Clear Sky Science · en
Cancer-associated fibroblasts promote osimertinib resistance in non-small cell lung cancer cells via METTL1-mediated NET1 m7G modification
Why some lung cancers stop responding to a "smart" drug
Targeted drugs like osimertinib have transformed treatment for many people with a common type of lung cancer, offering longer life with fewer side effects than traditional chemotherapy. Yet for most patients, these benefits are temporary: tumors eventually find ways to outsmart the drug. This study asks a deceptively simple question with big implications: is the tumor’s supporting tissue—specifically cells called cancer‑associated fibroblasts—quietly helping cancer cells escape the effects of osimertinib?
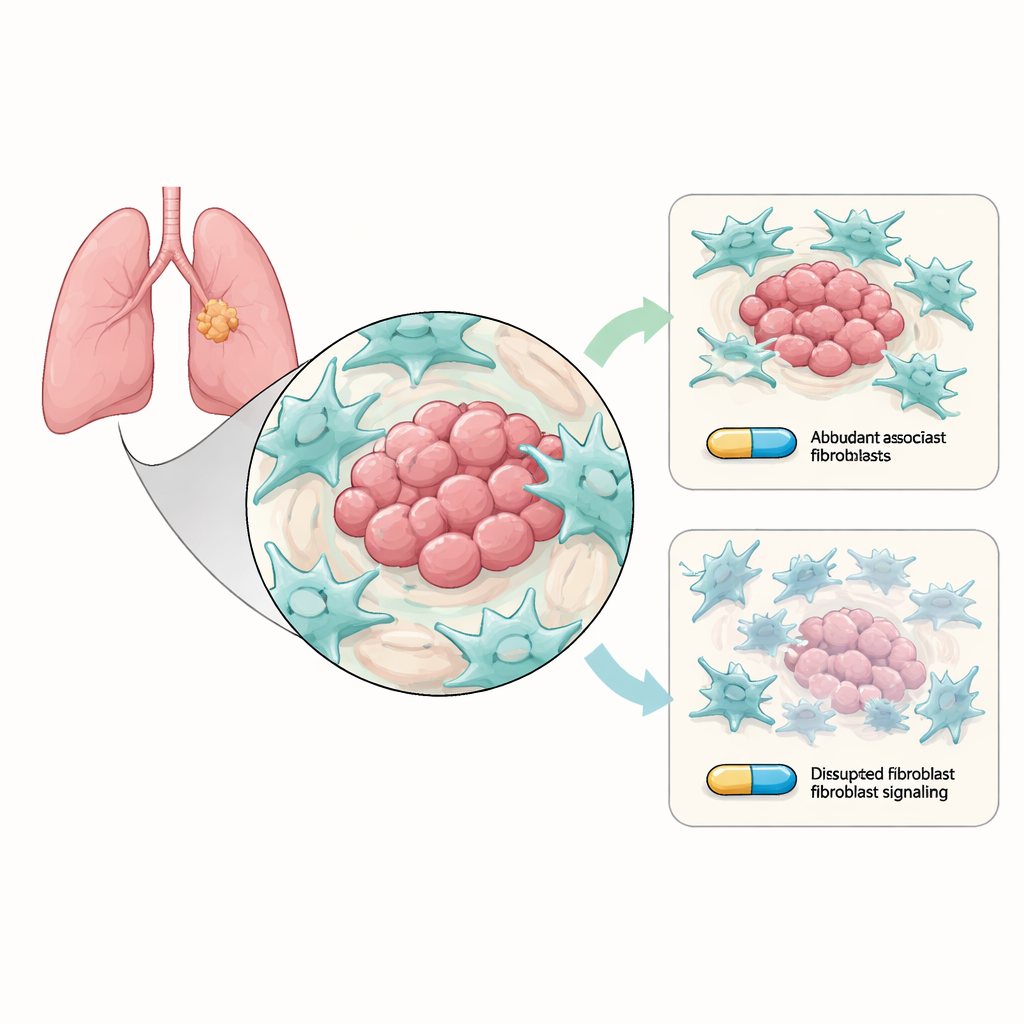
Helper cells that secretly shield the tumor
Non-small cell lung cancer grows within a bustling neighborhood of non-cancer cells known as the tumor microenvironment. Among the most abundant residents are cancer-associated fibroblasts (CAFs), connective-tissue cells that remodel the area around the tumor. The researchers grew lung cancer cells carrying the usual osimertinib-sensitive EGFR mutations together with CAFs in the lab. They found that when CAFs were present, cancer cells were harder to kill with osimertinib: they formed more colonies, self-destructed less often, and showed stronger "stem-like" traits linked to long-term survival and relapse. In other words, even though the drug still targeted the cancer cells, the surrounding fibroblasts quietly helped them endure.
A chemical tag on RNA that changes the rules
The team then looked beyond genes themselves to the chemical tags that decorate RNA—the working copies of genetic information inside cells. One such tag, called m7G, can influence how stable an RNA molecule is and how efficiently it is translated into protein. Using biochemical tests and genome-wide mapping, the researchers showed that CAFs increase overall m7G marking on RNAs inside lung cancer cells. This boost was driven mainly by a methyltransferase enzyme called METTL1, which places m7G marks on messenger RNAs. METTL1 levels were higher in lung tumors than in nearby normal tissue and were linked to more advanced disease and poorer patient survival, hinting that this enzyme does more than just fine-tune routine cell functions.
