Clear Sky Science · en
A small molecule inhibitor of ARF GTPase protein 1 limits liver and colon cancer cell growth and metastasis
Why this research matters
Liver and colon cancers are among the deadliest tumors, and current treatments often shrink tumors at the cost of harsh side effects. This study explores a new kind of drug that does not simply poison fast-growing cells, but instead slips inside cancer cells and pries apart a protein “control hub” they rely on to grow and spread. By homing in on this hub, the experimental compound aims to slow tumor growth while sparing healthy tissue.
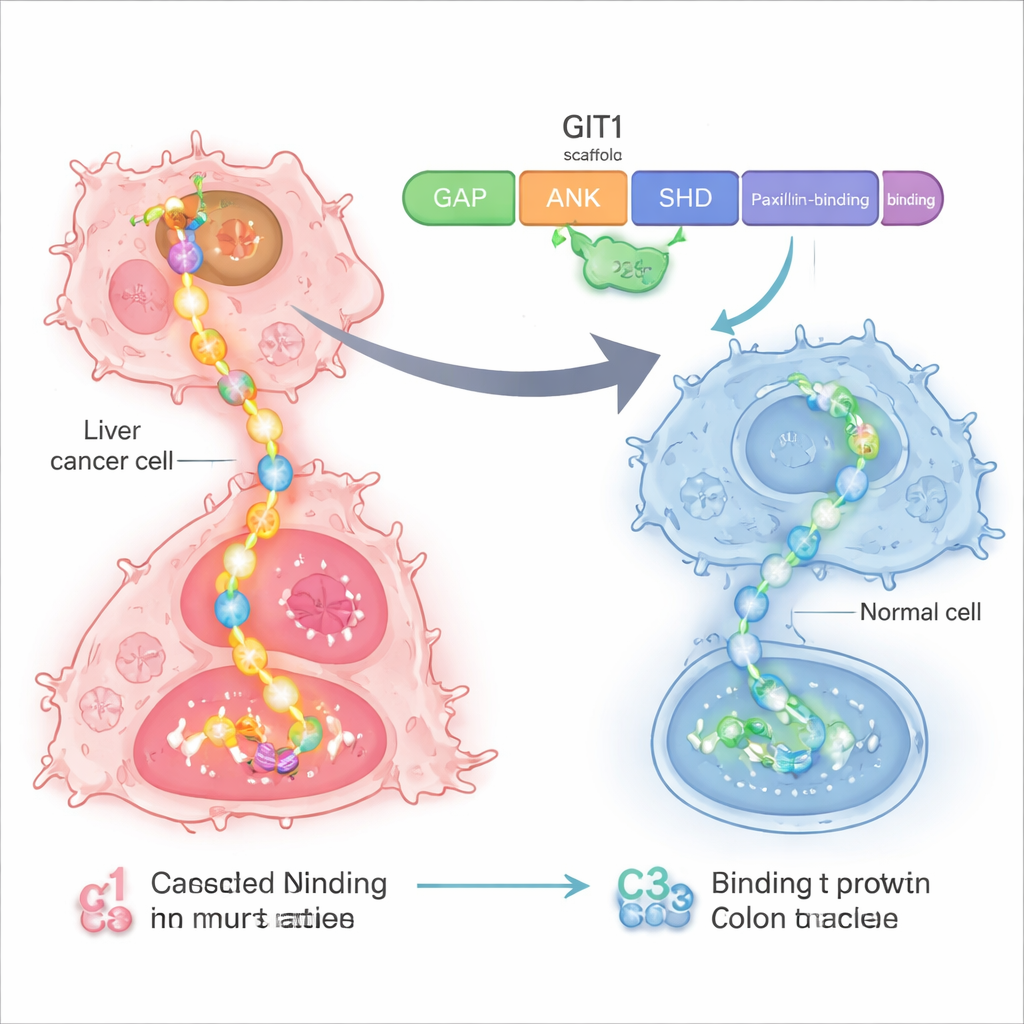
A vulnerable control hub inside cancer cells
The researchers focused on a protein called GIT1, which acts like a scaffold or power strip inside cells. In liver and colon cancers, GIT1 and its partner protein MAT2B are overproduced. Together, they assemble several pieces of a major growth pathway known as RAS–RAF–MEK–ERK, a chain of signals that tells cells to divide and migrate. Earlier work showed that when GIT1 and MAT2B are abundant, liver and colon tumors grow faster and spread more easily, and when these proteins are reduced, cancer growth slows. This made the GIT1–MAT2B scaffold an attractive target for a more precise therapy.
