Clear Sky Science · en
Lactylation-drived TRIM29 induces invasive behavior and lymph node metastasis in gastric cancer via hnRNPA1-mediated Wnt/β-catenin pathway
Why this research matters
Gastric (stomach) cancer is one of the world’s deadliest cancers largely because it spreads early to nearby lymph nodes, making cure by surgery difficult. This study uncovers how a chemical change linked to tumor metabolism supercharges a protein called TRIM29, helping cancer cells invade surrounding tissue, grow new lymph vessels, and resist chemotherapy. Understanding this chain of events points to new ways to slow or even block the spread of stomach cancer.
A dangerous partnership in stomach tumors
The researchers began by comparing stomach cancer samples with nearby healthy tissue from 100 patients, as well as large public gene databases. They found that TRIM29, a protein previously tied to immune regulation and other cancers, was consistently higher in tumor cells. Patients whose tumors had more TRIM29 were more likely to show cancer in their lymph nodes and blood vessels and had worse overall survival. These patterns suggested that TRIM29 is not just present, but actively involved in helping stomach cancer behave more aggressively.
How cancer cells learn to travel
To see what TRIM29 actually does, the team altered its levels in stomach cancer cell lines. When they reduced TRIM29, the cells became noticeably less mobile and less able to push through 3D gels that mimic body tissue. Boosting TRIM29 had the opposite effect, making cells more invasive. The researchers also examined lymphatic endothelial cells, the cells that line lymph vessels. TRIM29-rich cancer cells encouraged these vessel cells to sprout and form tube-like structures, a sign of new lymph vessel growth (lymphangiogenesis). In mouse models, tumors with high TRIM29 produced more liver and lymph node metastases, while those with TRIM29 knocked down spread less and formed fewer lymphatic vessels.
