Clear Sky Science · en
NSD2 inhibits the expression of PD-L1 via oxidative phosphorylation to control immune surveillance in hepatocellular carcinoma
Why this liver cancer story matters
Hepatocellular carcinoma, the most common form of liver cancer, is often diagnosed late and resists many treatments. Immunotherapy drugs that block the PD-L1 “invisibility cloak” on tumor cells help some patients, but not most. This study uncovers an unexpected player, a protein called NSD2, that links how liver cancer cells make energy to how visible they are to the immune system. Understanding this link may point to new ways to boost current therapies and identify which patients are likely to benefit.
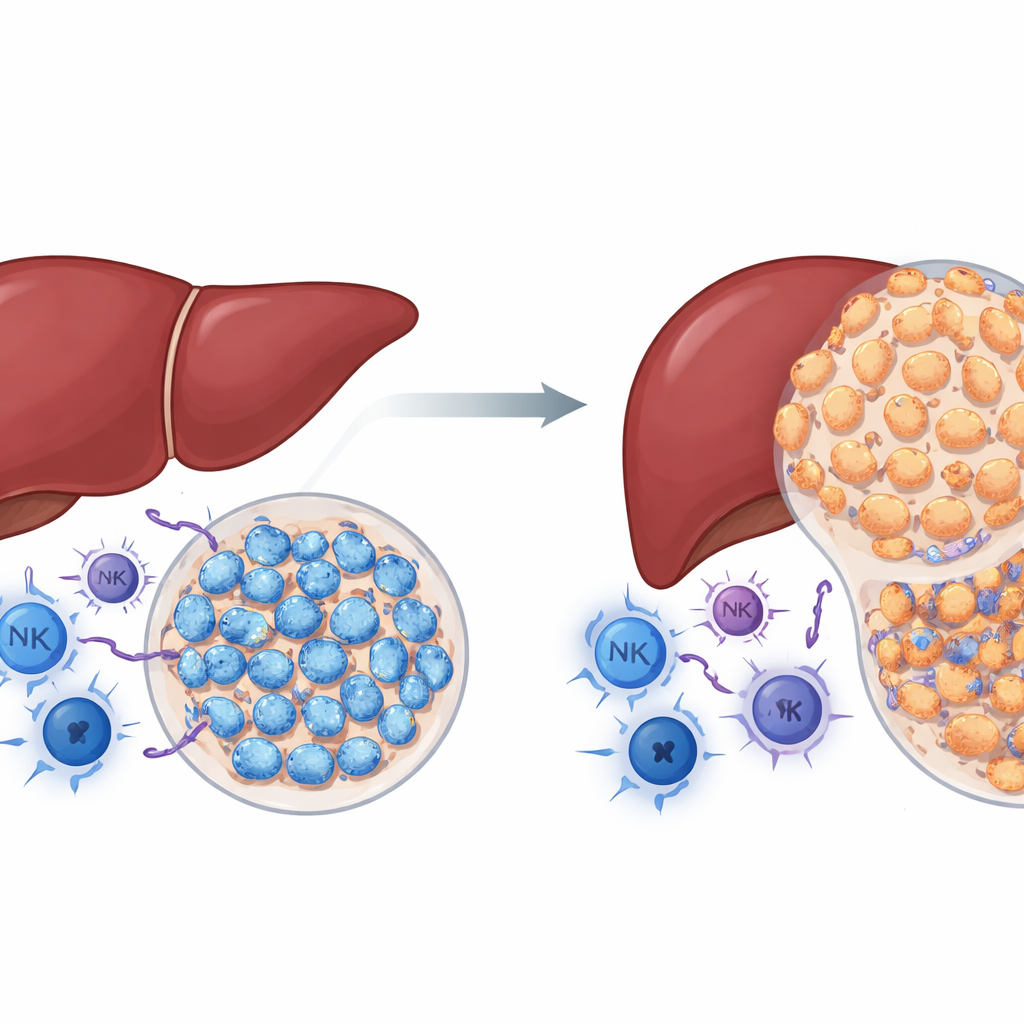
A surprising role for a known cancer protein
NSD2 is an enzyme that modifies histones, the proteins around which DNA is wrapped. In many cancers, NSD2 has been viewed as a driver of tumor growth. The authors asked whether the same is true in liver cancer. Using genetically engineered mice in which NSD2 is turned on specifically in liver cells, they treated animals with a chemical that reliably induces liver tumors. Contrary to expectations, mice with extra NSD2 developed far fewer and smaller tumors than normal mice, and their livers showed less damage, scarring and cell proliferation. At the same time, tumors from NSD2-overexpressing livers contained more cancer-fighting immune cells, including helper and killer T cells and natural killer cells. These findings suggested that, in the intact immune system, NSD2 behaves less like an accelerator and more like a brake on liver cancer.
