Clear Sky Science · en
TIGAR maintains intestinal epithelial regeneration by stabilizing HMGCL and promoting β-catenin β-hydroxybutyrylation in burn-induced sepsis
Why gut repair matters after severe burns
Severe burns don’t just damage skin; they can also wreak havoc deep inside the body, especially in the intestines. When the gut’s inner lining breaks down, bacteria and toxins can leak into the bloodstream, driving dangerous infections and organ failure. This study explores how a natural metabolic “switch” in intestinal cells helps the gut repair itself after burn-induced sepsis, and how disrupting this switch can turn a manageable injury into a life‑threatening crisis.
A hidden repair crew in the gut lining
The inside of the small intestine is covered with tiny finger-like projections called villi, which constantly renew themselves. This renewal depends on intestinal stem cells tucked in pockets at the base of the villi. After major burns and sepsis, the researchers found that the structure of the mouse intestine became badly damaged: villi shrank, regenerative pockets were lost, and cell growth markers dropped. At the same time, levels of a protein called TIGAR, usually enriched in these regenerative regions, fell sharply. Mice with lower TIGAR in their intestinal cells showed worse damage and poorer stem cell activity, suggesting that TIGAR acts as an internal protector of gut regeneration in severe illness. 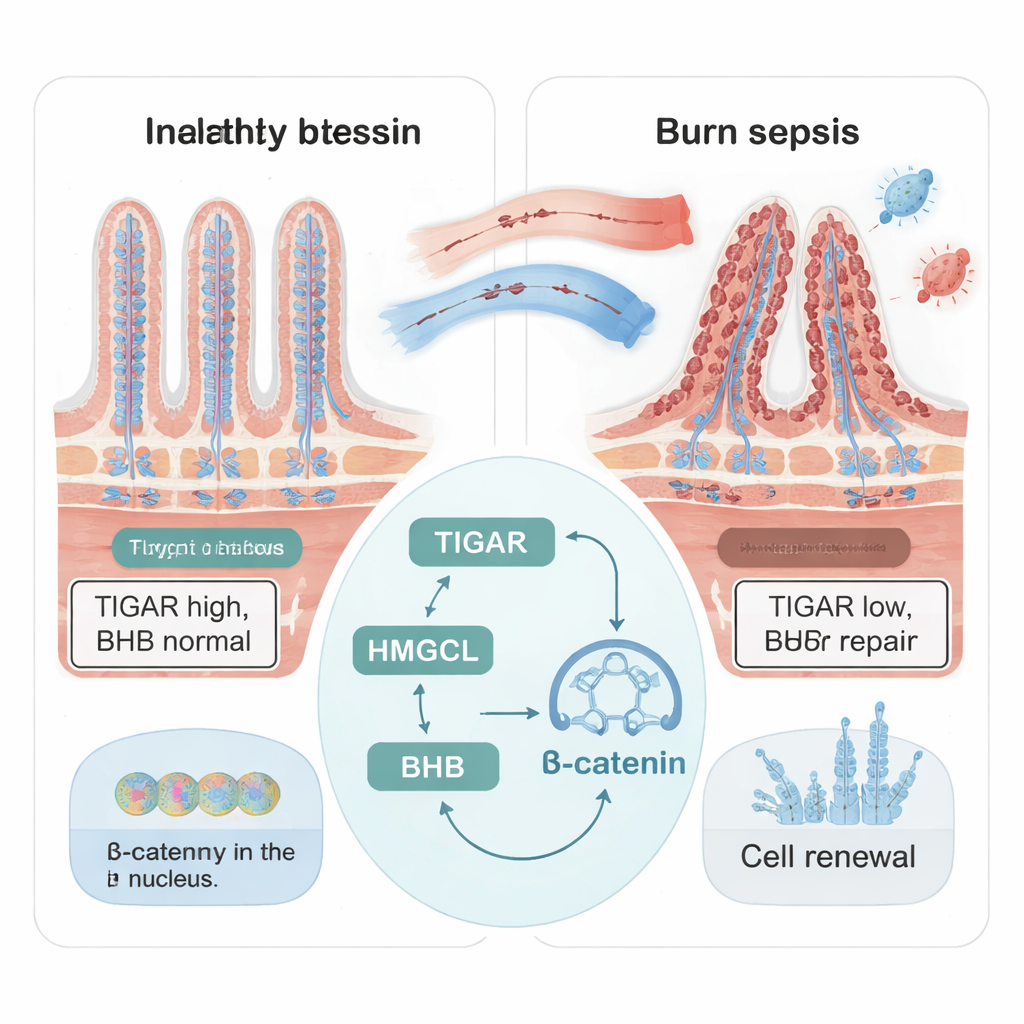
Fueling repair with a special fat-derived molecule
Intestinal cells can use not only sugar and amino acids for energy, but also ketone bodies—fat-derived molecules produced mainly in the liver and, to a lesser extent, in the gut. The team focused on beta‑hydroxybutyrate (BHB), the most abundant ketone body, known to support stem cells and tissue renewal. In burn‑sepsis mice, intestinal BHB levels first spiked and then collapsed to about one‑third of normal by day seven, matching the period of worst injury. When TIGAR was reduced in intestinal cells, BHB levels dropped as well. Adding extra BHB in cell and mini‑gut (organoid) models rescued cell growth, restored stem cell markers, and countered the damaging effects of inflammatory stress, showing that BHB is a key fuel and signal for gut repair.
