Clear Sky Science · en
DNMT2 inhibits anaplastic thyroid cancer progression by downregulating 5’tiRNAGly-GCC production
Why this hidden RNA chemistry matters
Anaplastic thyroid cancer is one of the deadliest human cancers, often killing patients within months despite surgery, radiation, and chemotherapy. This study uncovers a surprising culprit deep inside cancer cells: tiny chemical marks on transfer RNA, the molecules that help build proteins. The authors show that when a particular enzyme, DNMT2, is lost or reduced, thyroid tumors grow faster, spread more easily, and resist the common chemotherapy drug doxorubicin. Even more intriguing, they identify a small RNA fragment that emerges when DNMT2 is low and demonstrate that blocking this fragment could become a new treatment strategy.
A protective enzyme that goes missing
Transfer RNAs (tRNAs) normally carry amino acids to the cell’s protein-making machinery, and their stability depends on delicate chemical decorations. DNMT2 is an enzyme that adds one such mark, called a methyl group, to specific spots on tRNA. In many cancers DNMT2 is elevated and thought to help tumors, but here the story is reversed. By analyzing large patient datasets and tumor samples, the researchers found that DNMT2 levels are significantly lower in anaplastic thyroid cancer tissue than in normal thyroid. Patients whose tumors had more DNMT2 tended to live longer without their cancer worsening, suggesting that DNMT2 acts more like a brake than an accelerator in this disease.
How low DNMT2 fuels aggressive behavior
To see what DNMT2 actually does in cancer cells, the team manipulated its levels in anaplastic thyroid cancer cell lines grown in dishes and in mouse models. When DNMT2 was reduced, cells multiplied faster, invaded through membranes more easily, and formed more colonies, all hallmarks of an aggressive tumor. These cells also became less sensitive to doxorubicin, a standard drug for this cancer. In mice, tumors engineered to have less DNMT2 grew larger and spread to the lungs more readily, while boosting DNMT2 expression had the opposite effect. The researchers traced these changes in part to activation of a cellular program known as epithelial–mesenchymal transition, which makes cancer cells more mobile and invasive.
From damaged tRNA to harmful small fragments