Energy factories and fuel use in tumor cells
To understand how NSD2 restrains tumors, the team profiled gene activity in liver tissue. They found that NSD2 overexpression strongly dampened genes involved in oxidative phosphorylation, the process by which mitochondria—the cell’s “power plants”—generate energy using oxygen. Follow-up experiments in mouse and human liver cancer cell lines showed that boosting NSD2 lowered the expression of many oxidative phosphorylation genes, reduced oxygen consumption and ATP production, and dropped mitochondrial membrane potential, a sign of weaker mitochondrial activity. At the same time, these cells consumed less glucose but did not shift their reliance toward glycolysis, indicating a net reduction in energy output. Loss of NSD2 had the opposite effects, strengthening mitochondrial power and fuel use.
How NSD2 rewires energy and immune visibility
Diving deeper, the researchers combined genome-wide chromatin mapping with gene-expression data and identified a set of genes directly activated when NSD2 is present. Two of these, Camk2d and Prkce, are known to restrain mitochondrial activity. NSD2 increased a particular histone mark (H3K36me2) at their promoters, boosting their expression. Reintroducing these genes into NSD2-deficient liver cancer cells reduced mitochondrial oxygen use and ATP production, confirming that they act as NSD2’s downstream brakes on oxidative phosphorylation. Importantly, previous work had suggested that highly active mitochondria favor high PD-L1 on tumor cells. Consistent with this, the authors found that NSD2 overexpression reduced PD-L1 levels, while NSD2 loss raised PD-L1 on liver cancer cells and in tumor tissue.
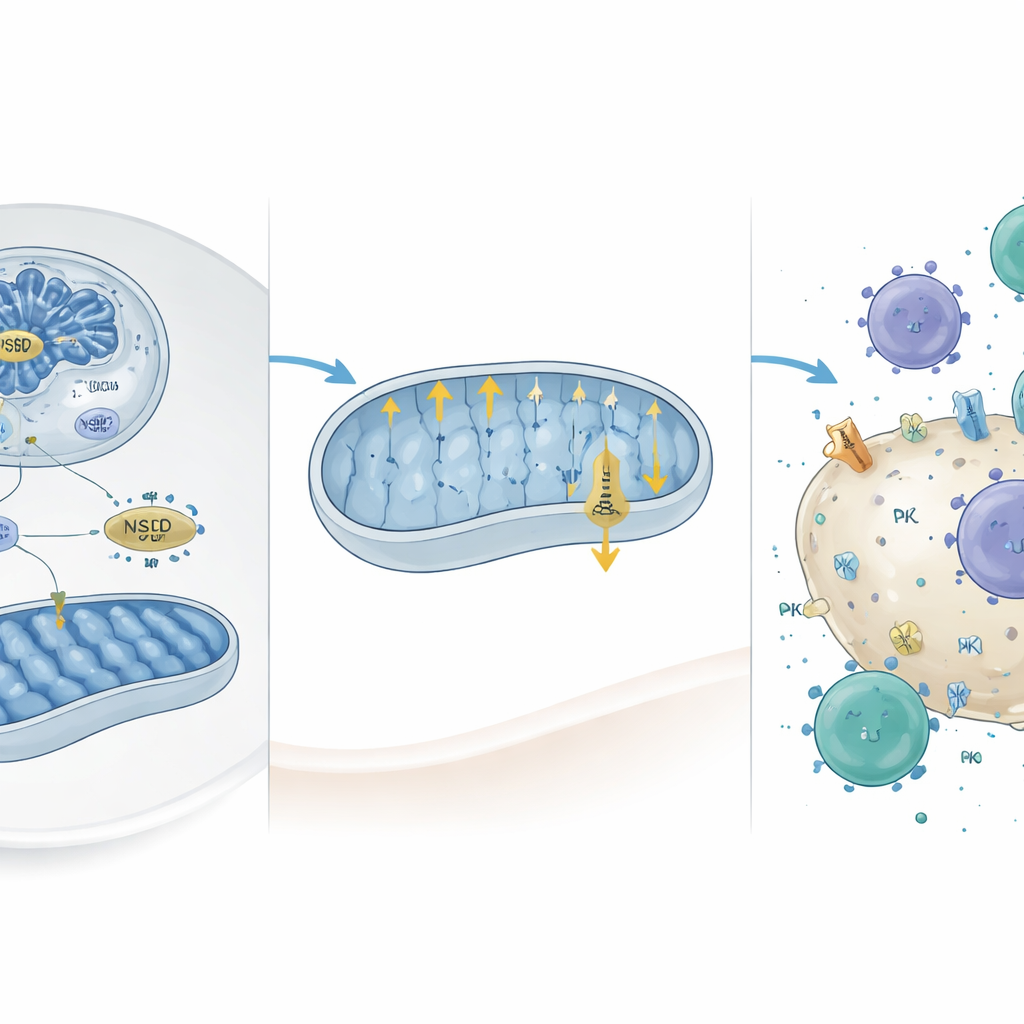
Turning down the shield and awakening immune attack
