Clear Sky Science · en
HIF2α-induced lysyl oxidase safeguards successful pregnancy by remodelling collagens at the feto-maternal interface
Why the Uterus Must Prepare for a New Arrival
Before a pregnancy can take hold, an early embryo must do something remarkable: burrow into the wall of the uterus and build a life-support system, the placenta. This paper explores how the mother’s tissue quietly reshapes itself to welcome the embryo, focusing on how low oxygen levels and a little-known enzyme help prepare the uterine lining. Understanding this finely tuned dance may shed light on why some pregnancies fail very early and suggest new ways to prevent infertility and pregnancy complications.
A Delicate Meeting Between Mother and Embryo
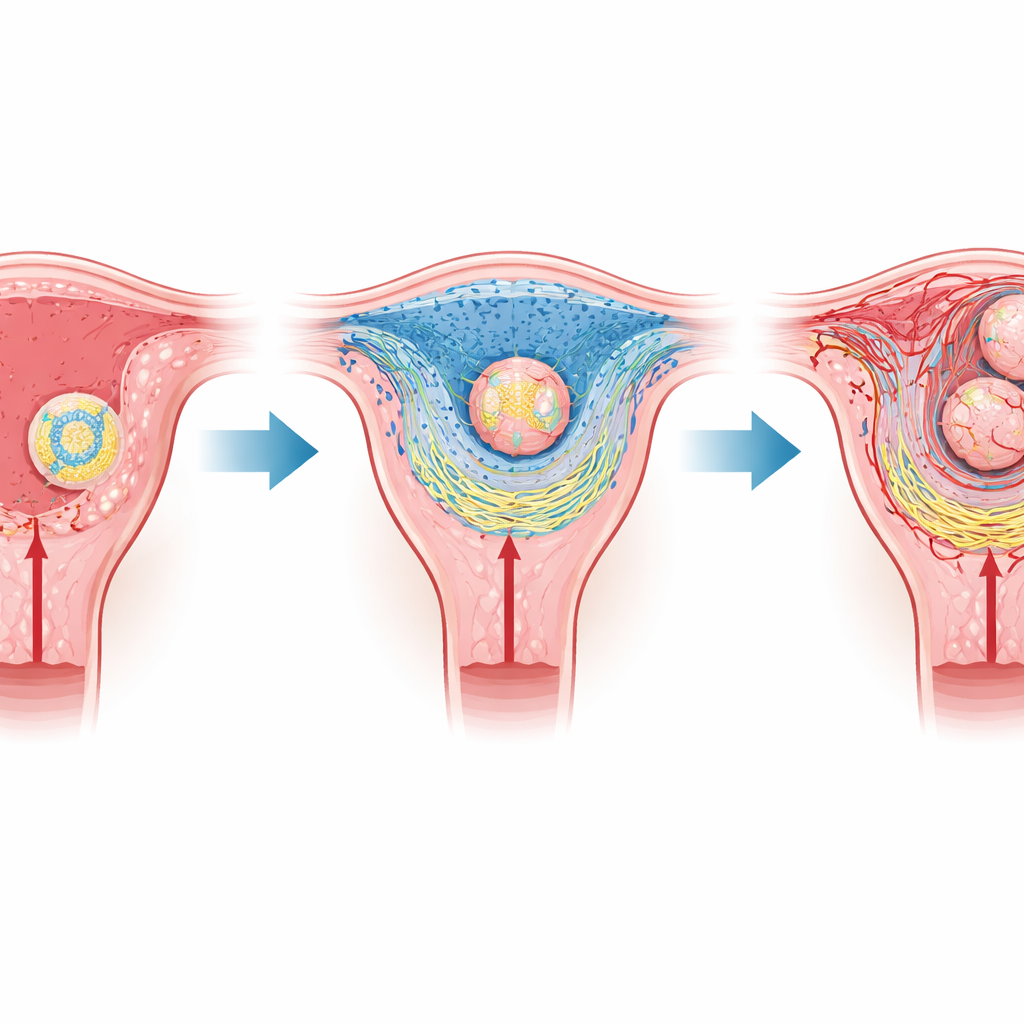
In mammals such as mice and humans, the earliest stage of pregnancy hinges on implantation, when the embryo attaches to and then invades the uterine lining. The embryo is wrapped in a shell of specialized cells that will later form the placenta, while the mother’s tissue transforms into a cushion-like layer called the decidua. Closest to the embryo lies a thin region known as the primary decidual zone, which lacks blood vessels and therefore contains little oxygen. Earlier work showed that this naturally low-oxygen pocket helps trigger implantation, but how such a simple physical condition could reorganize whole tissues was not well understood.
Low Oxygen as a Hidden Switch
The authors used a powerful technique called spatial transcriptomics, which maps gene activity directly onto tissue slices, to study mouse uteri during the days when embryos attach and invade. They focused on a protein called HIF2α, which turns on in low-oxygen conditions and acts as a genetic switch. In normal mice, cells in the primary decidual zone showed strong activity of genes linked to low oxygen and to the construction of the surrounding scaffolding material, including collagens that form the extracellular matrix. When HIF2α was removed only from the uterus, the overall arrangement of cell types looked surprisingly normal. But at high resolution, the collagen network was disorganized, the epithelial surface did not open up properly, and embryonic cells struggled to penetrate the maternal tissue.