Digging deeper, the study shows that DNMT2 places a methyl mark at a specific position (called C38) on just three types of tRNA. Without this protection, the tRNAs become more vulnerable to cutting by another enzyme, angiogenin. That cut produces a short piece of RNA derived from tRNA-Gly-GCC, termed 5’tiRNAGly-GCC. Using sequencing and biochemical tests, the authors found that when DNMT2 is low, this fragment builds up, especially from the glycine-carrying tRNA. In test-tube experiments, adding the methyl mark strongly shielded tRNA from being sliced, confirming that DNMT2 normally guards these molecules from being chopped into smaller, potentially harmful pieces.
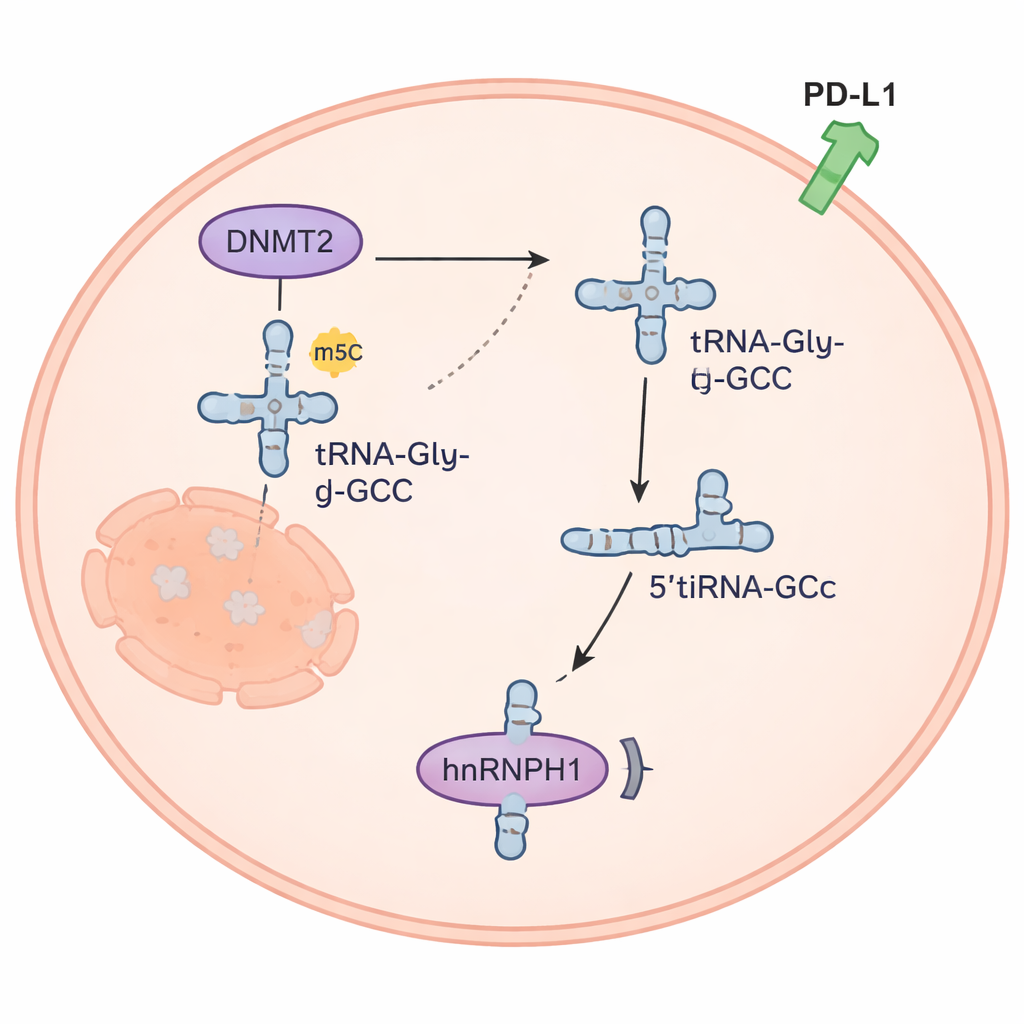
A tiny RNA fragment rewires cancer signals
The 5’tiRNAGly-GCC fragment turned out to be more than molecular debris. It was abundant in anaplastic thyroid cancer cells but scarce in normal thyroid cells. When the researchers artificially raised its levels, cancer cells proliferated faster, invaded more, and became more resistant to doxorubicin; lowering the fragment had the reverse effect. In mice, a chemically designed inhibitor of 5’tiRNAGly-GCC slowed tumor growth, and combining this inhibitor with doxorubicin worked even better than either treatment alone. The team discovered that 5’tiRNAGly-GCC binds to a protein called hnRNPH1, reducing its amount inside cells. Because hnRNPH1 normally helps keep levels of the immune-related protein PD-L1 in check, losing hnRNPH1 allows PD-L1 to rise, a change that can help tumors escape immune attack and is linked to regulatory T cell buildup around tumors.
What this means for future treatments
In simple terms, the study reveals a chain reaction: when DNMT2 is low, certain tRNAs lose a protective mark, are cut into small fragments, and one of these fragments (5’tiRNAGly-GCC) disables a protein that restrains PD-L1. The result is a more aggressive, drug-resistant, and immune-evasive cancer. Rather than trying to boost DNMT2 directly—which might be risky in other tissues—the authors suggest targeting the harmful RNA fragment itself. Their mouse experiments with a 5’tiRNAGly-GCC inhibitor, especially when combined with doxorubicin, provide early proof that interrupting this microscopic RNA circuitry could help tame one of the most lethal forms of thyroid cancer.
Citation: Zhou, R., Li, B., Cao, M. et al. DNMT2 inhibits anaplastic thyroid cancer progression by downregulating 5’tiRNAGly-GCC production. Cell Death Dis 17, 240 (2026). https://doi.org/10.1038/s41419-026-08488-5
Keywords: anaplastic thyroid cancer, DNMT2, tRNA fragments, chemoresistance, PD-L1