Clear Sky Science · en
SNRPD2-mediated regulation of DDX39B splicing promotes endometrial cancer progression by suppressing the activation of CTSC cryptic exons
Why Hidden Messages in Cancer Genes Matter
Endometrial cancer, a common cancer of the uterus, is rising worldwide, yet many patients with advanced disease still have few effective options. This study uncovers how a subtle process inside cells—how they cut and paste genetic messages—can drive endometrial tumors to grow and spread. By revealing a fragile "wiring diagram" that cancer cells rely on, the work points to new ways to shut tumors down using precision genetic medicines.
A Growing Cancer in Need of New Answers
Endometrial cancer is now the most frequent gynecologic cancer in many Western countries, with cases and deaths steadily increasing in both the West and China. Surgery, radiation, chemotherapy, hormone treatment and immunotherapy are all used, but they are far from perfect. Removing the uterus ends fertility, hormonal treatments often fail, and aggressive tumors commonly come back. These challenges have pushed researchers to look deeper inside cancer cells for molecular weak spots that can be targeted with more precise therapies.
The Cell’s Splicing Machinery as a Weak Link
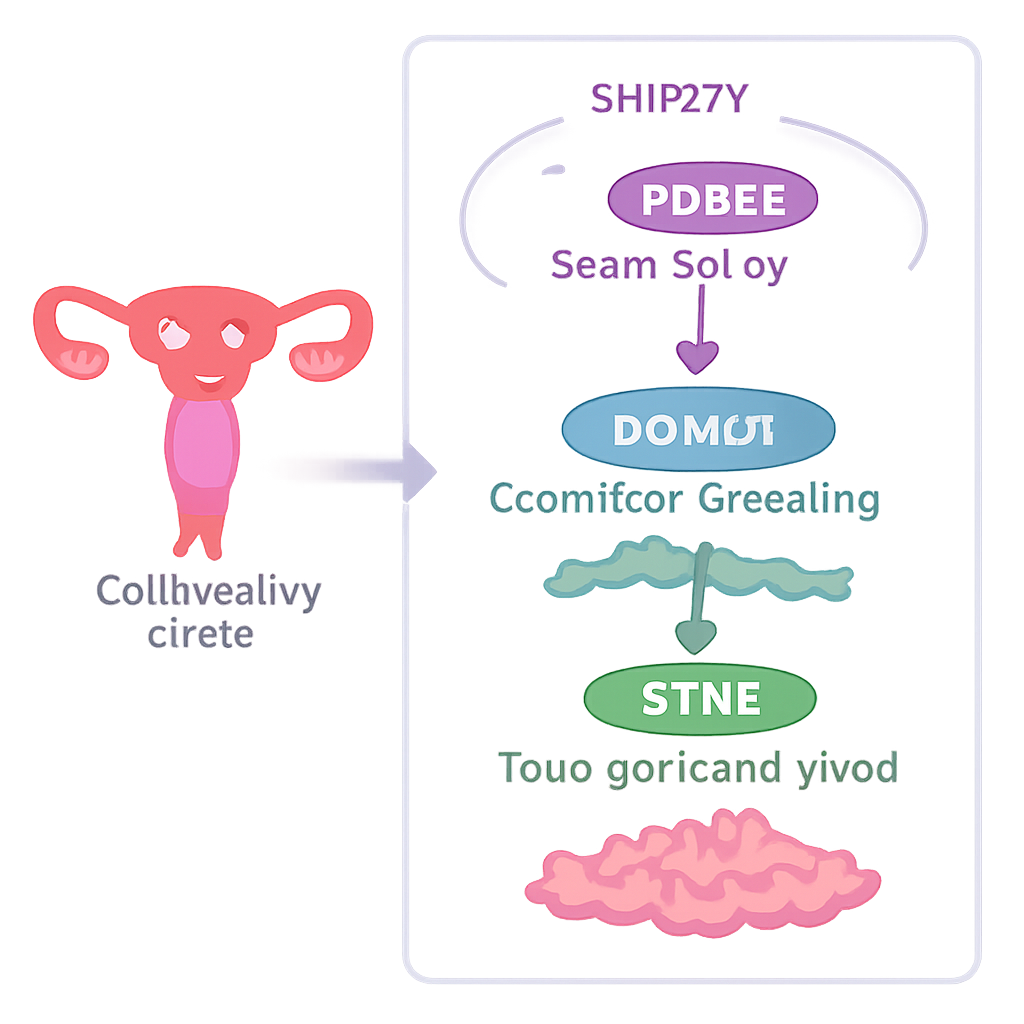
To function, our genes are first copied into long RNA molecules that must be trimmed and stitched together in a process called splicing. Specialized protein machines called spliceosomes decide which pieces are kept and which are thrown away, effectively editing the script that cells use to make proteins. The team focused on a spliceosome component called SNRPD2, part of a family of “Sm” proteins that help assemble the splicing machinery. By mining large cancer gene and protein databases and examining patient samples, they found that SNRPD2 levels are strikingly higher in endometrial tumors than in normal uterine lining, and that patients whose tumors have more SNRPD2 tend to fare worse.
Switching Off a Master Editor Slows Tumors
To test whether SNRPD2 is simply a bystander or an active driver, the researchers turned it down in endometrial cancer cell lines grown in the lab. When SNRPD2 was silenced, cells divided more slowly, formed fewer colonies, and were less able to move and invade through membranes—behaviors linked to metastasis. When these altered cells were implanted into mice, the resulting tumors were much smaller and showed fewer cells in active division. Importantly, the team designed antisense oligonucleotides—short, drug-like pieces of synthetic DNA—that specifically target SNRPD2’s RNA. In a patient-derived xenograft model, in which human tumor fragments grow in immune-deficient mice, these antisense drugs sharply reduced SNRPD2 levels and tumor size, suggesting that SNRPD2 could be drugged in a clinically meaningful way.
