Clear Sky Science · en
Colony-stimulating factor 1 receptor inhibition is neuroprotective to photoreceptors in retinal detachment
Why this research matters for eyesight
Retinal detachment is an emergency that can steal sight even after surgery successfully reattaches the retina. Much of this lasting damage comes from the body’s own cleanup crew of immune cells, which can harm light-sensing cells while trying to help. This study in mice explores whether dialing down a key immune switch, called CSF1R, can protect these fragile photoreceptors and open a new path to preserve vision.
The eye’s cleanup crew and how it can backfire
When the retina peels away from its support layer, photoreceptors suddenly lose access to oxygen and nutrients. Many of these cells die, and the tissue sends out distress signals. Local immune cells in the retina, known as microglia, change shape, move toward the injury, and ramp up their eating of debris. At the same time, immune cells from the bloodstream, including different types of monocytes, stream into the retina. Together, they try to clear dead cells and start repair. But this beneficial reaction can also overshoot, leading to extra inflammation that kills additional, still-viable photoreceptors and worsens sight loss.
A drug that targets a shared immune switch
Both resident microglia and incoming monocytes depend on the same surface receptor, CSF1R, for survival and function. The researchers used PLX5622, a pill-like compound that blocks CSF1R, to ask how interfering with this switch changes immune behavior in retinal detachment. They first mapped its effects in the blood and retina. In the circulation, brief treatment did not change total monocyte numbers but did rebalance their subsets: pro-inflammatory “classical” monocytes increased, while patrolling “non-classical” monocytes dropped. In the eye, PLX5622 efficiently depleted retinal microglia and altered how many blood‑derived immune cells entered the damaged retina and what shapes and behaviors they adopted.
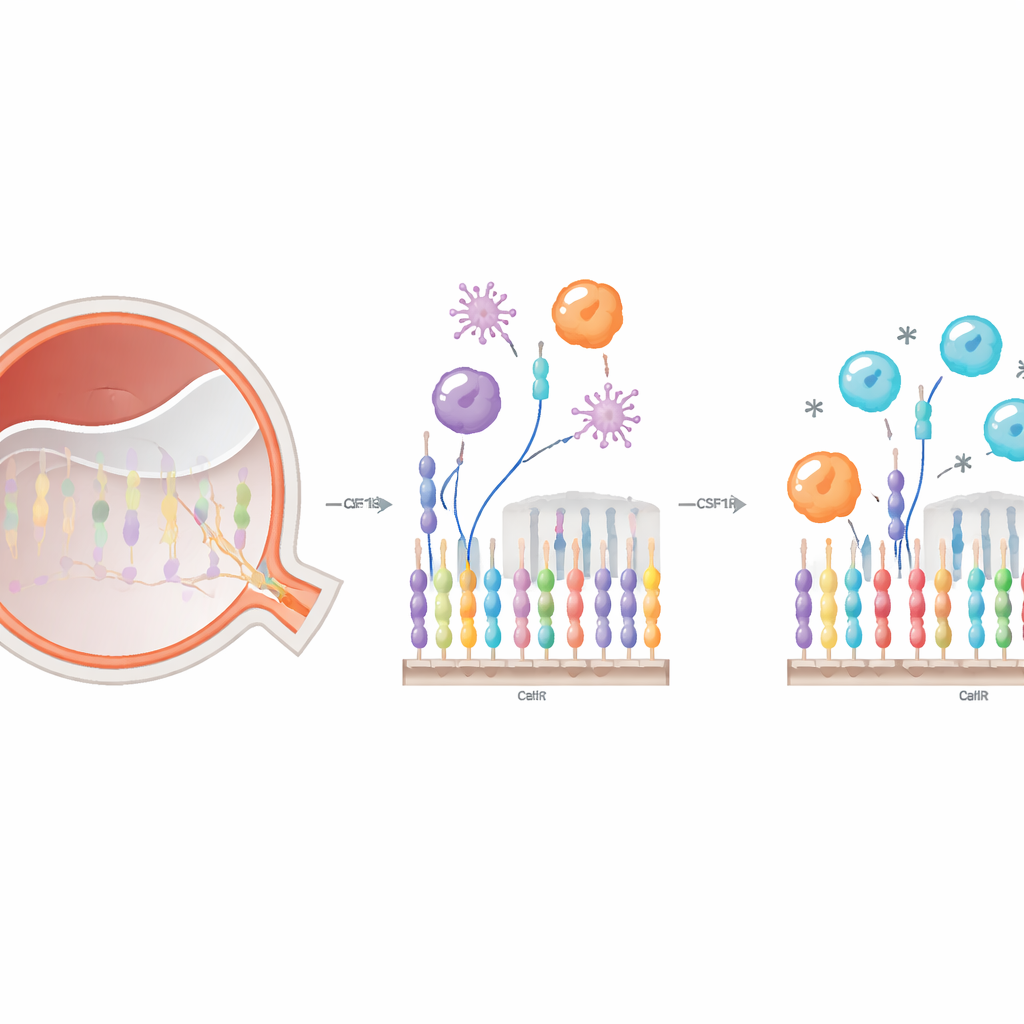
Tracking local and incoming cells with color tags
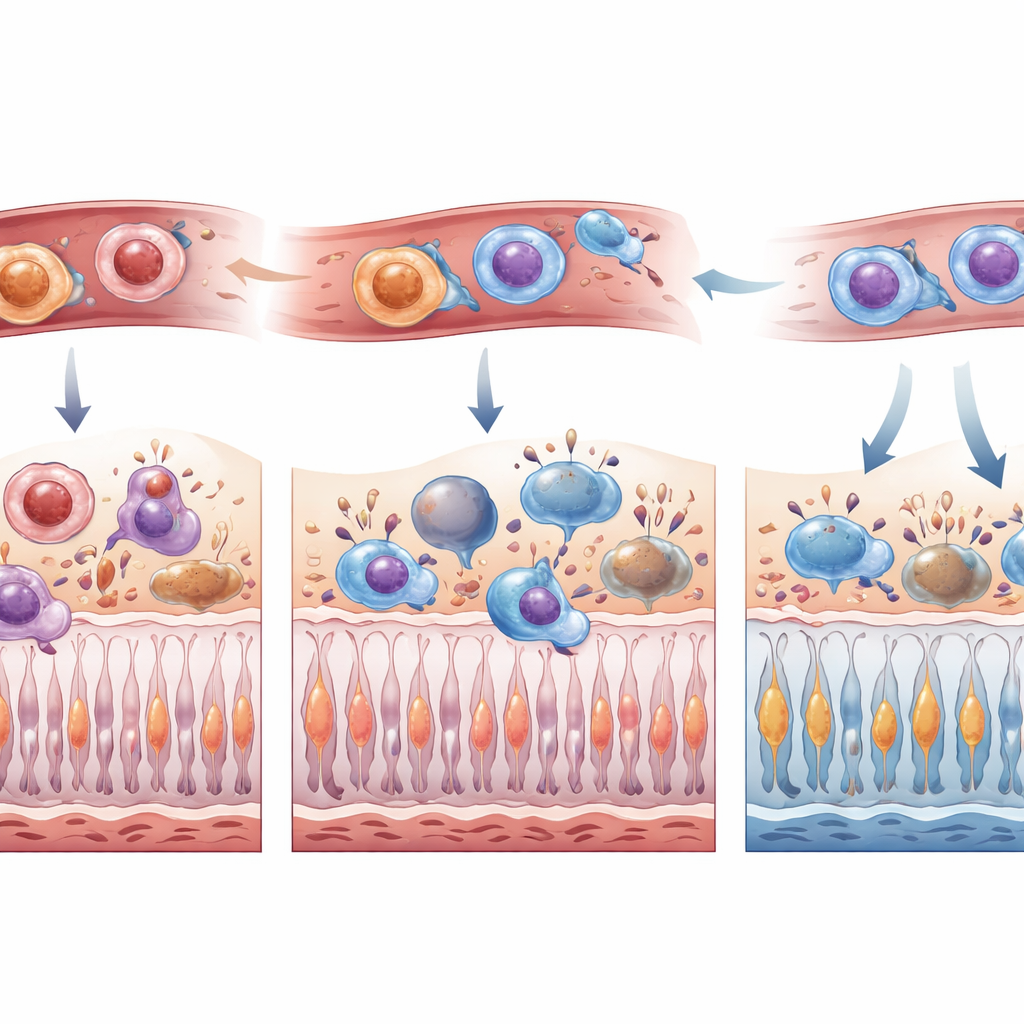
Because microglia and infiltrating monocytes can look very similar once they enter the retina, the team created a dual-label mouse system: resident microglia glowed one color and bone‑marrow–derived cells another. After inducing a controlled retinal detachment, they followed where these cells moved and how they changed over the first week. Without treatment, microglia accumulated mainly in the