Clear Sky Science · en
Extracellular vesicles from stem cells rescue cellular phenotypes and behavioral deficits in SHANK3-associated ASD neuronal and mouse models
Tiny Messengers with Big Potential
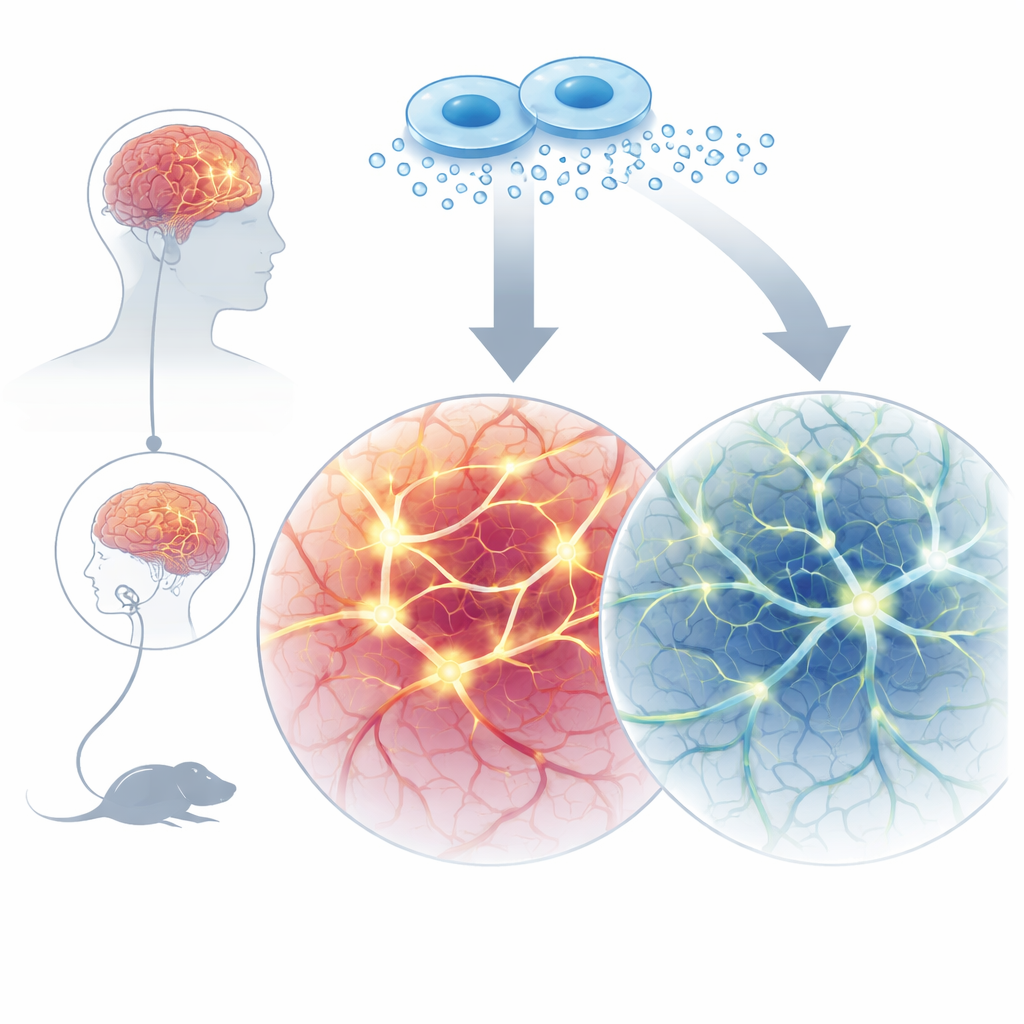
Autism spectrum disorder and related conditions often trace back to subtle glitches in how brain cells connect and communicate, long before any symptoms are visible. This study explores an emerging idea: that microscopic “packages” released by cells—called extracellular vesicles—might both spread harmful changes between brain cells and, if engineered correctly, help reverse them. By working with human stem-cell–derived neurons and a well-established mouse model, the researchers examine how these vesicles influence brain activity in a form of autism tied to the SHANK3 gene, and whether vesicles from healthy stem cells can restore more typical brain function and behavior.