A Three-Step Chain from Splicing Error to Cancer Growth
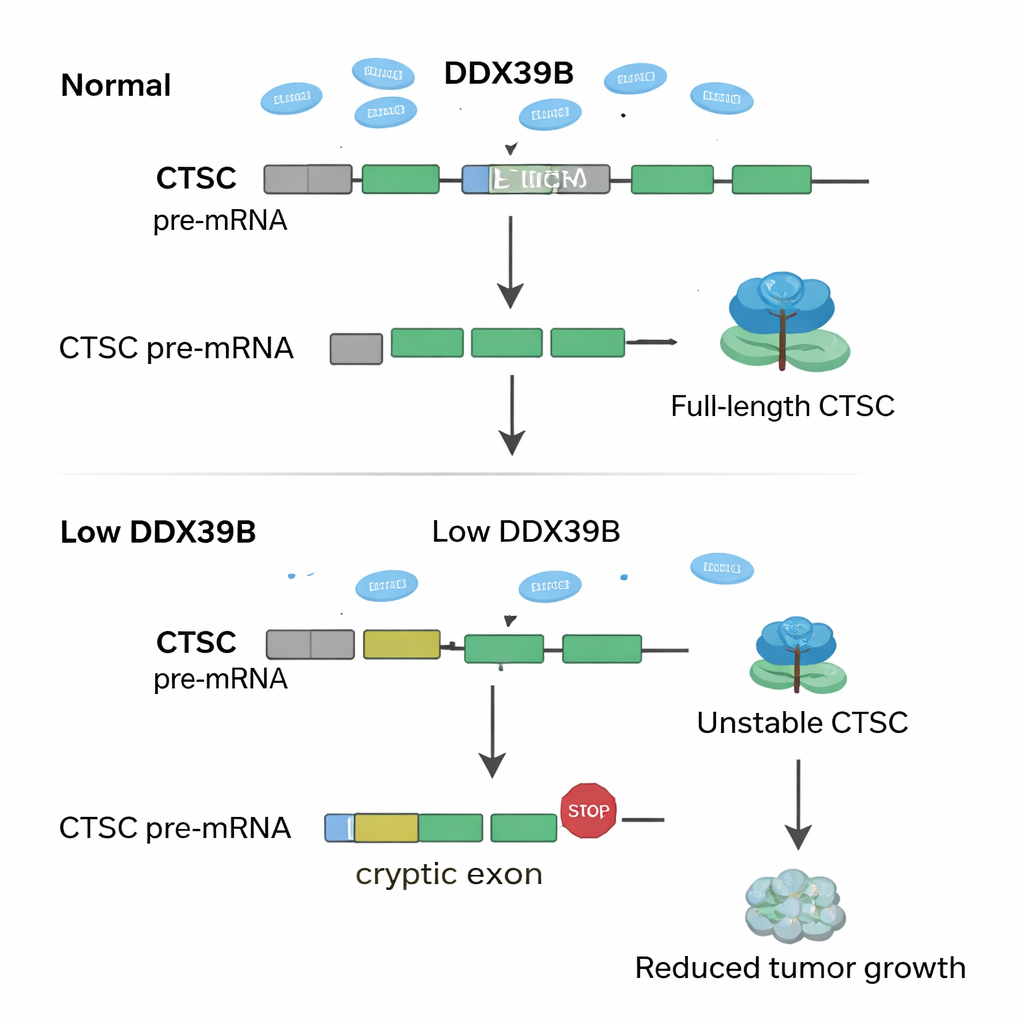
Diving into the genetic data, the investigators asked how SNRPD2 loss rewires RNA messages across the genome. They discovered that when SNRPD2 is reduced, a key RNA-processing enzyme called DDX39B begins to misfire. Normally, DDX39B helps remove a particular intron—an RNA segment that should be cut out—from its own message. Without enough SNRPD2, this intron is left in, creating a faulty version of DDX39B RNA that the cell quickly destroys. Lower DDX39B then has a ripple effect on another gene, CTSC, which encodes a protein-cutting enzyme known to support tumor growth and spread. Under healthy conditions, a hidden “cryptic” exon inside CTSC’s RNA is ignored, allowing cells to make full-length, active CTSC protein. With reduced DDX39B, this cryptic exon is mistakenly included, inserting premature stop signals into the RNA. The malformed CTSC message is degraded before it can produce much protein, and cancer cells lose some of their aggressive edge.
What This Means for Future Treatments
Put simply, the authors uncover a relay system—SNRPD2 → DDX39B → CTSC—that endometrial cancer cells exploit to thrive. High SNRPD2 keeps DDX39B abundant; DDX39B in turn prevents CTSC from using damaging cryptic exons, preserving full-strength CTSC protein that helps tumors grow and spread. When SNRPD2 is blocked, this chain collapses, CTSC levels fall, and tumors weaken. For non-specialists, the key idea is that cancer cells depend on very precise RNA editing, and even small forced “mistakes” can selectively poison their growth. Targeting SNRPD2 with antisense drugs, or directly forcing cryptic exon use in CTSC, could offer new, more tailored therapies for patients with endometrial cancer.
Citation: Li, Y., Chen, Z., Liu, Y. et al. SNRPD2-mediated regulation of DDX39B splicing promotes endometrial cancer progression by suppressing the activation of CTSC cryptic exons. Cell Death Dis 17, 239 (2026). https://doi.org/10.1038/s41419-026-08489-4
Keywords: endometrial cancer, RNA splicing, SNRPD2, DDX39B, CTSC