Clear Sky Science · en
UBE3A-mediated mH2A1 Ubiquitination activates TERT transcription to promote senescence resistance in pancreatic cancer
Why this research matters for patients
Pancreatic cancer is among the deadliest cancers, in part because tumor cells are unusually good at dodging the body’s built‑in aging brakes. This study uncovers a hidden molecular “sabotage circuit” that helps pancreatic cancer cells stay young, keep dividing, and resist treatment. By understanding this circuit, the researchers point to a new way to push these cells into aging and then selectively clear them out, opening the door to smarter, combination therapies.
Aging as the body’s natural tumor brake
All cells carry an internal clock that eventually makes them stop dividing, a process known as senescence. In early pancreatic lesions, this clock can slow tumor development by putting precancerous cells into permanent retirement. Many anticancer treatments also work partly by forcing tumor cells into this aged state. However, if these senescent cells are not cleared, they can fuel inflammation and help the cancer come back. That has led to a new treatment idea: first push cancer cells into senescence, then eliminate those aged cells with “senolytic” drugs that are specially toxic to them.
A cancer-promoting enzyme in the spotlight
To find genes that help pancreatic cancer cells defy senescence, the team mined large patient datasets and then validated their findings in tumor samples and cell lines. They homed in on a protein called UBE3A, an enzyme that tags other proteins for destruction. UBE3A levels were much higher in pancreatic tumors than in nearby normal tissue, and patients with more UBE3A tended to have larger, more advanced cancers and poorer survival. In lab-grown cells, boosting UBE3A sped up growth, reduced classic markers of cellular aging, and cut the inflammatory secretions typical of senescent cells. Silencing UBE3A had the opposite effect and, in mouse models, sharply reduced tumor growth and liver metastasis. 
How cancer cells erase a chromatin brake
Diving deeper, the researchers asked which molecules UBE3A acts on. Using advanced mass‑spectrometry screens, they found that UBE3A binds to and chemically tags a special histone variant called macroH2A1 (mH2A1), which helps compact DNA and usually acts as a brake on gene activity. UBE3A attaches a chain of “destroy me” tags to mH2A1 at a specific position, causing it to be shredded by the cell’s protein disposal machinery. In pancreatic tumors, mH2A1 levels were lower where UBE3A was high, and restoring mH2A1 countered UBE3A’s ability to block senescence and drive tumor growth. This reveals a key step: pancreatic cancer cells strip away a protective chromatin mark by using UBE3A to eliminate mH2A1.
Unlocking telomerase to keep cells young
The next question was which genes are freed when mH2A1 is removed. By combining RNA sequencing and DNA‑binding maps, the team identified the telomerase gene TERT as a central target. Telomerase maintains the protective caps on chromosome ends, allowing cells to keep dividing instead of entering senescence. The study shows that mH2A1 normally sits at an enhancer region within the TERT gene, where it recruits another enzyme, EZH2, to lay down a repressive chemical mark on nearby histones. This combination keeps TERT turned down and lets telomeres shorten. When UBE3A destroys mH2A1, the repressive complex dissolves, the enhancer becomes active, TERT switches on, telomeres are maintained, and pancreatic cancer cells gain resistance to aging. Mutant forms of UBE3A that cannot tag proteins, or mutant mH2A1 that cannot be tagged, break this circuit and restore the senescence brake.
