Clear Sky Science · en
α-ketoglutarate/succinate ratio imbalance impairs thymine DNA glycosylase function and base excision repair process increasing susceptibility to pancreatic cancer
When Everyday Metabolism Meets Hidden DNA Damage
Pancreatic cancer is one of the deadliest cancers in part because it is often found too late. This study explores a subtle but powerful idea: how long-lasting problems like obesity, high blood sugar, and high-fat diets can quietly reshape the chemistry inside pancreatic cells, weaken their DNA repair systems, and make it easier for cancer to take hold. By following the trail of small molecules inside cells, the researchers uncover how a disturbed metabolic balance can tip DNA from being well maintained to dangerously fragile.
How Diet-Driven Metabolism Primes the Pancreas
To understand how an unhealthy metabolic state affects the pancreas, the researchers used mice genetically prone to developing pancreatic lesions and fed them either a low-fat or high-fat diet. The high-fat diet quickly triggered weight gain, elevated blood sugar and cholesterol, and changes in genes linked to growth and inflammation. At the tissue level, the pancreases of high-fat–fed mice showed earlier and more pronounced precancerous changes compared with controls. Metabolite profiling revealed that specific fats shifted in abundance and that molecules involved in one-carbon metabolism and methyl donation—chemical processes that influence how DNA is marked and read—were notably altered, especially the key methyl donor S-adenosyl methionine.
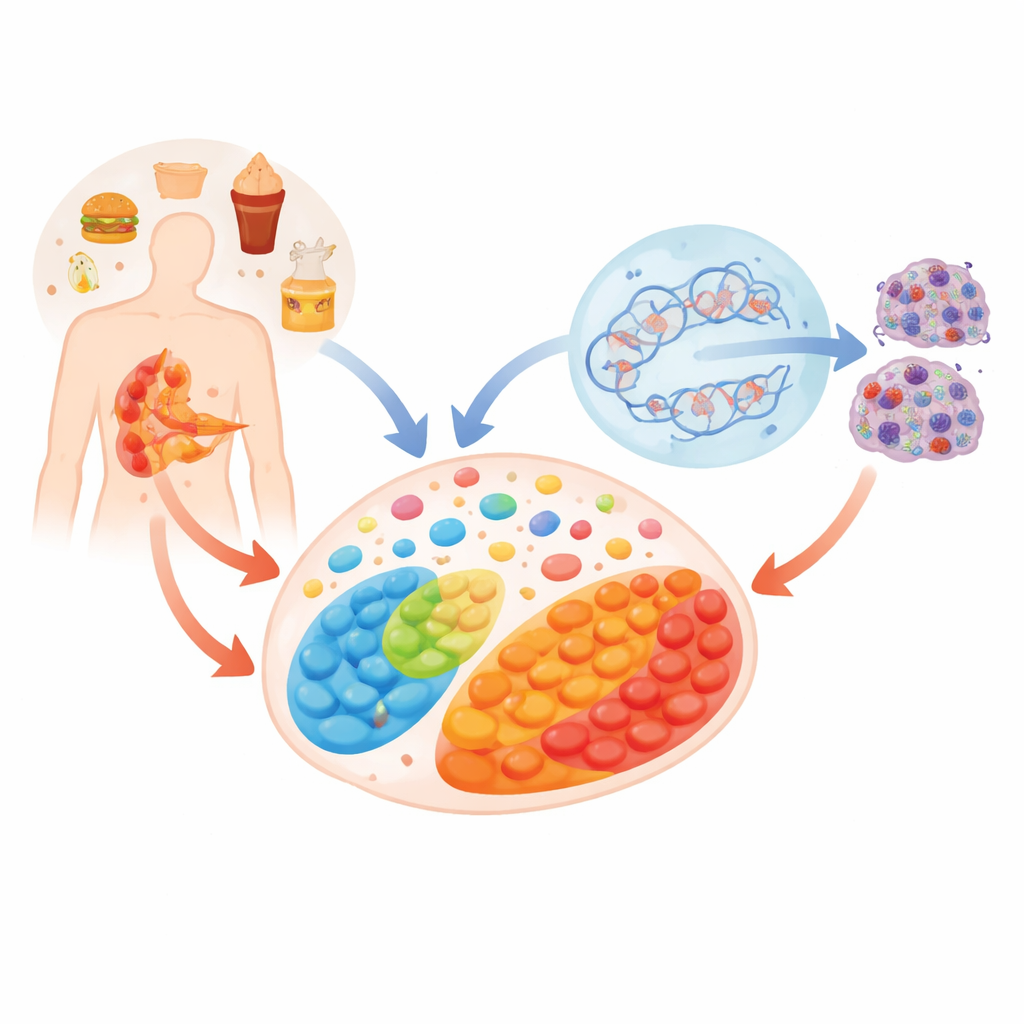
Small Molecules That Steer DNA Marks