The functional impact of these molecular shifts was tested in mice receiving liver injections of cancer cells engineered to gain or lose NSD2. Cells lacking NSD2 rapidly formed larger tumors, expressed more PD-L1 and were infiltrated by fewer T cells and natural killer cells, especially activated killer T cells. When the researchers chemically blocked mitochondrial complex V, a key component of oxidative phosphorylation, PD-L1 levels in NSD2-deficient cells dropped back toward normal and immune cells were better able to induce tumor cell death. Similarly, overexpressing Camk2d or Prkce in NSD2-lacking cells, or treating tumors with a PD-L1–blocking antibody, reduced tumor size and restored immune-cell infiltration. In patient datasets, low NSD2 expression was linked to poorer survival but, intriguingly, to a better response to PD-L1–targeting therapy, suggesting that NSD2 status could help predict who benefits from such drugs.
What this means for future liver cancer care
For a general reader, the key message is that this study redefines NSD2 as a tumor-suppressing factor in liver cancer, at least in the presence of a working immune system. By dialing down the cell’s oxygen-based energy production, NSD2 indirectly lowers PD-L1 on tumor cells, removing part of their invisibility cloak and allowing immune cells to recognize and attack them. This newly described NSD2–energy–PD-L1 chain suggests two practical ideas: first, patients whose tumors lack NSD2 may be especially good candidates for PD-L1–blocking drugs; and second, combining drugs that inhibit mitochondrial energy production with immunotherapy could offer a powerful one-two punch against liver cancer.
Citation: Zhang, W., Feng, W., Ma, C. et al. NSD2 inhibits the expression of PD-L1 via oxidative phosphorylation to control immune surveillance in hepatocellular carcinoma. Cell Death Dis 17, 284 (2026). https://doi.org/10.1038/s41419-026-08490-x
Keywords: hepatocellular carcinoma, NSD2, oxidative phosphorylation, PD-L1, tumor immunotherapy