Clear Sky Science · en
KIF20A inhibits TRIM21-dependent ubiquitination of DHX9 to boost SOX2 stability, enhancing OSCC stemness and ferroptosis resistance
Why this research matters for oral cancer patients
Oral squamous cell carcinoma, a common form of mouth cancer, often comes back and resists treatment. Many failures of surgery, chemotherapy, and radiation trace back to a small, stubborn group of "cancer stem cells" that can regrow the tumor and resist cell death. This study uncovers a key molecular circuit that helps those cells stay alive and resist a newer form of cell death called ferroptosis, and it points to a drug strategy that could make standard chemotherapy work better.
A hidden driver in mouth tumors
The researchers began by comparing tumor samples from oral cancer patients with nearby healthy tissue. They found that a protein called KIF20A was consistently much higher in cancer cells. When they examined larger patient datasets, people whose tumors had more KIF20A tended to live for a shorter time, suggesting that this protein is linked to aggressive disease. 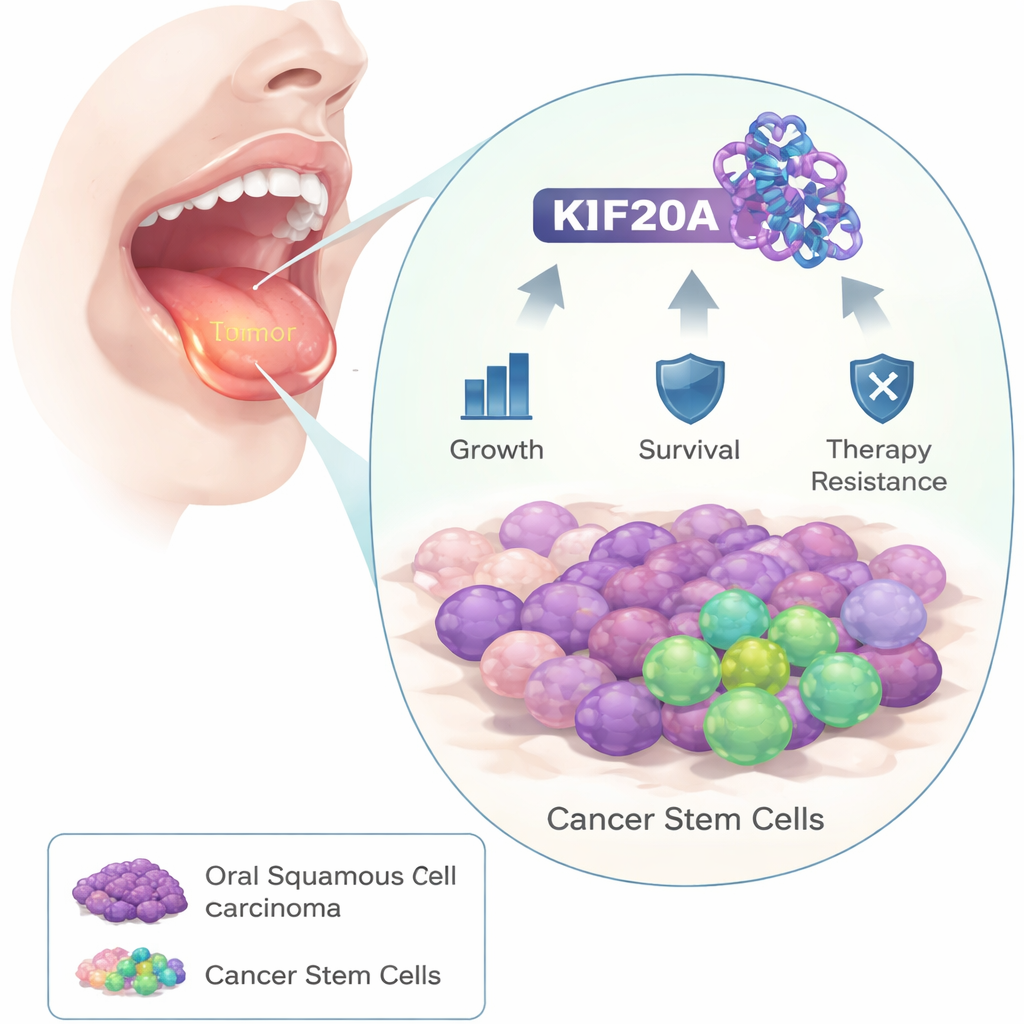
How KIF20A protects a key RNA helper
To understand what KIF20A actually does, the team used protein-mapping methods to look for its partners inside cancer cells. A central partner that emerged was DHX9, a protein that helps manage RNA molecules and influences how long certain messages last in the cell. The scientists discovered that KIF20A binds to DHX9 and keeps it from being marked for destruction by another protein, the E3 ligase TRIM21, which normally tags DHX9 with "ubiquitin" chains that send it to the cell’s protein shredder. By blocking this tagging, KIF20A slows DHX9 breakdown and increases its levels, especially in the cell’s cytoplasm, where it can act on specific RNA targets.
From RNA stability to cancer stem cells and ferroptosis resistance
With more stable DHX9, another crucial player comes into focus: SOX2, a master gene that helps cells stay in a stem-like state and resist treatment. The study shows that DHX9 protects SOX2’s RNA message from decaying, allowing more SOX2 protein to be made. In oral cancer cells, extra KIF20A led to higher DHX9 and SOX2, more tumorsphere formation in culture, and a greater fraction of cells expressing stem cell markers—signs of stronger "cancer stemness." KIF20A also made cells less vulnerable to ferroptosis, a type of iron-driven, lipid-related cell death that is being explored as a way to kill resistant cancer cells. When KIF20A was switched off, tumors shrank more readily in mice, and markers of ferroptosis went up, indicating that the tumors had lost this protective shield.