Pairing senescence induction with senolytic clean‑up
Finally, the researchers tested whether this pathway could be exploited therapeutically. Shutting down UBE3A pushed pancreatic cancer cells toward senescence and increased their reliance on anti‑death proteins of the BCL‑2 family—a known vulnerability of senescent cells. The team then combined UBE3A inhibition with ABT‑263, a senolytic drug that targets these survival proteins. In cultured cells, the combination caused many more cancer cells to undergo programmed cell death than either treatment alone. In mouse models, tumors with reduced UBE3A shrank even further when animals were given ABT‑263, and markers of cell proliferation inside the tumors dropped sharply. 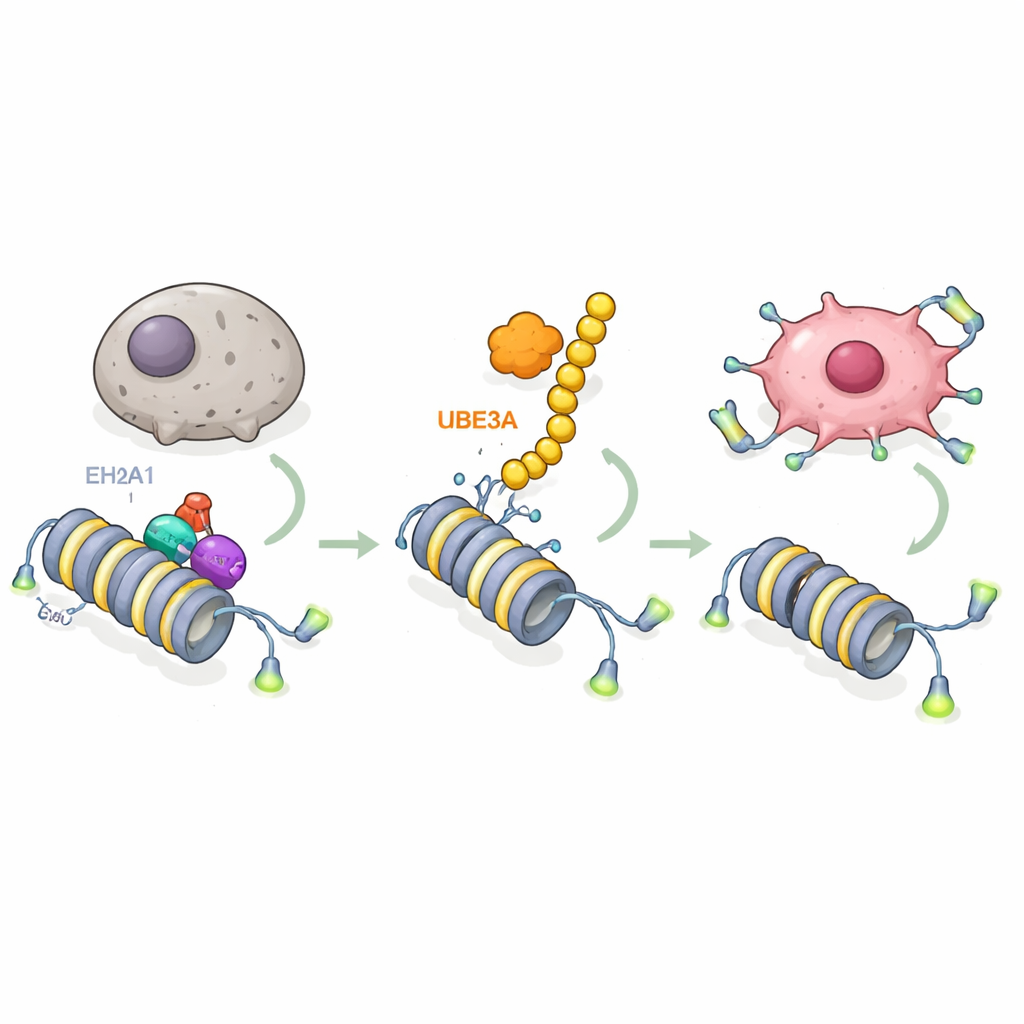
What this means for future treatment
This work reveals a detailed chain of events by which pancreatic cancer cells avoid growing old: UBE3A destroys the chromatin brake mH2A1, which in turn lifts repression of the telomerase gene TERT and helps maintain telomeres, allowing cells to keep dividing. By targeting UBE3A, doctors may be able to re‑engage the cell’s natural aging program and then use senolytic drugs like ABT‑263 to selectively eliminate these vulnerable, aged cancer cells. While more studies are needed to translate this mechanism into patient therapies, the UBE3A–mH2A1–TERT axis offers a promising new handle on one of the most treatment‑resistant cancers.
Citation: Ren, L., Lu, R., Fei, X. et al. UBE3A-mediated mH2A1 Ubiquitination activates TERT transcription to promote senescence resistance in pancreatic cancer. Cell Death Dis 17, 274 (2026). https://doi.org/10.1038/s41419-026-08480-z
Keywords: pancreatic cancer, cellular senescence, telomerase, ubiquitin ligase, senolytic therapy