Clear Sky Science · en
mTOR-driven autophagy suppression defines metabolic vulnerability in CDK4/6 inhibitor-resistant HR+/HER2− breast cancer
Why this matters for breast cancer patients
Many women with advanced breast cancer now live longer thanks to drugs called CDK4/6 inhibitors, which slow down how fast tumor cells divide. Unfortunately, most tumors eventually learn to escape these medicines. This study asks a hopeful question: when breast cancer becomes resistant to CDK4/6 drugs, does it gain a new weakness that doctors could exploit with existing, relatively simple metabolic medicines such as Metformin or dichloroacetate (DCA)?
When cell-cycle drugs stop working
Doctors often treat the most common type of breast cancer—hormone receptor–positive, HER2-negative disease—with hormone therapy plus CDK4/6 inhibitors like Palbociclib or Ribociclib. These drugs target proteins that push cells through the division cycle. Over time, however, some cancer cells adapt and become strongly resistant, continuing to grow even in the presence of high drug doses. The researchers recreated this problem in the lab by exposing breast cancer cell lines (T47D and MCF7) to gradually increasing amounts of CDK4/6 inhibitors. They ended up with two kinds of survivors: partially resistant cells and a smaller group of highly resistant clones that no longer responded well to treatment.
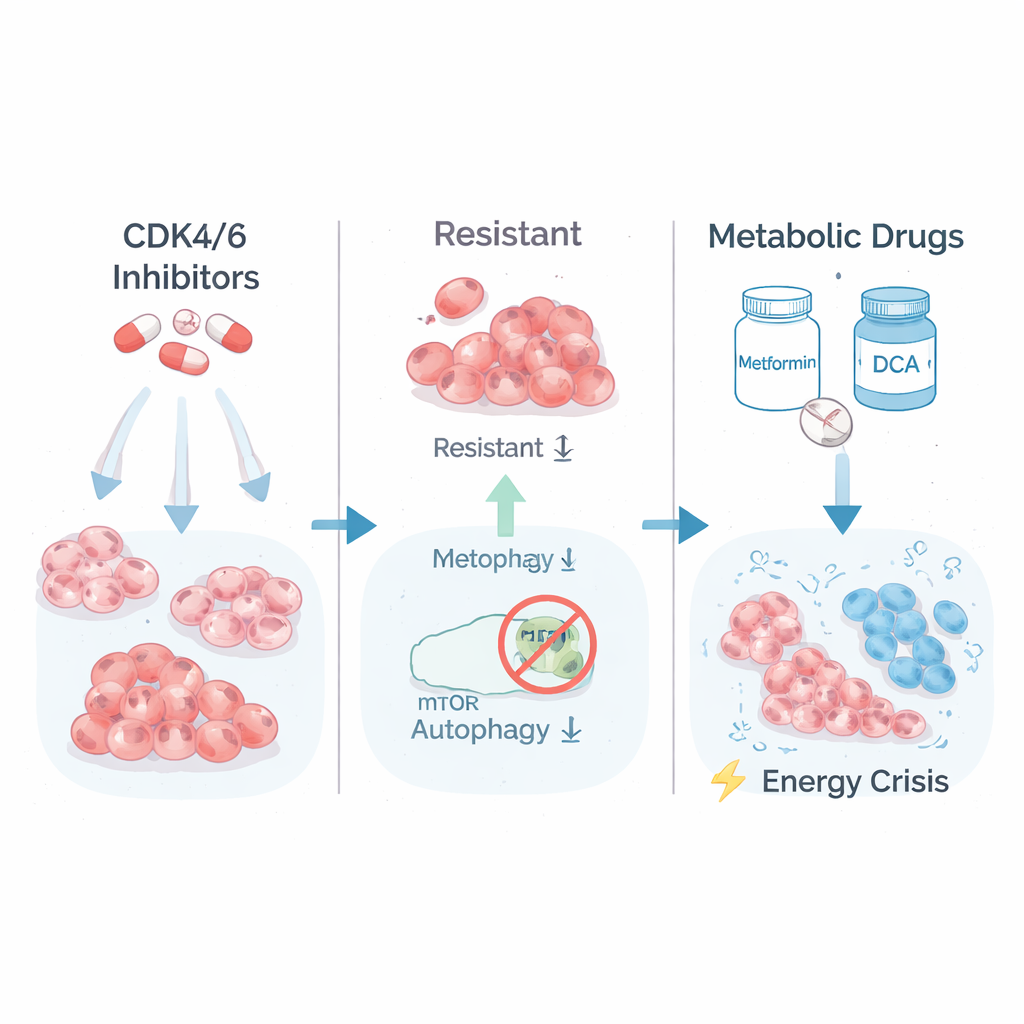
A hidden weakness in tumor energy use
To understand what made the highly resistant cells different, the team examined which genes were switched on or off. They found that the most resistant cells had cranked up pathways involved in burning sugar (glycolysis) and in growth signaling controlled by a protein complex called mTORC1. In other words, these cells lived fast, burning through fuel at a high rate to keep growing. The scientists then tested whether this “high-gear” metabolism could be turned against the cells. They treated the resistant and parental cells with Metformin, a common diabetes drug that stresses the cell’s energy factories, and DCA, which forces cells to process fuel more efficiently. Strikingly, the most drug-resistant clones were now the most vulnerable: their long-term growth in dish cultures collapsed under metabolic treatment, while parental and only partially resistant cells were much less affected.
