Clear Sky Science · en
Splice-switching of the oncogenic BCS1L isoform suppresses ovarian cancer progression by disrupting mitochondrial function
Power Plants Inside Cancer Cells
Every cell in our body depends on tiny power plants called mitochondria. Cancer cells, which grow and spread quickly, are especially hungry for energy. This study uncovers how ovarian cancer rewires one of its key mitochondrial helpers, and shows that gently flipping a molecular “switch” on this helper can starve tumors while largely sparing healthy tissue.
Why Tumors Lean on Their Engines
Ovarian cancer cells, like many cancers, can flexibly tap different energy sources. By combing through large patient databases and single-cell profiles, the researchers found that many ovarian tumors run their mitochondria in a high-gear mode known as oxidative phosphorylation, or OXPHOS. Tumors with this boosted mitochondrial activity were more aggressive and more common in advanced disease. Among the many proteins that build the mitochondrial power machinery, one stood out: BCS1L, a chaperone that helps assemble Complex III, a central hub in the cell’s energy pipeline. High-OPXHOS tumors and certain tumor cell subtypes showed especially strong BCS1L activity, hinting that cancer cells were leaning heavily on this helper to keep their engines humming.
Two Faces of a Mitochondrial Helper
The team discovered that the BCS1L gene can produce two versions, or isoforms, of its protein. The full-length form, BCS1L-L, carries a built-in address tag that sends it into mitochondria, where it supports proper Complex III assembly and strong energy output. The shorter form, BCS1L-S, lacks a key segment and cannot reach the mitochondria; instead, it sits in the cell’s fluid and nucleus and does not support power production. In healthy tissues, the short version is common, but in ovarian cancers the balance flips: the long, mitochondria-bound version dominates. When cells were forced to make more BCS1L-L, their mitochondria generated more energy, maintained stronger membrane potential, produced fewer damaging oxygen byproducts, and resisted cell death. When BCS1L was reduced, mitochondria fragmented, energy production fell, harmful oxygen molecules rose, and cancer cells were more likely to die.
How Cancer Rewires the Message