Molecular relay: protecting a key regulator
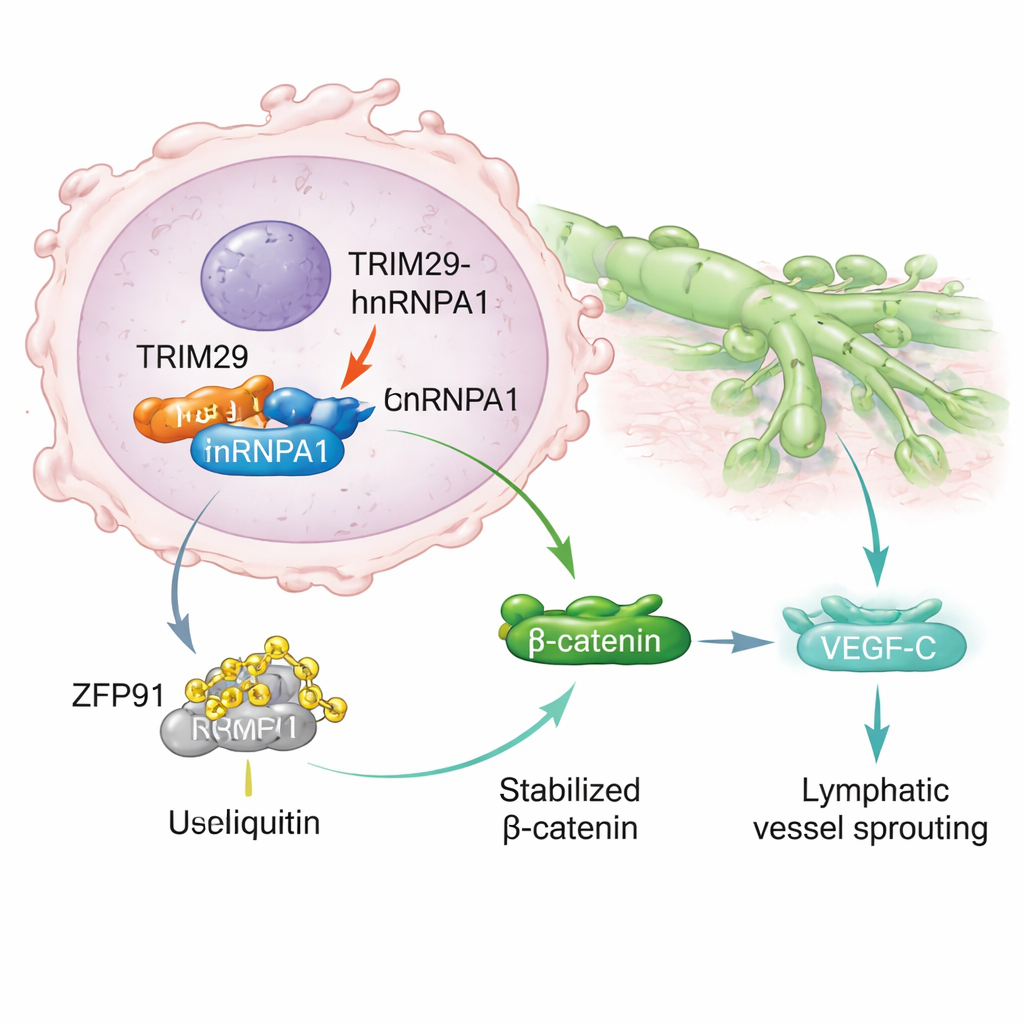
Diving deeper, the scientists discovered that TRIM29 physically binds to another protein, hnRNPA1, which is known to influence how genes are processed and linked to cancer spread. Normally, hnRNPA1 can be tagged for destruction by yet another protein, an enzyme called ZFP91, through the cell’s waste-disposal system. TRIM29 interferes with this process by competing with ZFP91 for binding to hnRNPA1. When TRIM29 is abundant, it shields hnRNPA1 from being tagged and broken down. As a result, hnRNPA1 becomes more stable and accumulates in cancer cells, setting off a chain of signals that end in activation of the Wnt/β-catenin pathway, a well-known driver of tumor growth and movement. This in turn boosts production of VEGF‑C, a molecule that stimulates new lymph vessels, giving cancer cells more “roads” to escape.
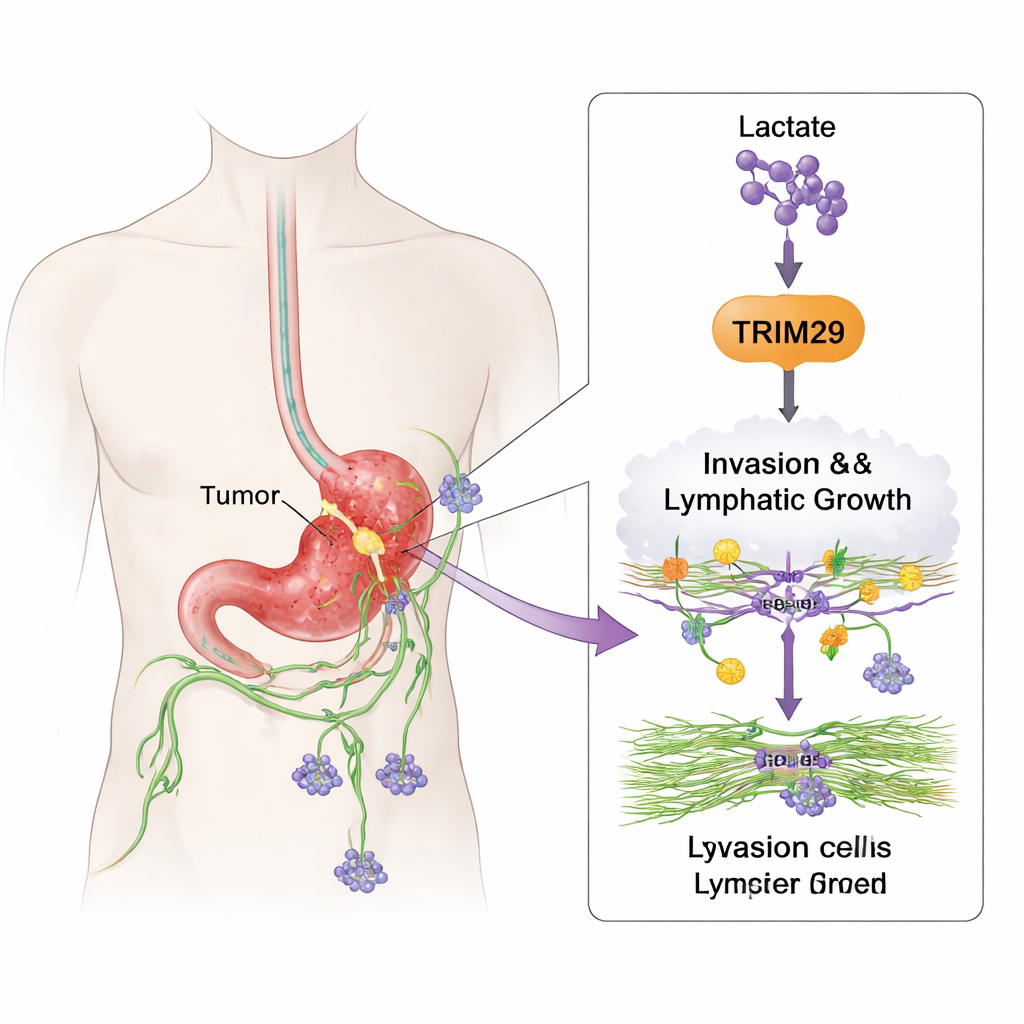
Fueling cancer with lactate
The study also links this invasive behavior to how tumors process sugar. Rapidly growing cancers often rely on a form of metabolism that produces large amounts of lactate, a byproduct once considered mere waste. Here, the authors show that lactate can chemically modify histone proteins that help package DNA, specifically at a site called H3K9. This “lactylation” acts like an on-switch for the TRIM29 gene, turning up its activity. When they lowered lactate production using metabolic drugs, TRIM29 levels fell; when they raised lactate, TRIM29 rose. Stomach cancer tissues from patients showed higher levels of this H3K9 lactylation, which correlated with higher TRIM29, more lymph node spread, and poorer survival. In essence, the tumor’s altered metabolism helps write a more aggressive genetic program.