A secreted signal and a vulnerable molecular chain
How do CAFs turn up METTL1 in cancer cells? The study points to a protein called HMGB1, which CAFs release into their surroundings. Compared with cancer cells, CAFs secreted much larger amounts of HMGB1. When the researchers added HMGB1 to lung cancer cells, METTL1 levels and m7G marks rose; when they blocked HMGB1 in CAF-conditioned medium, this effect faded. Zooming in further, they identified one key RNA target of METTL1: NET1, a gene that helps drive cell growth and movement. CAFs increased both the m7G tagging and the abundance of NET1 RNA and protein, and METTL1 physically bound NET1 RNA to stabilize it. Less METTL1 meant less modified NET1, weaker cancer cell survival signals, and greater sensitivity to osimertinib.
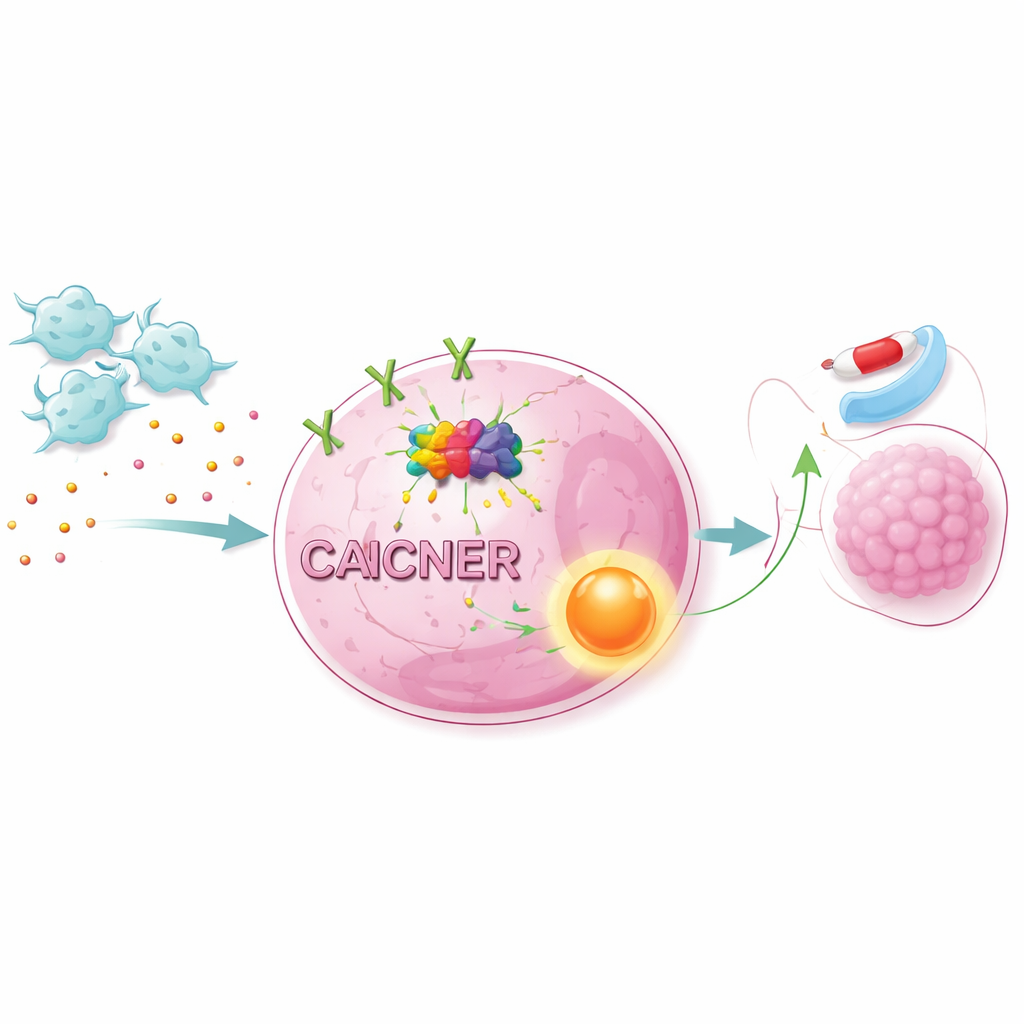
Turning on survival pathways inside cancer cells
The strengthened NET1 signal did not act alone. By combining large-scale RNA data with pathway analysis, the scientists showed that CAFs activate a major growth and survival route inside cells known as the AKT/NF-κB pathway. Blocking AKT or NF-κB with specific inhibitors made lung cancer cells more vulnerable to osimertinib, even in the presence of CAFs. Changing NET1 levels had similar effects: reducing NET1 made CAF protection weaker, while boosting NET1 enhanced resistance. In mice implanted with human lung cancer cells, adding CAFs made tumors grow faster and less responsive to osimertinib. Knocking down METTL1 in the cancer cells cut m7G marks, lowered NET1 and pathway activation, and sharply reduced CAF-driven tumor growth.
What this means for future lung cancer treatment
For people facing EGFR-mutant lung cancer, this work offers a new way to think about why powerful drugs like osimertinib eventually fail. Rather than blaming only new mutations in cancer cells, it highlights a molecular chain that starts outside the tumor cell—with CAFs releasing HMGB1—and runs through METTL1, m7G marks on NET1, and activation of survival pathways that blunt the drug’s impact. Interrupting this chain at any point could, in principle, restore or prolong sensitivity to treatment. While drugs that directly target METTL1, HMGB1, or specific m7G-marked RNAs are still in early stages, the study suggests that combining osimertinib with therapies aimed at the tumor’s supportive cells or their RNA-modifying signals may help keep this form of lung cancer under control for longer.
Citation: Qian, Y., Gong, Z., Jia, Y. et al. Cancer-associated fibroblasts promote osimertinib resistance in non-small cell lung cancer cells via METTL1-mediated NET1 m7G modification. Cell Death Dis 17, 248 (2026). https://doi.org/10.1038/s41419-026-08505-7
Keywords: osimertinib resistance, non-small cell lung cancer, cancer-associated fibroblasts, RNA methylation, METTL1 NET1 pathway