When Brain Wiring Develops Too Fast
Some people with autism or Phelan-McDermid syndrome carry changes in a gene called SHANK3, which helps organize the contact points where neurons talk to one another. In earlier work, the team showed that human neurons grown from a patient’s cells with a SHANK3 mutation matured unusually quickly and fired too many electrical signals, a pattern called hyperexcitability. Similar early overactivity appears in mouse models lacking Shank3. These findings support the idea that, during early development, certain forms of autism are marked not by weakness in brain circuits, but by an initial surge of connections and firing that later leads to imbalance.
Cellular Packages That Can Spread Trouble
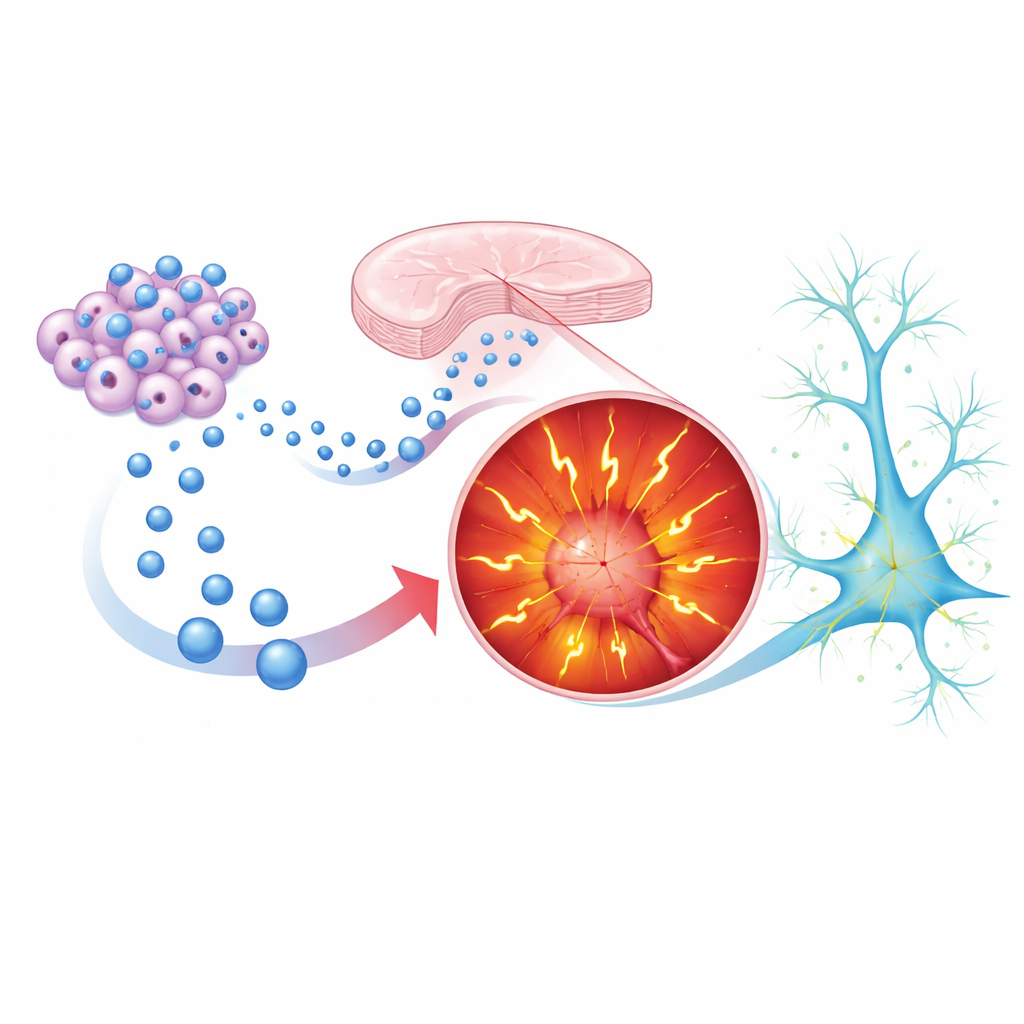
Almost all cells, including neurons, release tiny, membrane-wrapped vesicles packed with proteins, RNA, and other molecules. These extracellular vesicles act as couriers, allowing cells to influence their neighbors. The researchers asked whether vesicles released by SHANK3-mutant neurons might carry signals that alter the behavior of healthy neurons. They grew human cortical neurons from both a patient with a SHANK3 mutation and an unaffected relative, collected vesicles from each group, and then “swapped” them between cultures. Remarkably, healthy neurons exposed to vesicles from SHANK3-mutant neurons began to look and act more like the mutant cells: they fired more action potentials, showed greater spontaneous activity, and displayed electrical properties consistent with hyperexcitability. In contrast, mutant neurons did not improve when treated with vesicles from healthy neurons, suggesting that those vesicles lacked sufficient restorative cargo.
