Clear Sky Science · en
Selenomethionine as a dual-mechanism ferroptosis inhibitor: selenium-supply-driven GPX4 biosynthesis beyond transsulfuration and reductive-capacity-mediated ROS scavenging independent of GPX4 activity
Why protecting our cells from a “rust-like” death matters
Cells in our body can die in many ways, and one of the newest and most intriguing is called ferroptosis—a kind of iron-driven, “rust-like” damage to cell membranes. This process is now linked to diseases ranging from kidney injury to neurodegeneration. The trace element selenium, which many people know from dietary supplements, turns out to play a key role in stopping this dangerous form of cell death. This study explores how a common selenium-containing amino acid, selenomethionine, can shield cells from ferroptosis through not just one, but two distinct protective strategies.
A new defender found in a huge drug search
To find small molecules that can block ferroptosis, the researchers screened thousands of compounds from FDA-approved drugs and natural products. They used a chemical called RSL3 to trigger ferroptosis in human cancer cell lines and then monitored cell death and the buildup of damaged, oxidized fats in cell membranes. Among all candidates, selenomethionine emerged as a strong protector: it reduced both lipid damage and cell death across different cell types and in two major ferroptosis models—one driven by direct GPX4 inhibition (with RSL3) and another by starving cells of cystine, a building block for the antioxidant glutathione. These results positioned selenomethionine as a broad and robust ferroptosis inhibitor. 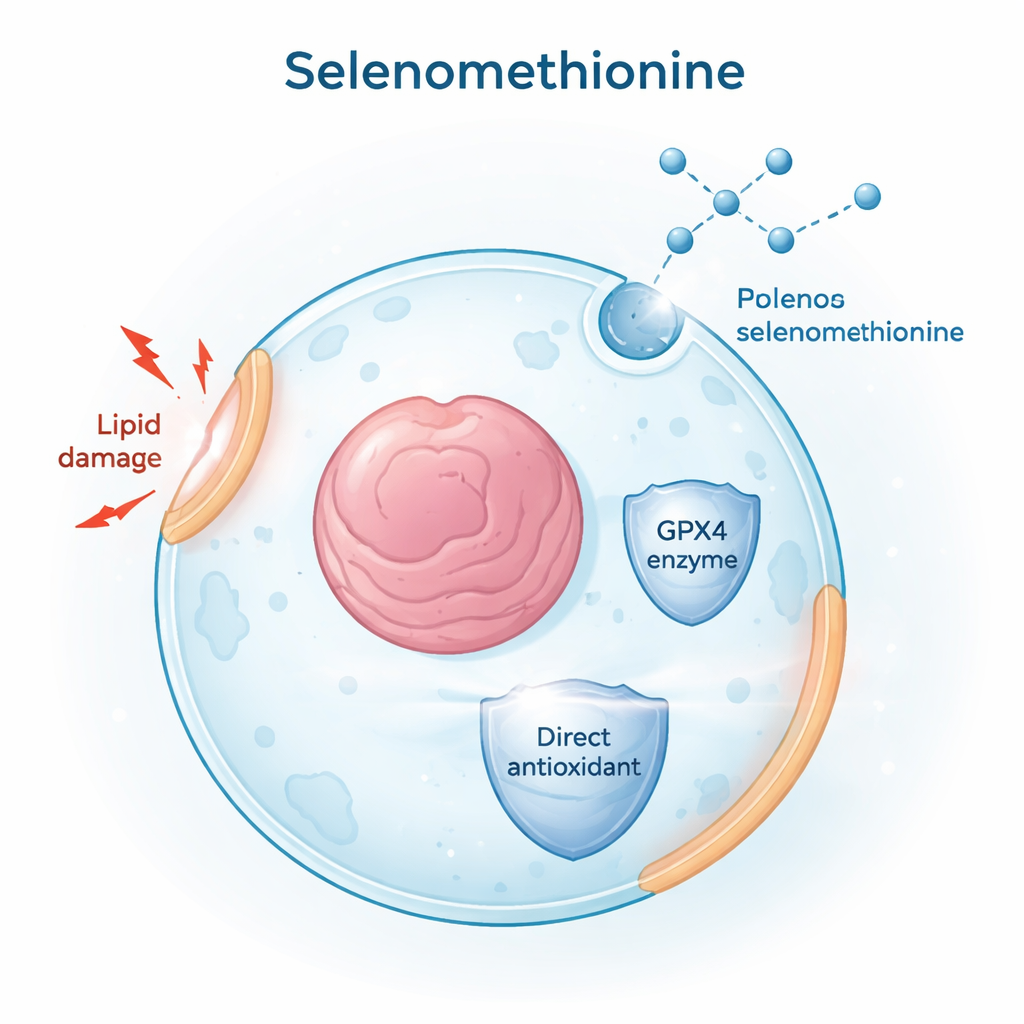
Selenium supply for a key protective enzyme
A core anti-ferroptosis shield in cells is an enzyme called GPX4, which depends on selenium to function. The team set out to determine whether selenomethionine simply feeds this pathway or does more. They traced how selenium from selenomethionine could be converted into forms suitable for building GPX4, even when a usual route, called the transsulfuration pathway, was impaired or blocked. Using genetic knockdowns and chemical inhibitors of enzymes in these sulfur–selenium pathways, they found that selenomethionine still boosted GPX4 levels and protected cells. This showed that cells can tap multiple metabolic routes to extract selenium from selenomethionine and keep GPX4 production going, even in tissues where the classic pathway is weak.
