons remove a liver tumor, they must clamp blood vessels and injure surrounding tissue. This damage triggers a wave of inflammation that helps the liver heal. By measuring ten different inflammatory molecules in the blood of patients one week after surgery, the team found that one chemokine, CCL11, stood out. Patients whose liver cancer later recurred had significantly higher levels of CCL11 than those who remained cancer-free. High CCL11 levels were also linked with worse survival. When the researchers combined CCL11 with routine clinical data such as blood tests and tumor features, they built a risk “scorecard” that predicted five‑year recurrence more accurately than traditional models.
The liver tissue that looks normal still matters
Most attention in cancer research goes to the tumor itself, but this study shows that the surrounding “normal” liver tissue can be just as important. In samples from over 100 patients, CCL11 was actually higher in non-tumor liver than in the cancer nodules or in healthy donor livers. Patients whose nearby liver tissue made more CCL11 were more likely to have advanced disease, distant spread, and relapse after surgery. In mouse models that mimic the brief loss and return of blood flow during an operation, liver injury rapidly drove up CCL11 levels. Under low-oxygen conditions in the lab, liver-supporting cells such as myofibroblasts and blood-vessel lining cells also produced more CCL11, suggesting that surgical stress primes the entire liver remnant to become a pro-cancer environment.
How CCL11 turns immune defenders into bystanders
To see how CCL11 shapes immunity, the authors zoomed in on macrophages, versatile white blood cells that can either attack tumors or protect them. They found that in non-tumor liver tissue, higher CCL11 went hand in hand with more of a macrophage subtype that carries the marker CCR5 and displays a so‑called M2-like, tumor‑friendly profile. In patients, livers rich in these CCR5+ M2-like macrophages were linked to poorer survival. In cell culture, adding CCL11 pushed monocytes to become this immunosuppressive macrophage type, ramping up molecules such as PD‑L1 and IL‑10 and releasing other signals that attract and skew more immune cells. These CCL11‑conditioned macrophages then encouraged naïve helper T cells to turn into regulatory T cells, a population known for dampening anti-tumor responses. Mechanistic experiments showed that CCL11 activates a chain of signals inside macrophages that ends in the NF‑κB pathway and PD‑L1 production, helping shield cancer cells from immune attack.