Stem Cell Vesicles as a Calming Influence
Because vesicles can cross biological barriers and are less likely to provoke immune reactions than transplanted cells, they are attractive therapeutic candidates. The team therefore tested vesicles from two stem cell sources: mesenchymal stem cells (derived from bone marrow) and induced pluripotent stem cells (reprogrammed adult cells). When these stem-cell vesicles were added repeatedly to SHANK3-mutant human neurons as they matured, the neurons’ behavior shifted toward normal. Their sodium and potassium currents dropped, their tendency to fire bursts of action potentials declined, and the strength and frequency of their synaptic signals became more like those of control neurons. Proteomic analyses—deep surveys of the proteins inside vesicles—showed that mutant-neuron vesicles were enriched in synaptic structural proteins and actin regulators linked to abnormal wiring, while stem-cell vesicles carried molecules associated with synaptic pruning, plasticity, and homeostasis, such as complement components and growth factors. This contrast hints at why one type of vesicle worsens hyperactivity while another can counter it.
From Neuron Dishes to Living Behavior
To find out whether these effects extend beyond lab dishes, the researchers turned to Shank3B knockout mice, which display autism-like traits. Male mice lacking Shank3 showed normal interest in other mice and in the opposite sex, but struggled with a more subtle task: distinguishing between a stressed mouse and a calm one, a rough stand-in for recognizing others’ emotional states. The scientists gave vesicles from healthy induced pluripotent stem cells intranasally—from early infancy to the juvenile stage—using a noninvasive route already being explored for brain therapies. In adulthood, treated knockout mice regained a clear preference pattern in the emotion-recognition test, while their overall movement remained unchanged. This suggests that stem-cell vesicles helped adjust specific social processing circuits rather than simply making the animals more active or alert.
What This Could Mean for Future Treatments
Altogether, the study paints extracellular vesicles as double-edged messengers: vesicles from SHANK3-deficient neurons can spread hyperactive traits to healthy cells, but vesicles from carefully chosen stem cells can dampen this overactivity and rescue aspects of behavior in mice. For families and clinicians, the work does not yet translate into a ready-made therapy, but it points toward a future in which customized vesicles, loaded with protective cargo, might gently nudge developing brain circuits back toward balance. Because these vesicles can be delivered without surgery and produced at scale from stem cells, they offer a promising path to targeted, less invasive interventions for autism and related neurodevelopmental conditions.
Citation: Choudhary, A., Rosh, I., Hussein, Y. et al. Extracellular vesicles from stem cells rescue cellular phenotypes and behavioral deficits in SHANK3-associated ASD neuronal and mouse models. Cell Death Dis 17, 244 (2026). https://doi.org/10.1038/s41419-026-08474-x
Keywords: autism, SHANK3, extracellular vesicles, stem cell therapy, neurodevelopment