New ways to boost chemotherapy
Lymph vessels do more than carry cancer cells; they can also drain chemotherapy drugs away from tumors. Using patient-derived tumor grafts grown in mice, the researchers tested whether blocking TRIM29 and lymph vessel growth could improve the effect of 5‑fluorouracil (5‑FU), a standard stomach cancer drug. Inhibiting TRIM29 or blocking lymphangiogenesis each made 5‑FU more effective at shrinking tumors and reducing cell division. Combining both strategies produced the strongest response, suggesting a potential way to overcome some forms of drug resistance in advanced disease.
What this means for patients
This work paints a step-by-step picture: stomach tumors generate excess lactate, which switches on TRIM29; TRIM29 then protects hnRNPA1, stabilizes a growth pathway, and ramps up lymph vessel formation and spread to lymph nodes. Clinically, high TRIM29 and related lactylation marks identify patients at higher risk of aggressive disease. In the future, drugs that curb lactate signaling, block TRIM29, or prevent lymph vessel growth could be combined with existing chemotherapy to keep stomach cancer from spreading and to make treatments work better.
Citation: Hua, R., Yu, J., Niu, Y. et al. Lactylation-drived TRIM29 induces invasive behavior and lymph node metastasis in gastric cancer via hnRNPA1-mediated Wnt/β-catenin pathway. Cell Death Dis 17, 222 (2026). https://doi.org/10.1038/s41419-026-08468-9
Keywords: gastric cancer metastasis, TRIM29, lactate and lactylation, lymphangiogenesis, Wnt beta-catenin pathway