Clear Sky Science · en
ERRα-KDM5C restrains STING enhancer activity to modulate type I interferon signaling in breast cancer progression
Stopping Cancer Cells from Hiding
Cancer cells do more than just grow uncontrollably—they also learn to hide from the body’s immune system. This study uncovers how certain breast cancer cells dial down an important internal alarm system, called the STING pathway, which normally helps alert the immune system to danger. By understanding this “off switch,” scientists hope to find new ways to make tumors more visible to the immune system and improve the effects of existing treatments, including chemotherapy.
A Cellular Alarm System Under Pressure
The STING pathway acts like a built-in security system inside our cells. When DNA damage or viral-like signals are detected, STING helps trigger type I interferons—potent immune messengers that call in defenses such as killer T cells. Many tumors find ways to silence this alarm, making it easier for them to grow unchecked. The authors of this paper focused on breast cancer cells and asked: which molecules are responsible for keeping STING turned down, and how is this control wired into the cell’s genetic “switchboard” of enhancers—short DNA regions that boost nearby gene activity?
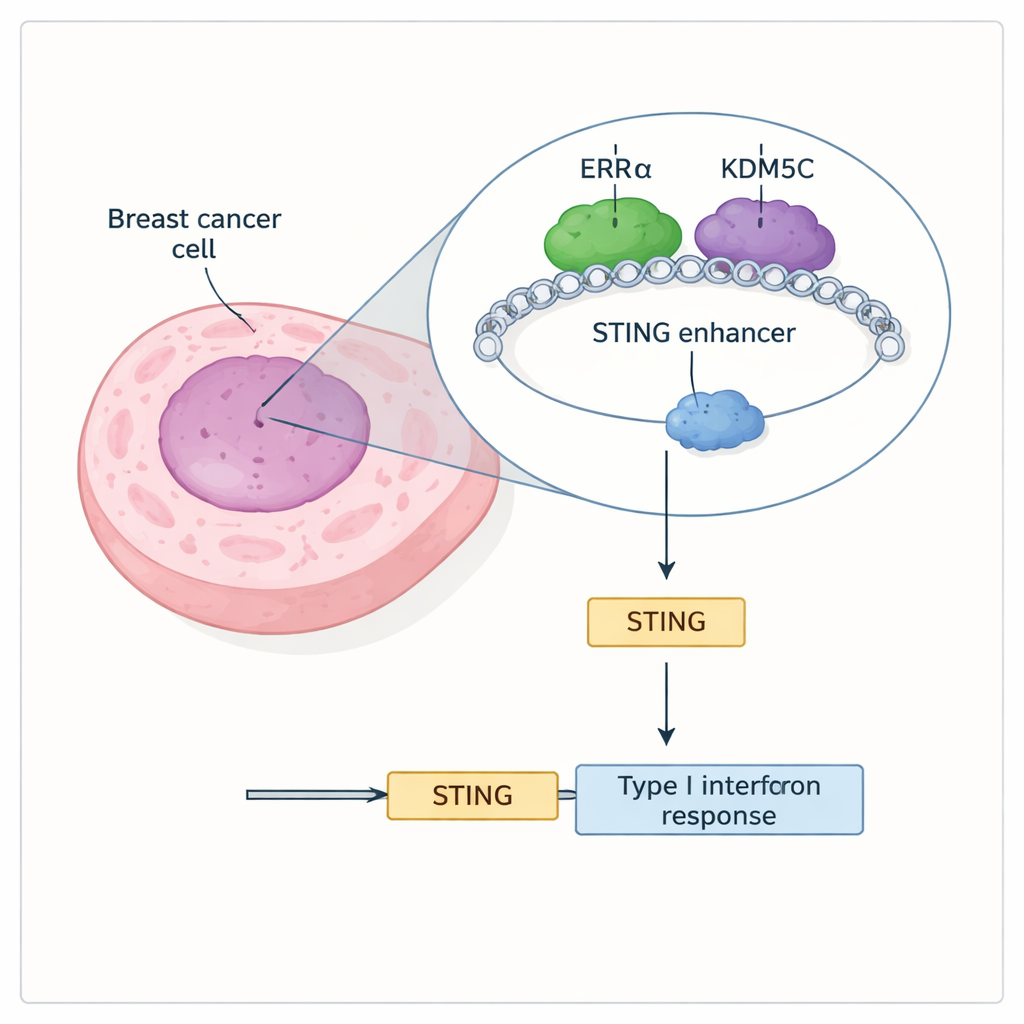
Two Proteins Team Up to Muffle STING
The researchers discovered that a nuclear receptor called ERRα and an enzyme called KDM5C physically interact and sit together on many active enhancer regions, including one that controls the STING gene. ERRα is known to help cancer cells rewire their metabolism and resist cell death, while KDM5C is a “chromatin editor” that erases a chemical mark (H3K4me3) associated with strong gene activation. Using genome-wide binding maps, the team showed that ERRα and KDM5C often co-occupy enhancer sites that carry marks of high activity and produce enhancer RNAs, a sign that these switches are on. At the STING enhancer in particular, both proteins are present, and their presence coincides with restrained STING output.