How TIGAR keeps the ketone engine running
Digging deeper, the researchers discovered how TIGAR keeps BHB production going. Inside intestinal cells, the enzyme HMGCL is a crucial step in making ketone bodies. TIGAR binds directly to HMGCL inside mitochondria, the cell’s energy factories. This binding shields HMGCL from another protein, Park2, which normally tags HMGCL with ubiquitin chains that send it to the cell’s “shredder” system. By blocking this tagging, TIGAR prevents HMGCL from being broken down, keeping ketone production active. When TIGAR was removed or cells were stressed with bacterial toxins, HMGCL became unstable, BHB production dropped, and cell proliferation slowed; restoring HMGCL or adding BHB reversed many of these defects.
A molecular nudge that sends growth signals to the nucleus
BHB does more than supply energy; it also modifies proteins. The study shows that BHB attaches a small chemical tag, called β‑hydroxybutyrylation, to a specific site (lysine 335) on β‑catenin, a master switch that drives genes for cell growth and stem cell renewal. With enough BHB, β‑catenin carrying this tag moves into the cell nucleus and partners with another factor, TCF4, to turn on pro‑growth genes. When BHB is low, or when this specific site on β‑catenin is mutated, β‑catenin struggles to enter the nucleus and the growth program stalls. TIGAR, by maintaining HMGCL and BHB, therefore indirectly controls whether β‑catenin can reach the nucleus and keep the gut lining renewing. 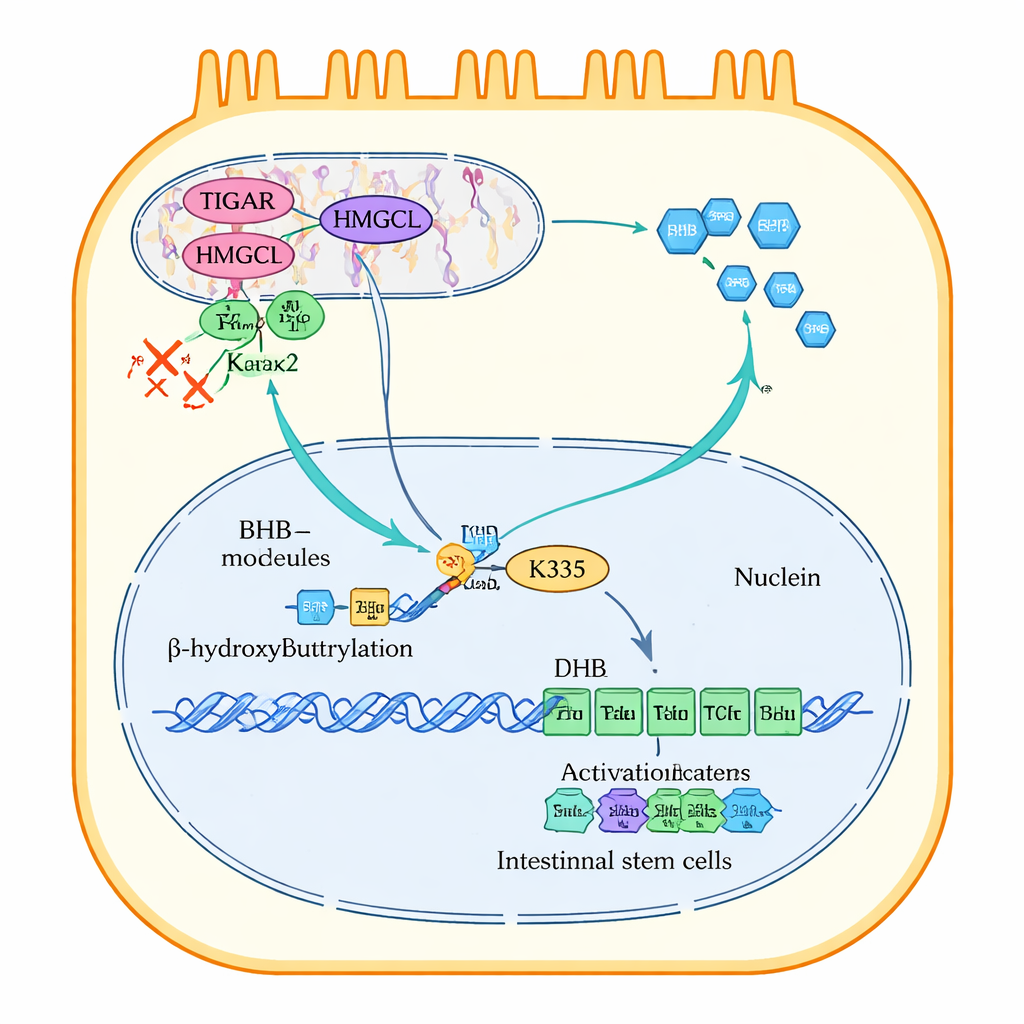
What this means for burn patients
In simple terms, this work reveals a chain reaction: TIGAR protects a ketone‑producing enzyme (HMGCL), which keeps BHB levels up; BHB then fine‑tunes a growth switch (β‑catenin) that tells intestinal stem cells to divide and rebuild the gut barrier. When TIGAR levels fall after severe burns and sepsis, this protective chain breaks, gut repair falters, and barrier failure worsens infection. Targeting this TIGAR–HMGCL–BHB–β‑catenin route—by boosting TIGAR function, stabilizing HMGCL, or safely supplementing BHB—could offer new ways to preserve gut integrity and improve survival in patients with severe burns and other critical illnesses that threaten the intestinal barrier.
Citation: Zhang, P., Wu, D., Wei, Y. et al. TIGAR maintains intestinal epithelial regeneration by stabilizing HMGCL and promoting β-catenin β-hydroxybutyrylation in burn-induced sepsis. Cell Death Dis 17, 233 (2026). https://doi.org/10.1038/s41419-026-08486-7
Keywords: intestinal barrier repair, ketone bodies, TIGAR, burn sepsis, intestinal stem cells