Clear Sky Science · en
Rewiring melanoma cell fate: TRPM8 modulators trigger apoptosis and boost NK cell cytotoxicity
Why turning a “cold sensor” against skin cancer matters
Melanoma is a dangerous form of skin cancer that often learns to dodge both drugs and the immune system. This study explores an unexpected weakness in melanoma cells: a protein called TRPM8, best known as the sensor that lets nerve cells feel cold or menthol. The researchers show that carefully designed drugs that block TRPM8 can push melanoma cells to self-destruct while also making them easier for natural killer (NK) immune cells to attack. This dual action hints at a new way to treat stubborn, advanced melanoma.
A hidden switch on melanoma cells
By mining large cancer databases and examining patient-derived melanoma cells in the lab, the team found that TRPM8 is much more abundant in metastatic melanoma than in normal skin, including healthy melanocytes and dermal fibroblasts. Using imaging, they showed that TRPM8 sits both on the cell surface and on internal membranes, placing it in strategic spots to influence cell behavior. When they applied a panel of new TRPM8-targeting molecules, two compounds (named 4 and 9) consistently stood out: they sharply reduced the survival of melanoma cells over time while leaving normal skin cells largely unharmed. Genetic tests confirmed that when TRPM8 was removed, these drugs lost their killing power, and when TRPM8 was increased, melanoma cells became even more sensitive, proving that the effect depends on this specific channel. 
Forcing cancer cells into an energy crisis
Unlike classic TRPM8 activators such as menthol, which drive a surge of calcium into cells, these new blockers did not trigger calcium changes. Instead, they launched a wave of oxidative stress inside melanoma cells. The compounds rapidly boosted reactive oxygen species, especially within mitochondria, the cell’s energy factories. This oxidative burst caused the mitochondria to lose their electrical charge, fragment, and leak cytochrome c, a key step in switching on the internal “suicide” program of the cell. Antioxidants that mopped up these reactive molecules largely rescued the cells, showing that redox imbalance is not a side effect but the engine of cell death. Downstream, classic death markers such as caspase-3 activation and PARP cleavage confirmed that the cells were undergoing programmed apoptosis rather than a random form of damage.
Disarming survival signals and awakening a guardian
The oxidative hit set off a broader stress response. The drugs activated ATM, a sensor of DNA damage, and led to the build-up and activation of p53, a famous tumor-suppressor protein that decides whether a damaged cell should repair itself or die. At the same time, the compounds weakened one of melanoma’s main survival routes: the PI3K–AKT pathway. Normally, TRPM8 physically interacts with PI3K components to help keep AKT in its active, growth-promoting form. After treatment, this partnership broke apart and AKT activity dropped, tipping the balance further toward cell death. Importantly, these changes were pronounced in melanoma cells but not in noncancerous cells, suggesting a therapeutic window where the tumor is hit hard while healthy tissue is spared.
