Clear Sky Science · en
Unveiling ZNF124 as a novel determinant in neurodegeneration: orchestration of photoreceptor homeostasis through MSX2 transcriptional regulation
Why this discovery matters for sight
Retinitis pigmentosa is a leading cause of inherited blindness, yet in nearly 40 percent of patients doctors still cannot pinpoint the faulty gene. This study uncovers a new genetic player, called ZNF124, that helps keep light-sensing cells in the eye alive. By showing how a mutation in this gene disrupts a chain of molecular “switches” in the retina, the researchers open new paths toward diagnosis and, potentially, targeted treatments for people losing their vision.
A hidden cause of inherited vision loss
The researchers studied a large family in which several children developed classic signs of retinitis pigmentosa: night blindness early in life, shrinking side vision, and gradual loss of central sight. Detailed eye exams revealed thinning of the retina and weak electrical responses to light, both hallmarks of damage to rod and cone photoreceptors. Yet none of the more than 80 known retinitis pigmentosa genes carried obvious defects in these patients, hinting that an undiscovered gene might be responsible for their disease.
Finding a faulty genetic switch
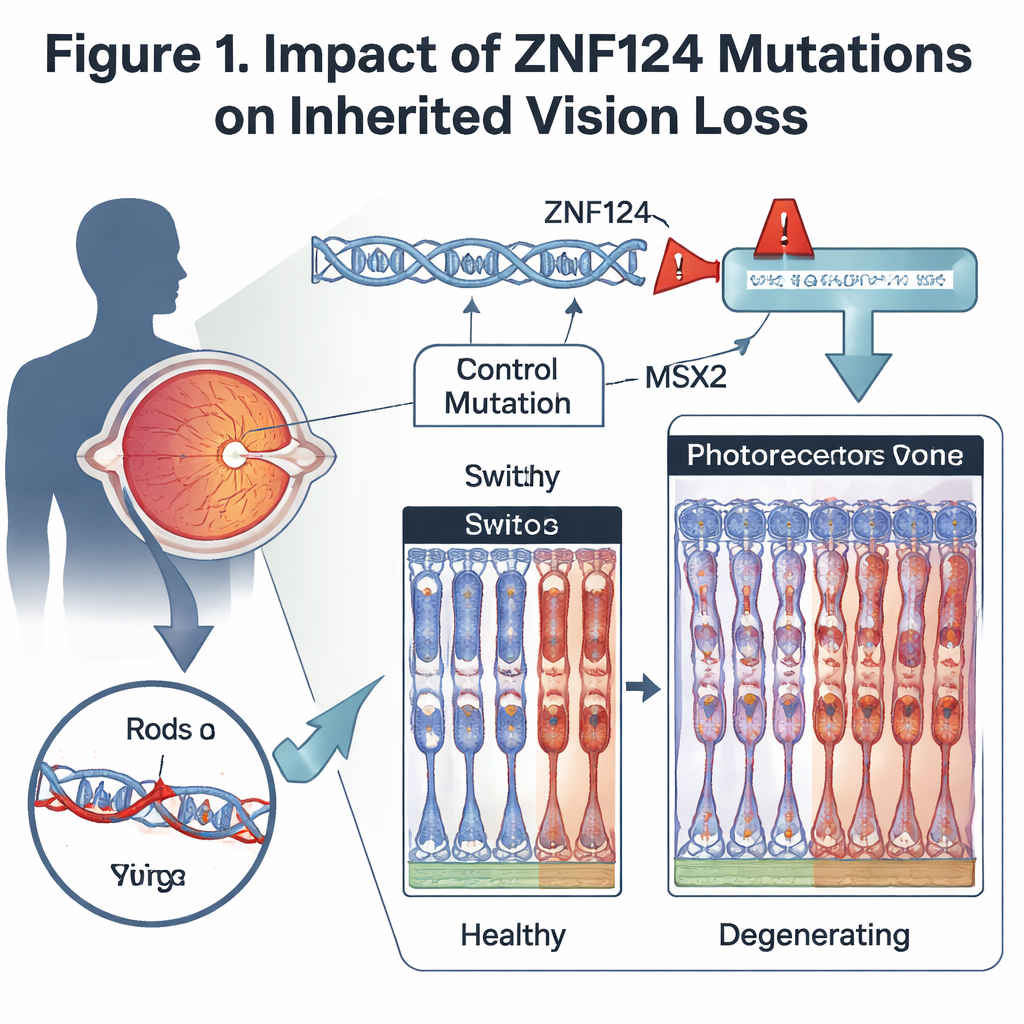
Using whole-exome sequencing, which reads the protein-coding parts of DNA, the team identified a rare, previously unreported mutation in the ZNF124 gene. This mutation alters the way the gene’s RNA is stitched together, deleting a few bases at a critical junction. As a result, the ZNF124 protein is cut short and loses its zinc-finger region, a structure normally used to recognize and bind specific DNA sequences. Because zinc-finger proteins often act as master switches that turn many other genes on or off, a damaged ZNF124 could have wide-ranging effects in retinal cells.
Testing the gene in animal models
To see how loss of this switch affects the eye, the scientists created mice lacking Gm20541, the closest mouse equivalent of ZNF124, specifically in the retina. These animals developed age-dependent vision problems: their electrical responses to dim and bright light weakened, and microscopic examination showed progressive thinning of the retinal layer that contains photoreceptors. Both rods, which support night vision, and cones, which support color and daytime vision, showed shortened outer segments and loss of key visual proteins. Other retinal cells, such as certain bipolar cells, were also reduced, and support cells became activated, a common response to chronic retinal injury.