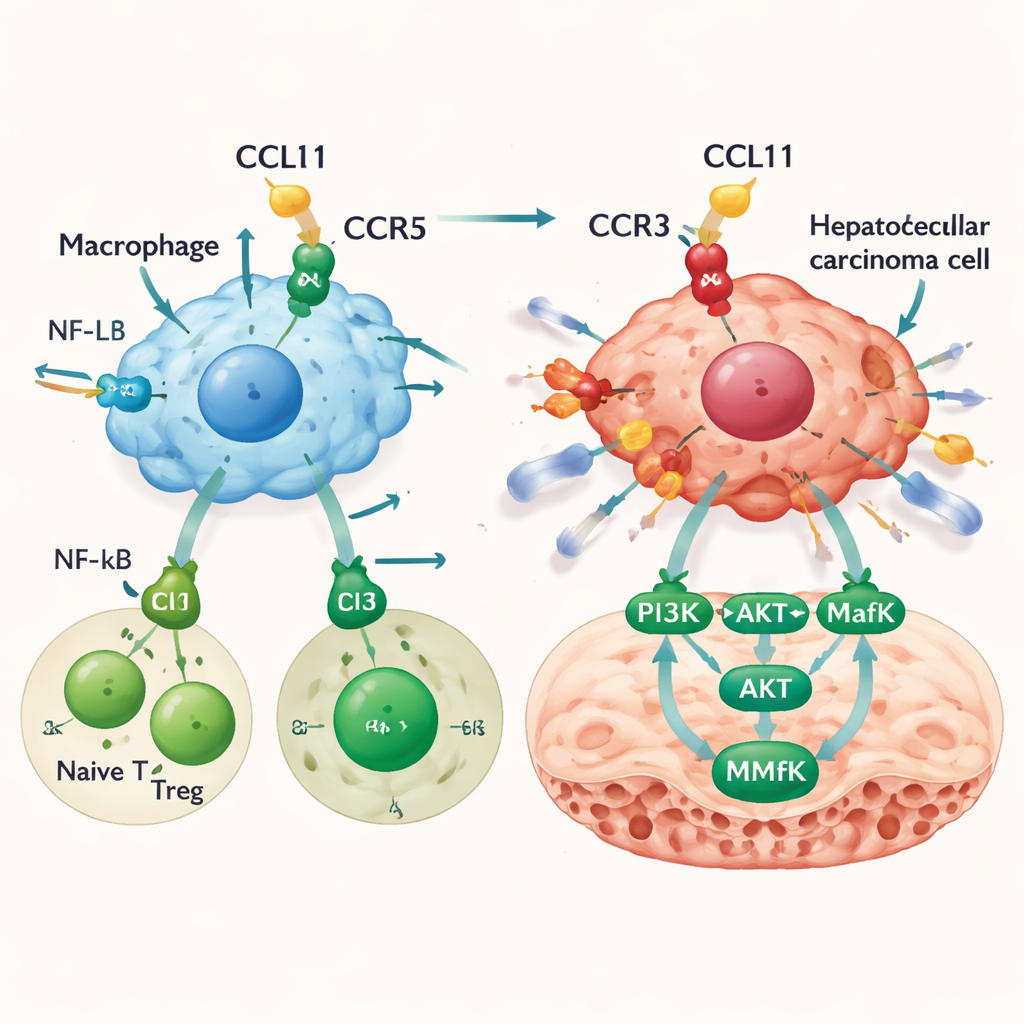
How CCL11 makes remaining cancer cells more invasive
CCL11 does not only reshape the immune landscape; it also acts directly on liver cancer cells. Although the cancer cells themselves make little CCL11, they carry one of its receptors, CCR3. In laboratory assays, CCL11 did not speed up liver cancer cell growth but did make them migrate and invade through barriers more readily, a hallmark of metastatic potential. Blocking the CCR3 receptor largely erased this effect. Gene analyses pointed to a pathway in which CCL11–CCR3 signaling switches on PI3K and AKT, which then activates a transcription factor called MafK. MafK in turn boosts production of MMP13, an enzyme that breaks down the surrounding tissue and opens paths for cancer spread. In mouse models, infusing CCL11 into the liver increased tumor burden and led to more metastases, along with higher levels of the PI3K–AKT–MafK–MMP13 pathway inside tumors.
Turning a risky signal into a treatment opportunity
Because CCL11 appears to both silence immune defenses and toughen up residual cancer cells, the researchers tested whether blocking it after surgery could change outcomes. In mice with liver tumors that were surgically removed, treatment with an antibody that neutralizes CCL11 dramatically reduced the rate of cancer returning in the liver and improved survival, without obvious side effects such as weight loss. Taken together, the findings present CCL11 as a master switch that links surgical injury to an immunosuppressive, invasion‑friendly liver environment. For patients, this work suggests that measuring CCL11 after surgery could help identify those at highest risk of relapse, and that drugs targeting the CCL11–CCR5/CCR3 axis might one day become part of strategies to keep liver cancer from coming back.
Citation: Wang, J., Yeung, O.WH., Qiu, W. et al. CCL11 promotes hepatocellular carcinoma recurrence after surgery by potentiating immunosuppressive CCR5 + CD206 + M2-like macrophages and promoting tumor invasiveness. Cell Death Dis 17, 236 (2026). https://doi.org/10.1038/s41419-026-08508-4
Keywords: liver cancer recurrence, CCL11 chemokine, tumor microenvironment, immune suppression, cancer metastasis