Inside cells, the chemical “tags” on DNA are set and removed by enzymes that depend on particular metabolites as fuel or helpers. In this work, the balance between two molecules from the cell’s energy cycle, alpha-ketoglutarate and succinate, emerged as crucial. In the high-fat diet setting and in human pancreatic duct cells exposed to high glucose and a common dietary fat, alpha-ketoglutarate levels fell while succinate rose, skewing their ratio. This shift disturbed a DNA demethylation partnership between the enzyme TET1 and the repair protein TDG. Instead of smoothly cycling DNA marks on and off, pancreatic epithelial cells accumulated intermediate cytosine modifications and chemical scars on DNA known as abasic sites—gaps where a base is missing.
When Repair Helpers Become Overzealous
The team dug deeper into how succinate might influence TDG itself. Using computer simulations, biophysical experiments, and enzyme tests, they found that succinate directly binds to a critical spot on TDG, the same region used by alpha-ketoglutarate. Unlike alpha-ketoglutarate, however, succinate pushed TDG into a hyperactive state. This overactivity led to an excess of DNA sites where bases had been cut out but not yet properly repaired. In pancreatic cells grown under dysmetabolic conditions, or treated with cell-permeable succinate, TDG activity and abasic site accumulation both increased, and this depended on the specific TDG site that senses these metabolites.
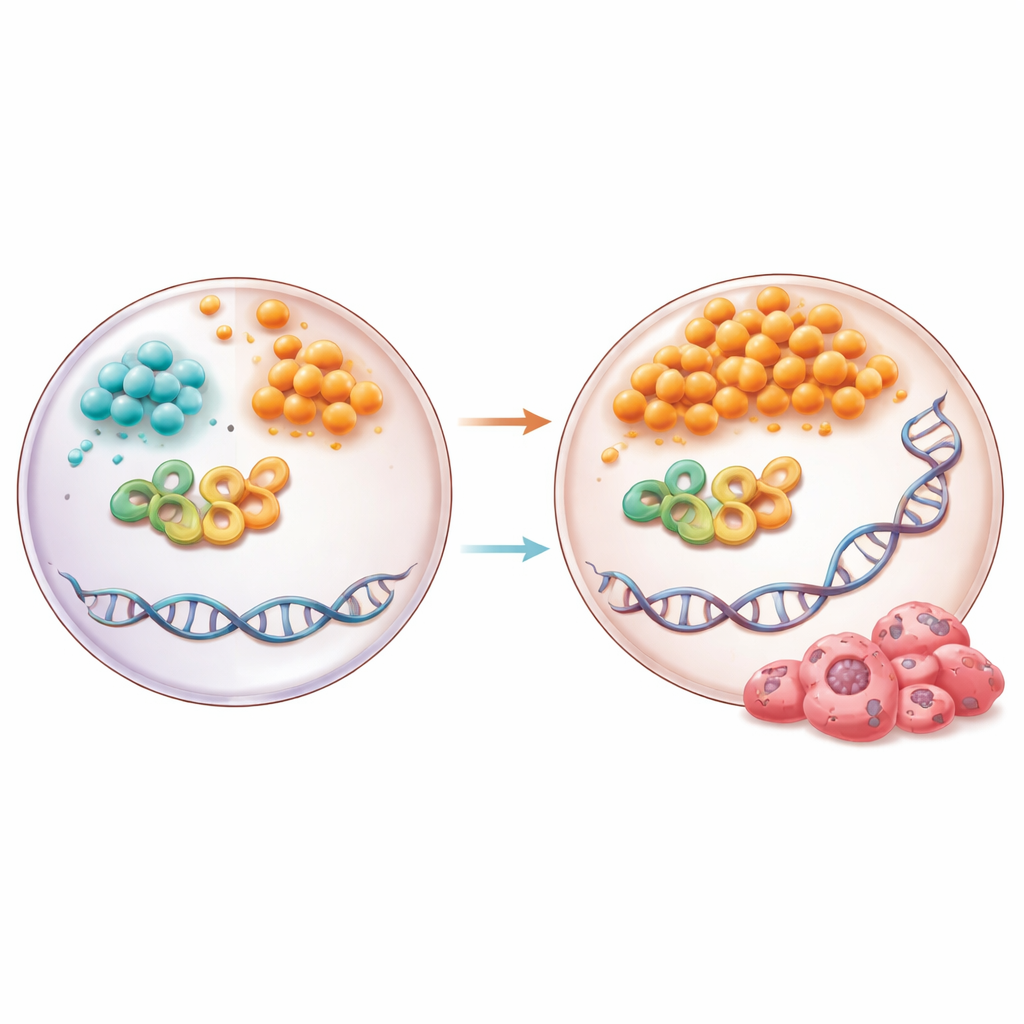
A Broken Finish Line for DNA Repair
Normally, after TDG and related enzymes create an empty spot in DNA, a repair pathway called base excision repair steps in to fill and seal the gap. Two DNA “ligases,” LIG1 and LIG3, perform the crucial final joining step. In both the high-fat–fed mice and the metabolically stressed human pancreatic cells, levels of LIG1 and LIG3 