Uncovering the ZNF124–MSX2 control pathway
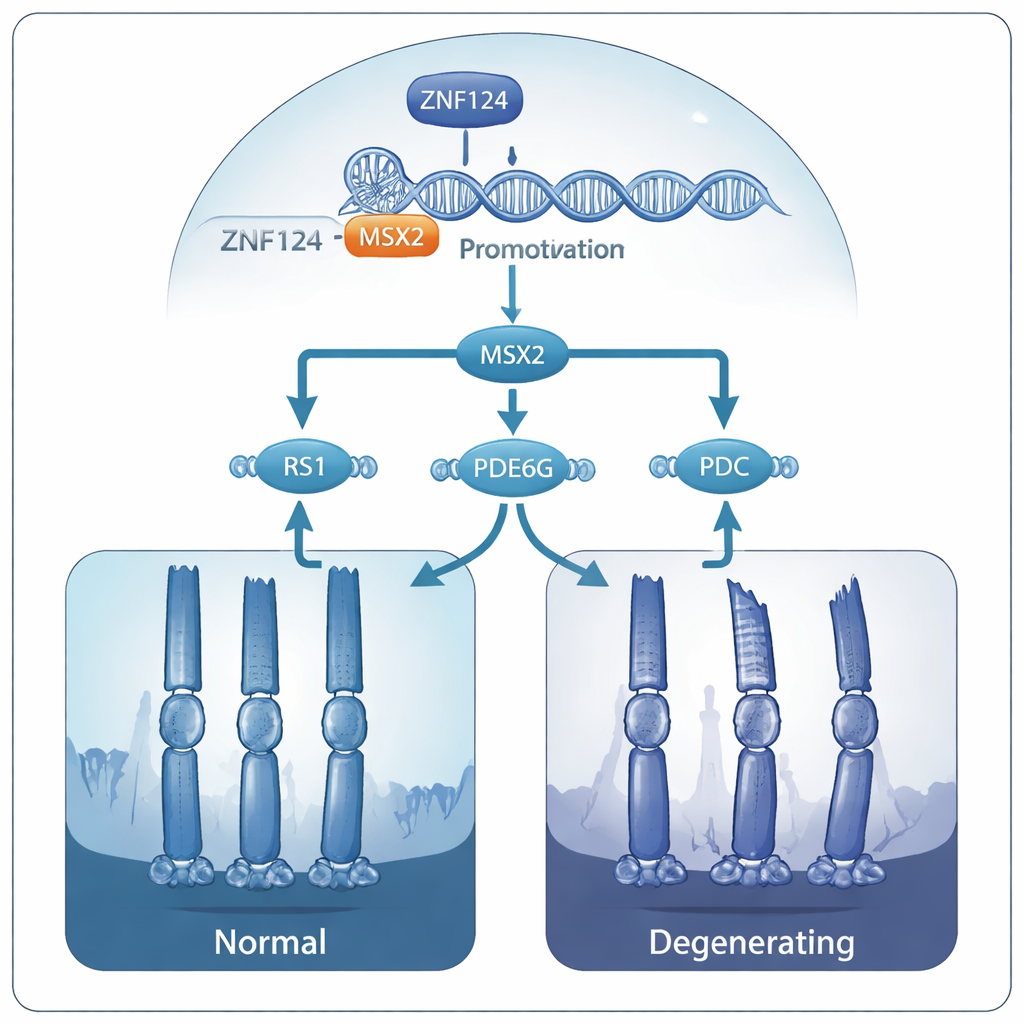
The next question was which genes ZNF124 normally controls. Using biochemical methods that map where proteins sit on DNA, combined with global RNA readouts from mouse retinas, the team found that ZNF124 binds to and activates another gene called MSX2. In healthy cells, ZNF124 attaches to a specific short sequence in the MSX2 “on-switch” region and boosts its activity. In mice missing Gm20541, MSX2 levels dropped by more than half. When the researchers deleted MSX2 specifically in rod cells, those animals also developed thinning of the photoreceptor layer and shortened outer segments, mirroring the defects seen in the ZNF124-like knockout mice. This placed MSX2 directly downstream of ZNF124 in a pathway essential for photoreceptor survival.
From gene switches to fragile photoreceptors
Further analysis showed that MSX2 in turn helps maintain several other genes already known to be linked to inherited retinal disease: RS1, PDE6G, and PDC. These genes support the structure of the retina and the chemistry of visual signaling. When MSX2 was reduced, all three genes became less active, and their protein products declined. The authors propose that in people with damaging ZNF124 mutations, this whole cascade is weakened: ZNF124 can no longer fully activate MSX2, MSX2 fails to sustain RS1, PDE6G, and PDC, and over time the photoreceptors lose their structural integrity and die, leading to progressive vision loss.
What this means for patients and therapies
To a non-specialist, the central message is that the retina depends on a finely tuned hierarchy of genetic switches. This work identifies ZNF124 as a new top-level switch whose failure can cause inherited blindness through a specific downstream partner, MSX2, and its target genes. Clinically, ZNF124 can now be added to genetic testing panels, helping more families receive precise diagnoses. In the longer term, therapies that restore activity of ZNF124, MSX2, or the affected downstream genes may help stabilize or rescue light-sensing cells, offering new hope for people with currently unexplained forms of retinitis pigmentosa.
Citation: Yang, Y., Jiang, X., Li, S. et al. Unveiling ZNF124 as a novel determinant in neurodegeneration: orchestration of photoreceptor homeostasis through MSX2 transcriptional regulation. Cell Death Dis 17, 234 (2026). https://doi.org/10.1038/s41419-026-08487-6
Keywords: retinitis pigmentosa, photoreceptors, ZNF124, MSX2, inherited retinal disease