Designing a small molecule without a blueprint
A key challenge was that the three-dimensional crystal structure of GIT1 is not available, so the team could not simply “look up” the protein’s shape. Instead, they used computer modeling to predict the structure of one region of GIT1 made up of ankyrin repeats, located near where MAT2B and other signaling proteins attach. They then virtually screened a large library of small molecules against this model to see which might bind to that area. Out of nine candidates tested in cells, one molecule—called compound 3 (C3)—stood out. C3 bound specifically to GIT1, but not to its close relative GIT2, and reduced the activity of the ERK growth signal in multiple cancer cell lines.
Stopping cancer cells from dividing and spreading
When liver and colon cancer cells were treated with C3, their growth slowed, and at higher doses many cells died, while noncancerous liver and kidney cells remained largely unaffected. C3 caused cancer cells to stall at the boundary between the G2 and M phases of the cell cycle, the checkpoint just before and during mitosis when a cell divides into two. The compound also sharply reduced the ability of cancer cells to form colonies and to migrate across a dish—two laboratory measures linked to tumor regrowth and metastasis. At the molecular level, C3 weakened the interactions between GIT1, MAT2B, and the RAF–MEK–ERK signaling proteins, leading to lower MEK and ERK activity and less of the cell-cycle driver cyclin D1.
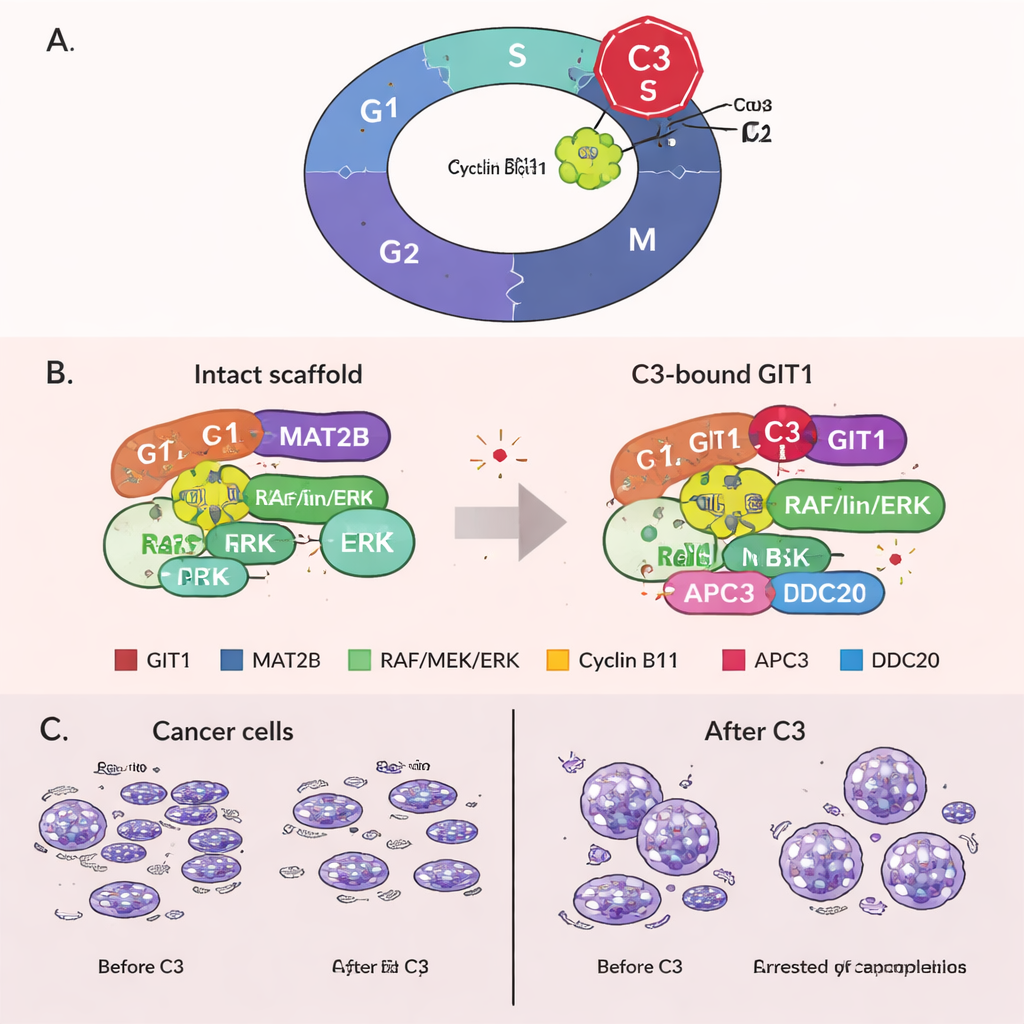
Locking cells in mitosis through a newly discovered role of GIT1
Unexpectedly, the team uncovered a new role for GIT1 in the final steps of cell division. They found that GIT1 binds to cyclin B1, a protein that helps push cells into mitosis, and also to parts of a protein machine called the anaphase-promoting complex (APC/C), which normally tags cyclin B1 for destruction so cells can exit mitosis. C3 treatment strengthened the link between GIT1 and cyclin B1 but weakened the connections among GIT1, cyclin B1, and APC/C components. As a result, cyclin B1 was no longer efficiently broken down, its levels stayed