A molecular circuit with a druggable weak spot
Putting these pieces together, the authors propose a KIF20A–DHX9–SOX2 axis: KIF20A stabilizes DHX9, DHX9 stabilizes SOX2 RNA, and SOX2 in turn maintains cancer stem cells and blocks ferroptosis. This circuit also feeds into the PI3K–AKT pathway, a major growth and survival route in many cancers. 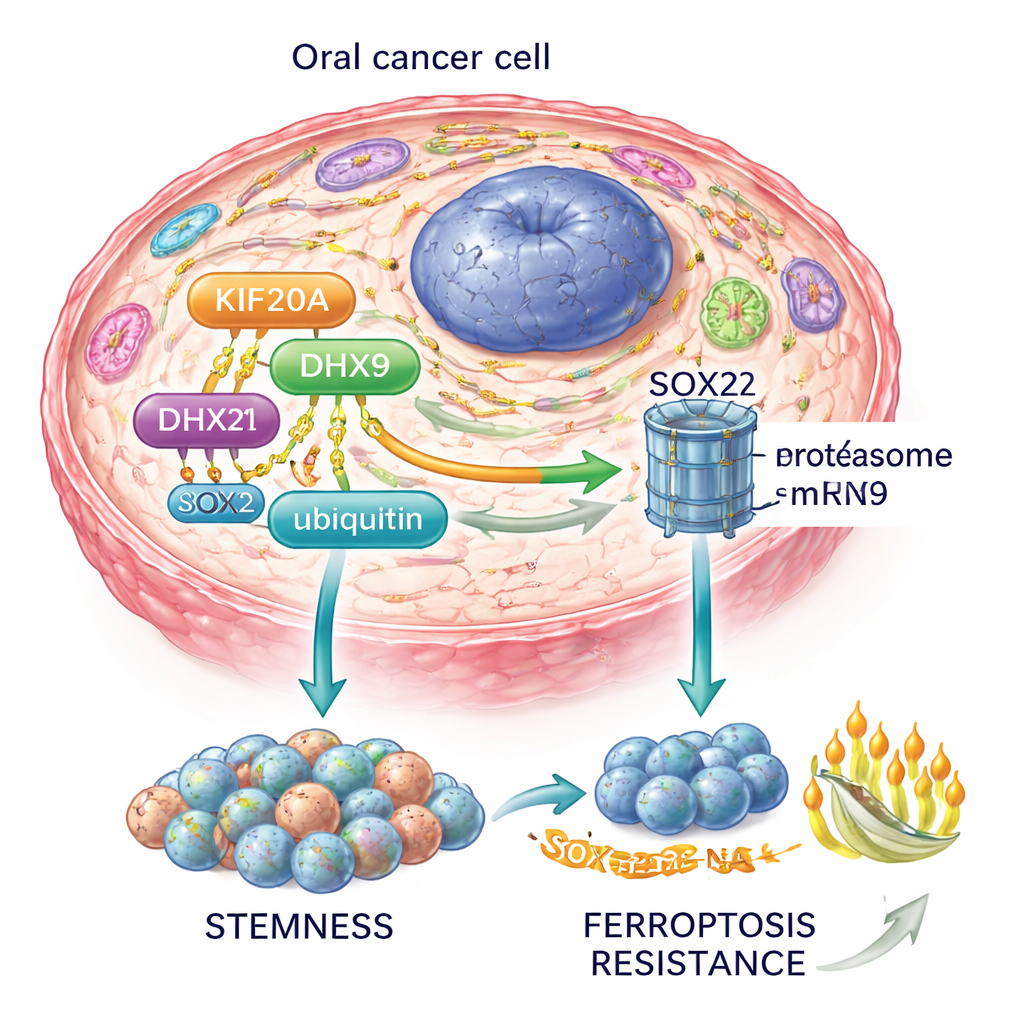
A candidate drug to weaken resistant tumors
To move toward treatment, the team searched large drug–gene databases for compounds predicted to counteract KIF20A-related gene patterns. They identified ENMD-2076, an oral multitarget kinase inhibitor already tested in other cancers. In oral cancer cells with high KIF20A, ENMD-2076 lowered KIF20A, DHX9, and SOX2 levels, dampened stemness-related signaling, and slowed growth. When combined with the standard chemotherapy drug cisplatin, ENMD-2076 produced a stronger anticancer effect than either drug alone in both cell cultures and mouse tumors, without obvious added toxicity.
What this means for future therapies
For a non-specialist, the takeaway is that this study reveals how one overactive protein, KIF20A, helps mouth tumors keep a small pool of "seed" cells alive and shielded from a promising form of cell death. By mapping this chain—from KIF20A to DHX9 to SOX2—the researchers highlight new targets for drugs and show that an existing experimental drug, ENMD-2076, can weaken this axis and make cisplatin more effective in models. While more work is needed to test safety and confirm benefits in patients, the findings support a future in which blocking this circuit could help prevent relapse and overcome drug resistance in oral cancer.
Citation: Zhang, Z., Li, Y., Hu, J. et al. KIF20A inhibits TRIM21-dependent ubiquitination of DHX9 to boost SOX2 stability, enhancing OSCC stemness and ferroptosis resistance. Cell Death Dis 17, 218 (2026). https://doi.org/10.1038/s41419-026-08467-w
Keywords: oral squamous cell carcinoma, cancer stem cells, ferroptosis, KIF20A, SOX2