Clear Sky Science · en
Transcriptional factor ZMYM3 promotes hepatocellular carcinoma metastasis by upregulating CTTN and inducing invadopodia formation
Why this liver cancer study matters
Liver cancer is among the deadliest cancers worldwide, largely because it is often discovered late and has a strong tendency to spread. This study looks under the microscope and into the cell nucleus to ask a crucial question: what makes some liver tumors so good at breaking away and invading other parts of the body? By uncovering one key molecular driver of this spread, the research points to new ways doctors might one day predict which patients are at highest risk and design treatments to slow or stop metastasis.
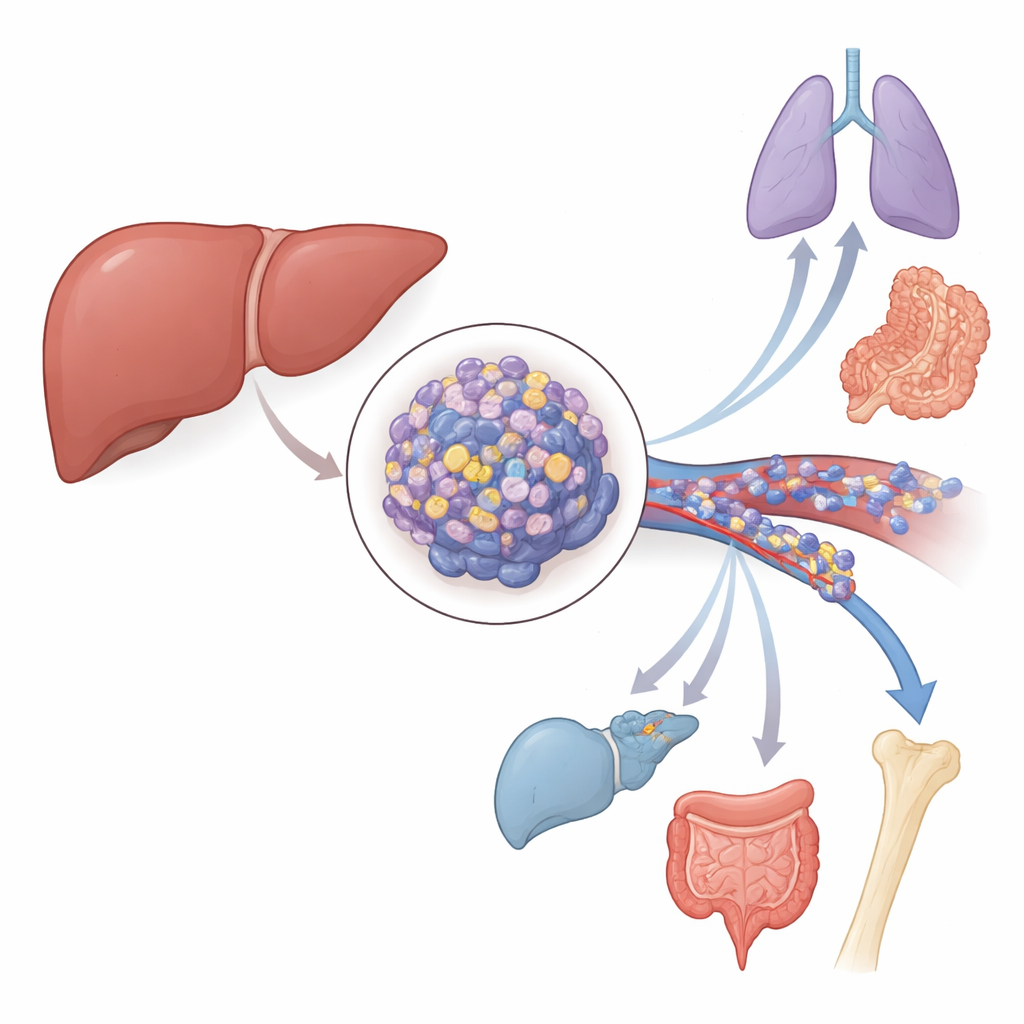
A hidden switch inside liver tumors
The researchers focused on hepatocellular carcinoma, the most common type of primary liver cancer. By mining large public cancer databases and analyzing patient tissue samples, they found that a gene called ZMYM3 is switched on much more strongly in liver tumors than in nearby healthy liver tissue. Patients whose tumors had higher ZMYM3 levels tended to have poorer overall survival and shorter periods without the disease coming back. The team also examined special clots formed by tumor cells inside the portal vein, a major blood vessel that drains the liver. In these highly invasive tumor clots, ZMYM3 levels were even higher than in the original liver tumors, tying this gene closely to the cancer’s ability to invade blood vessels and spread.
How ZMYM3 makes cancer cells more aggressive
To move beyond correlations, the scientists manipulated ZMYM3 levels in liver cancer cell lines. When they lowered ZMYM3, the cells grew more slowly, formed fewer colonies, and were more likely to undergo programmed cell death. When they boosted ZMYM3, the opposite happened: cells divided faster, resisted dying, and were better at migrating and invading through artificial barriers in the lab. In mouse models, tumors engineered to have extra ZMYM3 grew larger under the skin and produced more metastatic nodules in the lungs after being injected into the bloodstream. Together, these experiments show that ZMYM3 is not just a marker but an active promoter of liver cancer growth and spread.