dropped sharply. The study shows that their gene promoters become more methylated in this environment, likely because the methyl-donating chemistry is overly active while demethylation falters. As a result, the ligases are dialed down when they are most needed, causing repair to stall and abasic sites to build up. When the researchers inhibited ligases directly, abasic sites increased, underscoring how essential these enzymes are to preventing DNA damage from piling up.
Can the Damage Be Rebalanced?
Strikingly, restoring alpha-ketoglutarate in metabolically stressed pancreatic cells partially reset this harmful program. Extra alpha-ketoglutarate reduced methylation on the LIG1 and LIG3 promoters, revived their expression, and lowered the number of abasic sites. Together, the findings paint a picture of a metabolic–epigenetic axis: chronic dysmetabolism raises succinate and methyl donors, disrupts TET1–TDG cooperation, hyperactivates TDG, and silences key repair ligases. The result is unstable DNA in rapidly dividing pancreatic cells, which can pave the way for precancerous lesions. For non-specialists, this work suggests that the chemistry of metabolism does not just affect weight and blood sugar; it can quietly reshape how our DNA is maintained, potentially opening new doors for early biomarkers and metabolism-targeted strategies to reduce pancreatic cancer risk.
Citation: Malatesta, S., Vigiano Benedetti, V., Salviati, E. et al. α-ketoglutarate/succinate ratio imbalance impairs thymine DNA glycosylase function and base excision repair process increasing susceptibility to pancreatic cancer. Cell Death Dis 17, 242 (2026). https://doi.org/10.1038/s41419-026-08475-w
Keywords: pancreatic cancer, metabolism, DNA repair, epigenetics, succinate