Blocked self-cleaning and an energy crash
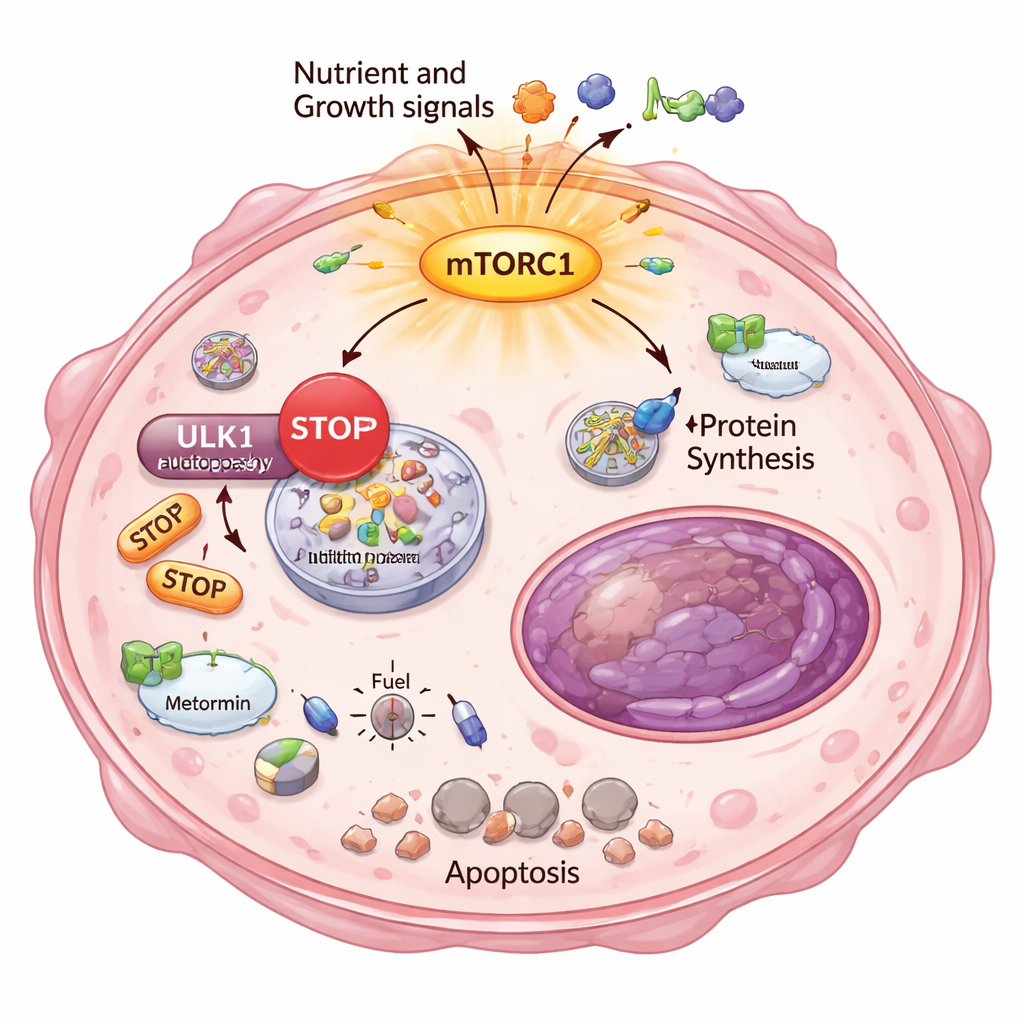
Why would these escapee cancer cells be so sensitive to energy stress? The answer lay in another basic survival process: autophagy, the cell’s built-in “recycling and cleanup” system. Normally, when energy runs low, cells break down and reuse their own components to stay alive. The researchers showed that in the highly resistant clones, mTORC1 was overactive and placed a chemical brake on ULK1, a key starter of autophagy. Using a sensitive reporter assay, they demonstrated that these cells could no longer properly turn on or ramp up autophagy, even when pushed with metabolic drugs. As a result, Metformin and DCA created a severe energy crisis: sensors of low energy lit up, fat production was shut down, and markers of irreversible cell death appeared, specifically in the strongly resistant cells.
Evidence from real patient tumors
Laboratory models do not always reflect what happens in patients, so the team turned to human breast tumor samples. They stained over a hundred cancers for a marker of mTORC1 activity (phosphorylated 4E-BP1) and for p62, a protein that builds up when autophagy is blocked. Tumors with high mTORC1 activity tended to have more p62, supporting the idea that an “overdriven growth signal plus poor recycling” pattern exists in real breast cancers, not just in cell lines. This suggests that a subset of patients may carry tumors with the same metabolic weakness seen in the lab.
What this could mean for future treatments
For a layperson, the key message is that when breast cancers evolve to escape powerful CDK4/6 drugs, they may also paint themselves into a metabolic corner. By switching growth signals permanently on and switching cellular recycling off, these tumors become heavily dependent on a constant energy supply. The study shows that this setup can be exploited with metabolic medicines like Metformin and DCA, which are already well known in other diseases. Importantly, the authors propose that simple tissue tests for mTORC1 activity and autophagy markers could help identify patients whose resistant tumors are primed for this kind of metabolic attack, opening the door to more personalized rescue treatments after CDK4/6 inhibitors fail.
Citation: von Wichert, L., Stroh, A., Witt, M. et al. mTOR-driven autophagy suppression defines metabolic vulnerability in CDK4/6 inhibitor-resistant HR+/HER2− breast cancer. Cell Death Dis 17, 235 (2026). https://doi.org/10.1038/s41419-026-08496-5
Keywords: breast cancer, CDK4/6 inhibitor resistance, mTOR, autophagy, metabolic therapy