Invading by growing tiny "feet"
One striking feature of aggressive cancer cells is their ability to grow tiny, actin-rich protrusions that dig into and dissolve the surrounding tissue. These structures, called invadopodia, act like cellular jackhammers, helping tumor cells chew through the supportive mesh around organs and slip into blood vessels. Using gene expression profiling, imaging of the cell skeleton, and functional assays, the researchers showed that ZMYM3 ramps up many genes linked to invadopodia and to a broader shape-shifting program known as epithelial–mesenchymal transition, which makes cells more mobile. When ZMYM3 levels were high, liver cancer cells formed more invadopodia and loosened their attachments to neighbors; when ZMYM3 was reduced, these invasive structures and behaviors diminished.
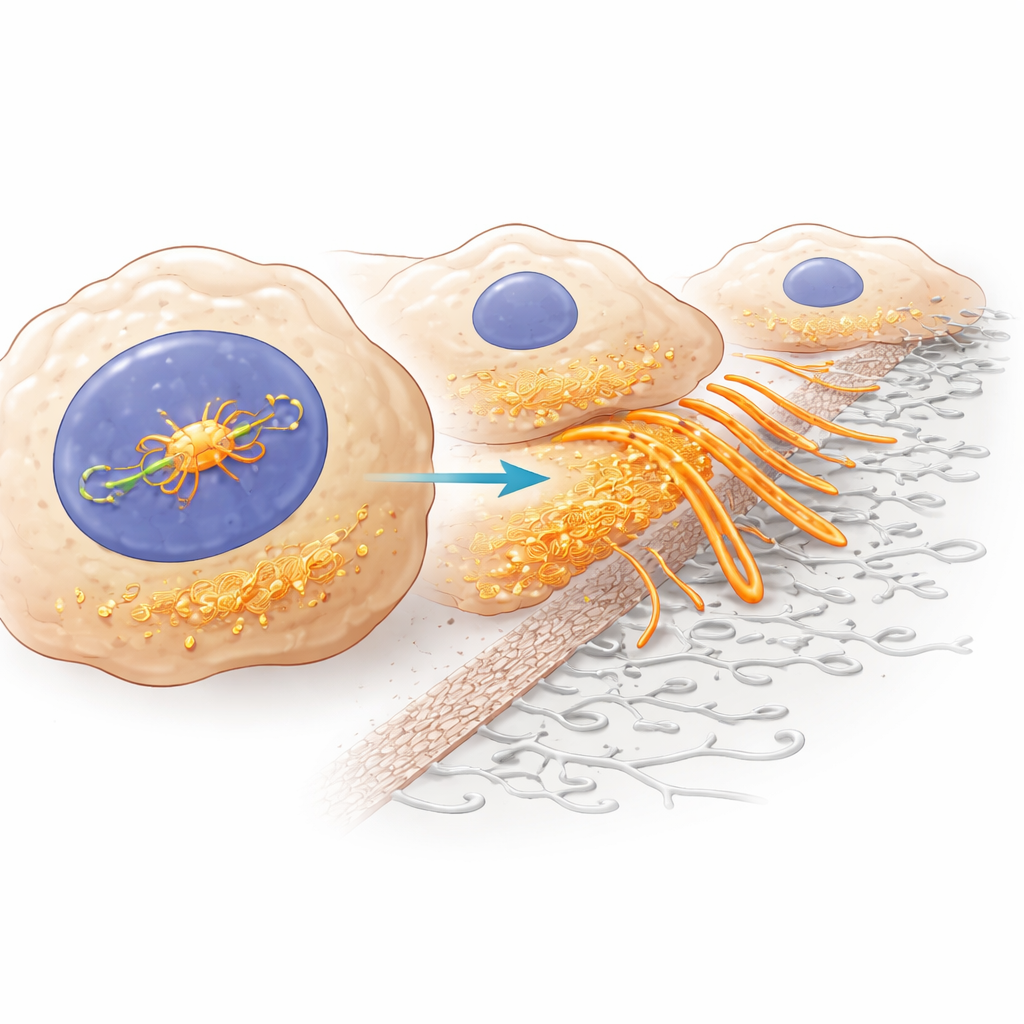
The key partner: a scaffold protein called cortactin
Digging deeper into the mechanism, the team used a technique that maps where proteins land on DNA across the genome. They discovered that ZMYM3 binds directly to the control region of another gene, CTTN, which encodes cortactin—a central organizer of invadopodia. Activating this region boosts cortactin production. In patient data, cortactin was also elevated in liver tumors and linked to worse outcomes, and its levels rose and fell in step with ZMYM3. When the scientists knocked down cortactin in cells that overproduced ZMYM3, the cancer cells lost much of their extra growth, survival, migration, and invasive power. This shows that a large part of ZMYM3’s impact is channeled through turning up cortactin and, in turn, strengthening invadopodia.
What this means for patients
For a layperson, the message is that this study identifies a molecular "accelerator"—ZMYM3—that helps liver cancer cells grow, resist death, and physically drill their way into blood vessels and distant organs. It does this largely by flipping on another gene, CTTN, which equips cancer cells with more powerful invasive "feet." Because high ZMYM3 and cortactin levels are tied to recurrence and poor survival, they could serve as warning flags to identify patients at higher risk of metastasis. In the longer term, drugs that interfere with this ZMYM3–cortactin pathway, or with invadopodia formation itself, might offer new ways to slow the spread of hepatocellular carcinoma and improve outcomes.
Citation: Zeng, F., Zhang, Z., Hu, T. et al. Transcriptional factor ZMYM3 promotes hepatocellular carcinoma metastasis by upregulating CTTN and inducing invadopodia formation. Cell Death Dis 17, 294 (2026). https://doi.org/10.1038/s41419-026-08506-6
Keywords: hepatocellular carcinoma, metastasis, ZMYM3, cortactin, invadopodia