high, and cells became stuck in mitosis. This extended arrest is known to trigger cell death, offering a second way in which C3 can kill cancer cells. Importantly, when GIT1 was experimentally reduced, C3 lost much of its ability to raise cyclin B1, block the cell cycle, and suppress growth, showing that its effects are truly GIT1-dependent.
Promising results in animal models
The researchers then moved to mouse models to see if C3 could work in living organisms. In immune-competent mice with implanted colorectal tumors, injections of C3 directly into the tumors sharply slowed their growth without obvious toxicity to major organs. In two additional models—human colon cancer cells growing in the livers of immune-deficient mice, and mouse colon cancer cells that had spread to the liver in immune-competent mice—C3 given by intraperitoneal injection reduced tumor burden and signs of spread. Blood tests and tissue exams suggested the treatments were well tolerated, and pharmacokinetic studies showed that drug levels in the bloodstream remained high enough for several hours after dosing.
What this could mean for future cancer treatments
To a non-specialist, the key message is that the investigators have found a way to attack cancer not by targeting a single mutated enzyme, but by disrupting a multi-protein assembly that cancer cells depend on for both growth signals and proper cell division. Their small molecule, C3, binds specifically to GIT1, reshapes its network of partners, dials down a major growth pathway, and traps cancer cells in a fatal traffic jam during mitosis. While C3 is still an experimental tool and not yet a drug for patients, the work demonstrates that targeting scaffold proteins like GIT1 may open a new avenue for treating liver and colon cancers with greater precision and potentially fewer side effects.
Citation: Peng, H., Chhimwal, J., Fan, W. et al. A small molecule inhibitor of ARF GTPase protein 1 limits liver and colon cancer cell growth and metastasis. Cell Death Dis 17, 238 (2026). https://doi.org/10.1038/s41419-026-08477-8
Keywords: liver cancer, colon cancer, targeted therapy, cell cycle arrest, scaffold protein