upper retinal layers, while bone‑marrow–derived cells flooded especially into the subretinal space, often taking on an amoeboid, highly phagocytic form. With CSF1R inhibition, microglia were markedly reduced across layers early on, and fewer bone‑marrow–derived cells, particularly the amoeboid type, reached the retina in the first days after injury. By day seven, the wave of incoming cells had largely caught up, suggesting that CSF1R blockade delays rather than permanently blocks their arrival.
How changing immune balance protects light-sensing cells
To understand what these immune shifts mean for vision, the researchers counted dying and surviving photoreceptors at multiple time points. In the early hours after detachment, animals on PLX5622 had fewer dying photoreceptors and more surviving ones. At one week, both short pre-treatment and continuous treatment preserved the thickness of the photoreceptor layer, a structural readout of cell survival, and reduced the presence of immune cells in the subretinal space. Detailed immune profiling showed that, despite an overall drop in total infiltrating leukocytes, those that did enter the retina under CSF1R inhibition tended to carry a more inflammatory signature. The authors suggest that this combination—fewer cells, but more tuned toward rapid debris clearance—may speed up the safe removal of dying photoreceptors while sparing their still-healthy neighbors.
What this could mean for future treatments
This work shows that a systemic CSF1R inhibitor can reshape both local and circulating immune responses in retinal detachment and, somewhat counterintuitively, that a more inflammatory but smaller cleanup crew can protect vision. By temporarily depleting microglia, delaying the first rush of blood‑derived cells, and shifting monocyte subtypes, PLX5622 created a time window in which early immune‑driven damage was reduced and more photoreceptors survived. While the study was done in mice and the balance of helpful and harmful inflammation will differ across eye diseases, these findings support CSF1R inhibition as a promising starting point for drugs designed to fine‑tune the immune response and preserve sight after retinal injury.
Citation: Pastor-Puente, S., Jung, R., Gonzalez-Buendia, L. et al. Colony-stimulating factor 1 receptor inhibition is neuroprotective to photoreceptors in retinal detachment. Cell Death Dis 17, 264 (2026). https://doi.org/10.1038/s41419-026-08470-1
Keywords: retinal detachment, microglia, photoreceptor survival, neuroinflammation, CSF1R inhibition