Producing either the long or short BCS1L version depends on how the cell edits its RNA message, a process called splicing. The researchers searched for proteins that touch the BCS1L RNA and influence this choice. They zeroed in on USP39, a splicing factor already linked to several cancers. In ovarian cancer cells, USP39 binds near the RNA segment that becomes BCS1L’s exon 2 and encourages its inclusion, thereby favoring the long, mitochondria-targeted version. When USP39 was silenced, cells skipped this segment more often, shifting toward the short BCS1L-S form. This shift weakened Complex III, reduced mitochondrial respiration and ATP output, increased oxidative stress, and triggered cancer cell death. Restoring BCS1L-L in these cells rescued much of their mitochondrial function and survival, showing that USP39 drives a critical mitochondrial switch largely through BCS1L.
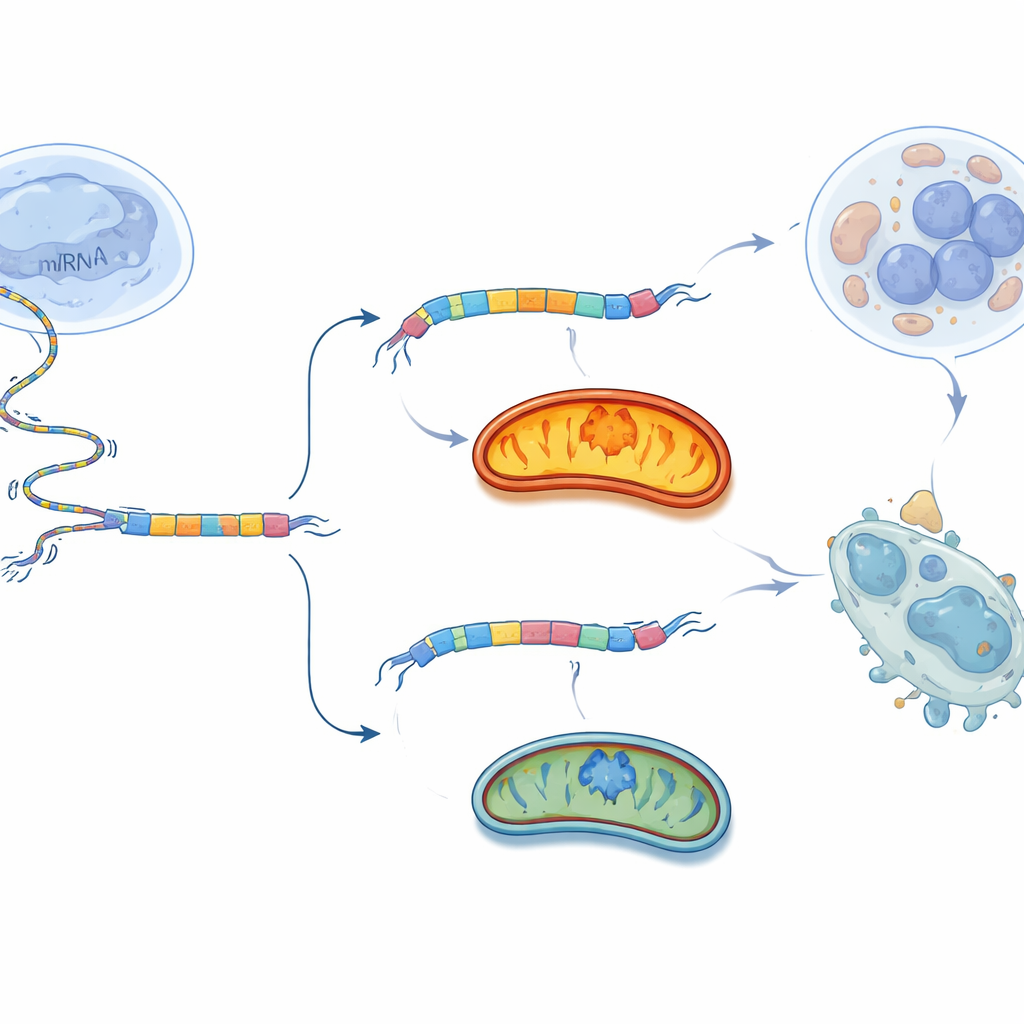
Turning the Switch Against the Tumor
Rather than blocking mitochondria broadly, which could harm many tissues, the team tried a more surgical tactic: nudging the splice choice of BCS1L itself. They designed short pieces of genetic material called antisense oligonucleotides (ASOs) that bind just around exon 2 of the BCS1L RNA. These ASOs act like tiny clothespins, changing how the splicing machinery reads the message and encouraging the cell to skip exon 2. In ovarian cancer cells grown in dishes, the best ASO strongly reduced the long BCS1L-L form and boosted the short BCS1L-S form. As a result, mitochondrial respiration and ATP production dropped, harmful oxygen levels rose, and many more cells underwent programmed death. When the same ASO was injected into ovarian cancer tumors in mice, tumor growth slowed and tumors weighed less, with a much stronger effect on cancer cells than on normal fibroblasts.
What This Could Mean for Patients
In simple terms, this work shows that many ovarian cancers depend on a “turbo” version of a mitochondrial helper protein to fuel their growth. A splicing factor, USP39, tilts the cell’s RNA editing machinery so that more of this turbo version is made, keeping the tumor’s power plants in top shape. By using precisely designed ASOs to flip the splicing choice back toward the weaker version, researchers can selectively cripple cancer cell mitochondria, triggering their death while leaving most normal cells less affected. Although delivery of such ASO drugs to tumors remains a major challenge, this study points to the BCS1L splicing switch as a promising and highly specific new handle for treating ovarian cancer.
Citation: Xu, M., Wang, Z., Yang, S. et al. Splice-switching of the oncogenic BCS1L isoform suppresses ovarian cancer progression by disrupting mitochondrial function. Cell Death Dis 17, 293 (2026). https://doi.org/10.1038/s41419-026-08495-6
Keywords: ovarian cancer, mitochondria, RNA splicing, BCS1L, antisense oligonucleotides