Making tumors more visible to immune “hit squads”
Melanoma’s danger also lies in its talent for escaping immune surveillance. The researchers therefore asked what happens to tumor cells that survive low doses of the TRPM8 blockers over longer periods. They discovered that these lingering cells started displaying more of a surface marker called ULBP1, which acts like a distress flag recognized by NKG2D receptors on NK cells. Other related flags did not change, indicating a selective effect. In three-dimensional melanoma spheroids that better mimic real tumors, pretreatment with TRPM8 blockers not only shrank the structures and increased cell death but also made them far more vulnerable to NK cell attack. When ULBP1 or NKG2D was blocked with antibodies, this extra killing largely disappeared, showing that the improved immune clearance runs through this specific handshake between tumor and NK cells. 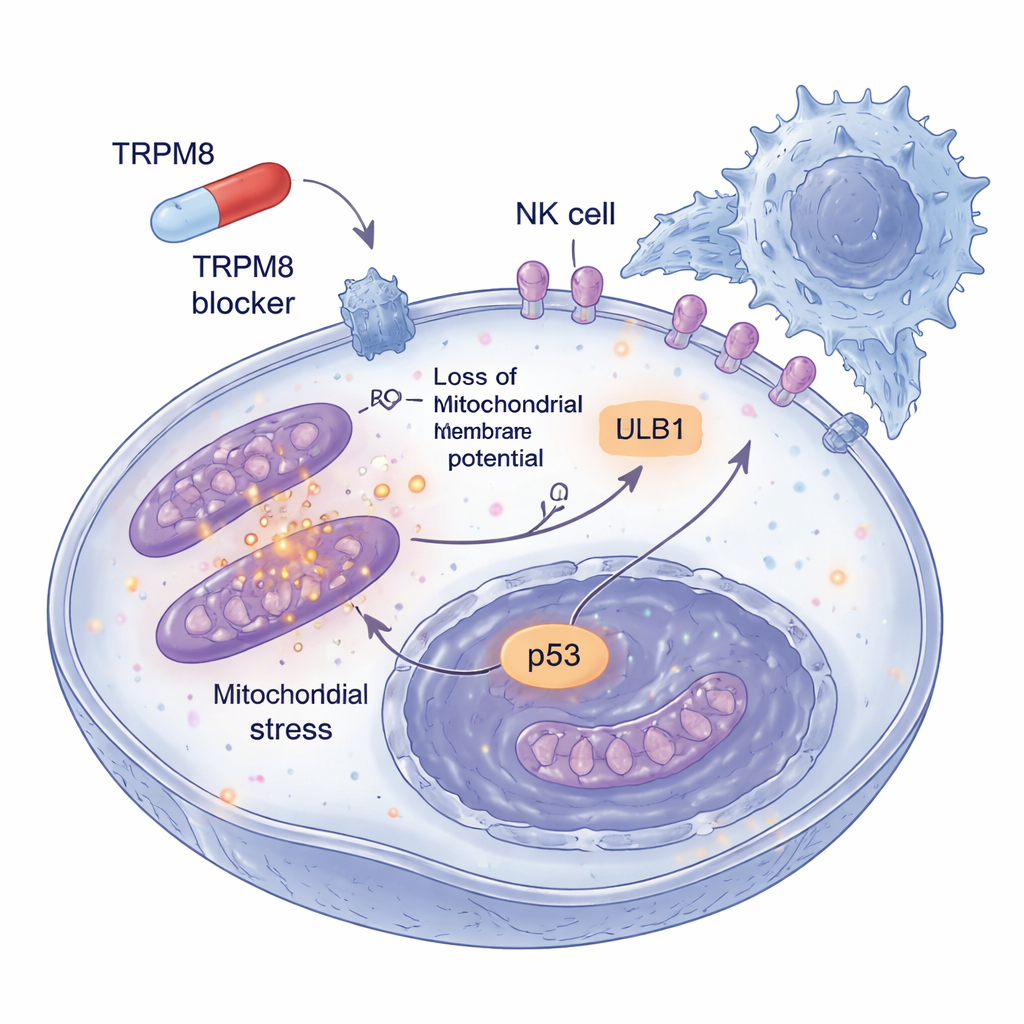
What this could mean for future melanoma care
In simple terms, blocking the TRPM8 “cold sensor” turns it into a therapeutic lever: it drives melanoma cells into a fatal mitochondrial and DNA crisis while flagging them for destruction by NK cells. Because the drugs spare normal skin cells and do not harm NK cells themselves, they offer a promising foundation for new treatments. The authors suggest that TRPM8 blockers could be combined with existing immunotherapies to attack advanced, drug-resistant melanoma on two fronts: killing tumor cells directly and making the survivors easier for the immune system to find and destroy.
Citation: Sorrentino, C., Lauretta, C., D’Angiolo, R. et al. Rewiring melanoma cell fate: TRPM8 modulators trigger apoptosis and boost NK cell cytotoxicity. Cell Death Dis 17, 223 (2026). https://doi.org/10.1038/s41419-026-08469-8
Keywords: melanoma, TRPM8, mitochondria, oxidative stress, natural killer cells