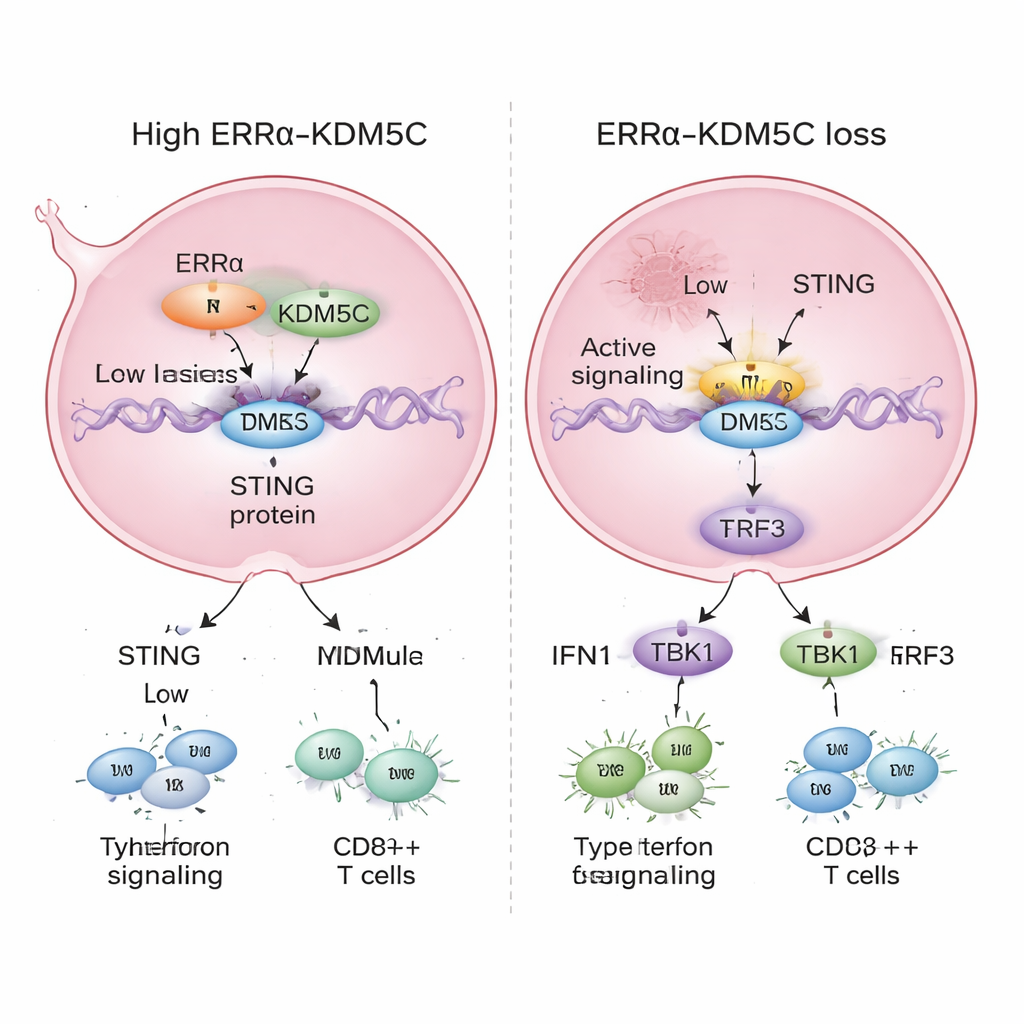
Releasing the Brake on Immune Signals
When the scientists reduced ERRα or KDM5C levels in breast cancer cells, the STING enhancer became overactive. Chemical marks associated with strong activation increased, those linked to a weaker state decreased, and enhancer RNA production went up. This was followed by a rise in STING gene and protein levels and stronger activation of its downstream pathway, including TBK1 and IRF3—key relay proteins that drive type I interferon production. As a result, cells began expressing higher levels of interferons and interferon-stimulated genes, which are central to antiviral defense and antitumor immunity. In other words, removing ERRα or KDM5C turned a muted alarm into a loud siren inside the cancer cells.
Slowing Tumor Growth and Boosting Therapy
These molecular changes had clear effects on how cancer cells behaved. Breast cancer cells with reduced ERRα or KDM5C grew more slowly and formed fewer colonies in culture. In mouse models, tumors derived from ERRα-depleted cells were smaller and lighter, indicating weaker growth in living animals. Importantly, when STING was also silenced, much of this growth inhibition was reversed, showing that activating STING is a key part of how ERRα and KDM5C loss restrains tumors. The team also tested a chemotherapy drug, etoposide, which causes DNA damage known to activate STING. Blocking ERRα made etoposide more effective at shrinking tumors, suggesting that loosening this brake makes standard treatments work better by amplifying immune-stimulating signals.
What This Means for Patients
By analyzing patient data, the researchers found that ERRα levels are higher in human breast cancers than in normal breast tissue, and that high ERRα expression is linked to poorer survival and fewer cancer-fighting CD8+ T cells inside tumors. Taken together, the work paints a clear picture: ERRα and KDM5C form a control module that keeps the STING alarm quiet, helping breast tumors grow and avoid immune attack. For a layperson, the key message is that some breast cancers use this duo to hide from the body’s defenses. Targeting ERRα, KDM5C, or their grip on the STING enhancer may help “unmask” tumors, strengthen the immune response, and increase the punch of existing therapies.
Citation: Xu, ZH., Chen, J., He, Y. et al. ERRα-KDM5C restrains STING enhancer activity to modulate type I interferon signaling in breast cancer progression. Cell Death Dis 17, 228 (2026). https://doi.org/10.1038/s41419-026-08499-2
Keywords: breast cancer, STING pathway, type I interferon, epigenetic regulation, tumor immunity