An Enzyme That Crosslinks the Scaffolding
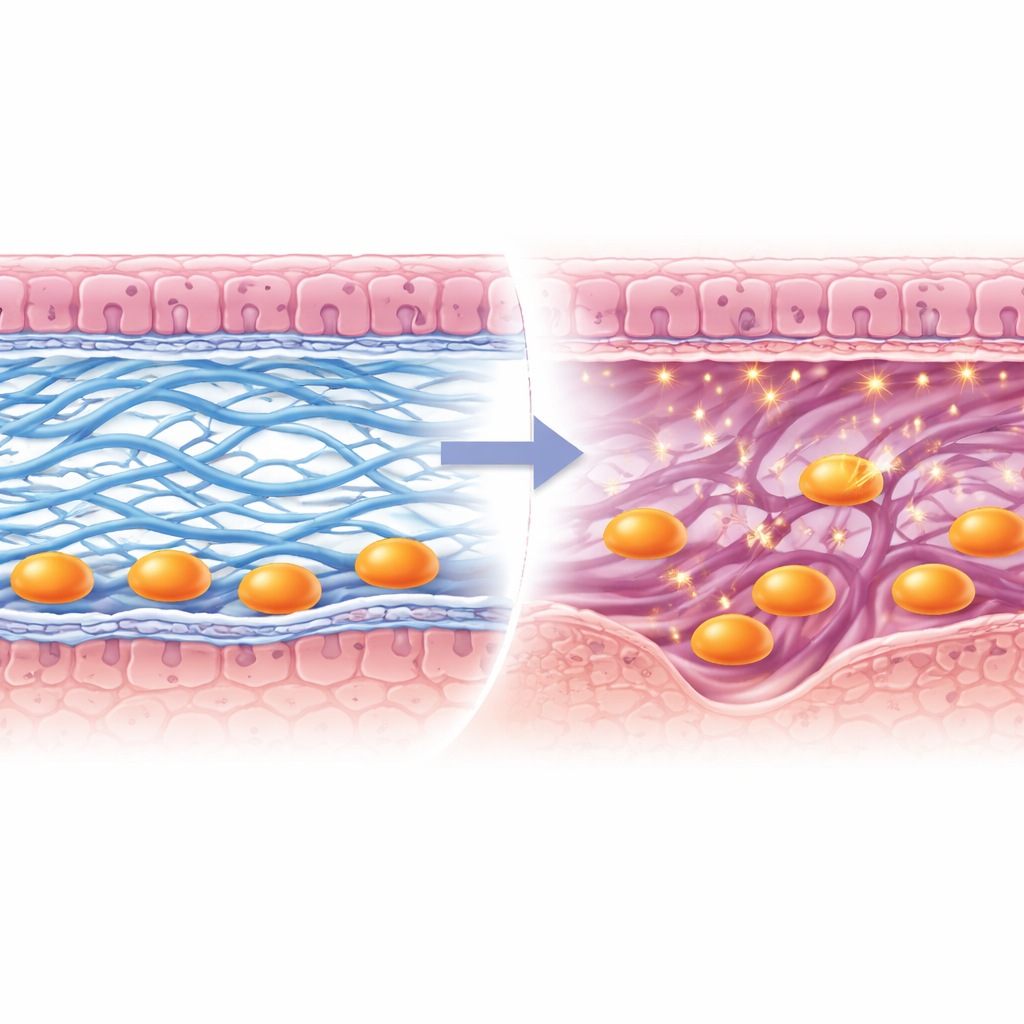
Digging deeper, the team identified a key player downstream of HIF2α: lysyl oxidase, or Lox, an enzyme that chemically crosslinks collagen fibers and helps stiffen and stabilize tissue. In normal pregnancies, Lox levels rose sharply around embryos precisely when they began to invade. In uteri lacking HIF2α, Lox expression dropped, and collagen fibers appeared fragmented instead of forming a dense mesh. To test Lox’s importance directly, the researchers engineered mice in which Lox was deleted only in the uterus. These females became pregnant less often, carried fewer embryos, and frequently showed signs of embryo loss and bleeding later in gestation, even though the earliest steps of attachment and tissue thickening initially looked normal.
Opening a Path for the Placenta
Careful imaging revealed what went wrong when Lox was missing. A thin protein-rich sheet called the basement membrane, which normally breaks down to let embryonic cells through, remained largely intact. Collagen type I fibers in the underlying stroma failed to form robust, continuous bundles, while collagen type IV in the basement membrane did not clear properly near invading cells. As a result, trophoblast cells—the embryo-derived pioneers that build the placenta—remained trapped at the surface instead of tunneling into the maternal layer. The team also found that a collagen-cutting enzyme, Mmp9, was not switched on in these trophoblasts, further hampering their progress. Over time, placental structures formed poorly, embryos grew abnormally, and pregnancy success dropped.
What This Means for Pregnancy Health
Put together, the findings outline a chain of events in which low oxygen in the early implantation zone activates HIF2α, which in turn boosts Lox. Lox then reshapes and crosslinks collagen fibers and helps dismantle the basement membrane, while also enabling enzymes like Mmp9 to clear a path. This remodeling provides both a structural scaffold and an open doorway for trophoblast cells to invade and construct the placenta. For a lay reader, the message is that successful pregnancy depends not only on healthy embryos and hormones but also on the quiet reworking of the mother’s tissue architecture. Disruptions in this hypoxia–HIF2α–Lox pathway could underlie some cases of unexplained infertility, miscarriage, and disorders linked to poor placental invasion, suggesting new diagnostic markers and therapeutic targets for supporting early pregnancy.
Citation: Aikawa, S., Shimizu-Hirota, R., Sakashita, A. et al. HIF2α-induced lysyl oxidase safeguards successful pregnancy by remodelling collagens at the feto-maternal interface. Cell Death Dis 17, 250 (2026). https://doi.org/10.1038/s41419-026-08485-8
Keywords: embryo implantation, uterine extracellular matrix, lysyl oxidase, trophoblast invasion, placental development