Protection that does not depend on the usual enzyme
Surprisingly, when the researchers completely removed GPX4 from cells using CRISPR gene editing, selenomethionine continued to guard against ferroptosis. It still lowered membrane lipid damage and cell death, indicating that GPX4 is not the whole story. Additional experiments hinted that some of this protection involves selenomethionine being built into newly made proteins, but that this is only part of the effect. The authors then focused on the chemical reactivity of selenomethionine itself. They found that it can directly neutralize reactive oxygen species (ROS), including hydrogen peroxide, and is converted into an oxidized “sulfoxide” form. Mass spectrometry confirmed this product, and giving cells the sulfoxide version also reduced ferroptosis, implying that cells can recycle it back to active selenomethionine in a redox cycle.
From cell culture to protecting kidneys in living animals
To test whether these mechanisms matter in a whole organism, the team turned to a mouse model of acute kidney injury caused by the cancer drug cisplatin, a condition known to involve ferroptosis. Mice treated with cisplatin showed weight loss, swollen kidneys, poor kidney function, and high levels of ferroptosis markers. When the animals received selenomethionine, many of these problems improved: body weight recovered, kidney swelling decreased, blood measures of kidney function normalized, inflammatory signals fell, and chemical markers of lipid damage dropped. Kidney tissue also showed higher GPX4 levels and fewer structural injuries. These benefits were comparable to those seen with ferrostatin-1, a well-known experimental ferroptosis blocker. 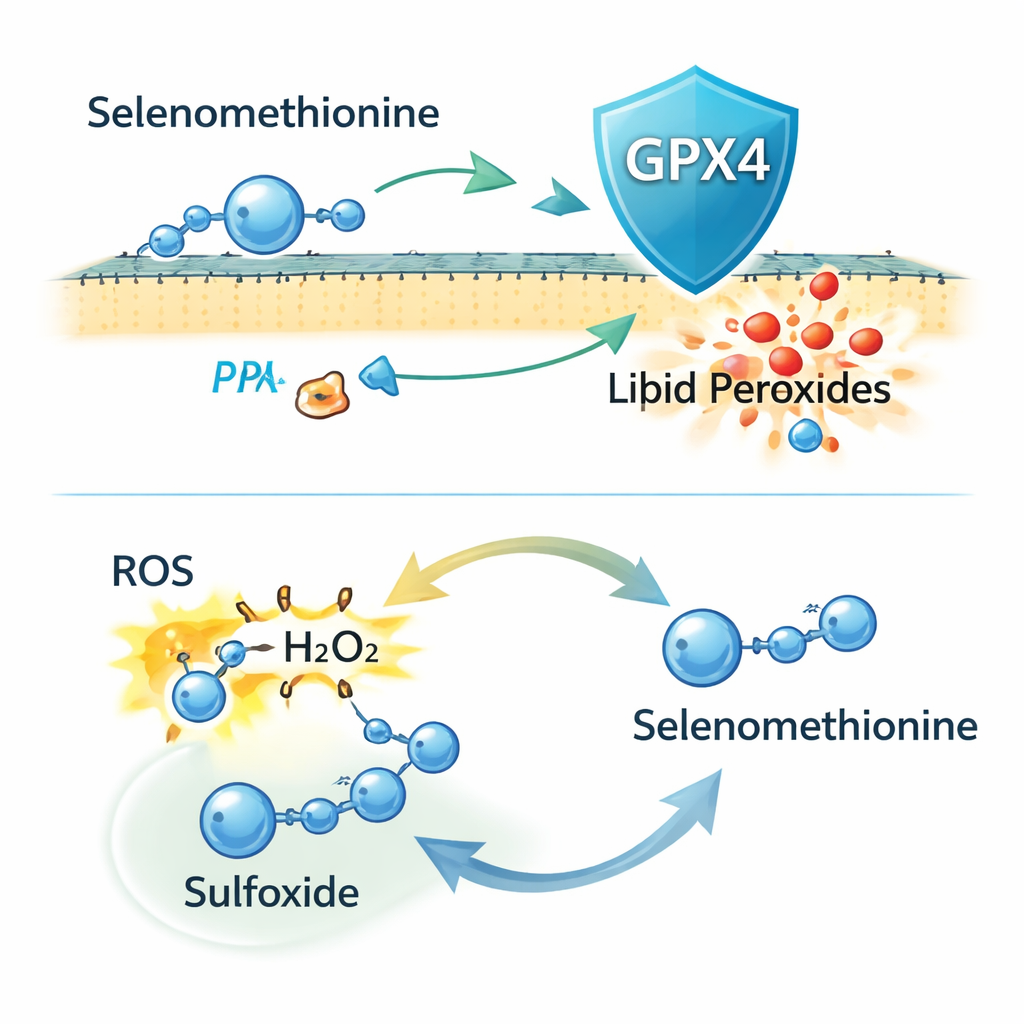
What this means for future therapies
In everyday terms, this work suggests that selenomethionine acts like a dual-purpose fire extinguisher against an iron-fueled “rusting” of cell membranes. On one hand, it supplies selenium so cells can build more of the GPX4 enzyme, which acts as an internal clean-up crew for damaged fats. On the other hand, selenomethionine itself can soak up harmful oxidants and be recycled, offering GPX4-independent backup protection. Together, these features make selenomethionine a promising candidate for preventing or treating diseases where ferroptosis plays a central role, such as drug-induced kidney damage and potentially other conditions driven by runaway oxidative stress.
Citation: Xia, C., Sun, X., Shao, J. et al. Selenomethionine as a dual-mechanism ferroptosis inhibitor: selenium-supply-driven GPX4 biosynthesis beyond transsulfuration and reductive-capacity-mediated ROS scavenging independent of GPX4 activity. Cell Death Dis 17, 224 (2026). https://doi.org/10.1038/s41419-026-08466-x
Keywords: ferroptosis, selenium, selenomethionine, oxidative stress, acute kidney injury