Clear Sky Science · en
The chaperonin TRiC component Cct3 is required for axonal transport, myelination, and neuromuscular junction refinement
When Nerve Wiring Goes Wrong
Our brains and bodies rely on long, cable-like nerve fibers to carry signals quickly and reliably. To do this job, many nerves are wrapped in insulating myelin, and their endings must form precise contacts with muscles. This paper explores what happens when a key cellular “helper” for protein folding, called Cct3, fails. Using zebrafish and human tissue, the authors show how this single component of a large chaperone machine can disrupt myelin, nerve–muscle connections, and the internal transport system that keeps nerve cells alive and functional.
A Cellular Tailor for Building Nerves
Inside every cell, newly made proteins must be folded into the right shapes, much like carefully tailoring a suit. The TRiC complex, which includes the Cct3 subunit, is one of the major “tailors,” helping about 10% of all cellular proteins—especially the structural proteins actin and tubulin—fold correctly. These building blocks form the cell’s inner scaffolding and the tracks along which vital cargo is moved. In patients carrying faulty versions of CCT3 and related genes, brain scans reveal reduced white matter (the myelin-rich wiring of the brain) and problems in the peripheral nerves and muscles. The authors set out to understand how losing Cct3 derails nerve development and function.
Zebrafish as a Window into Nerve Insulation
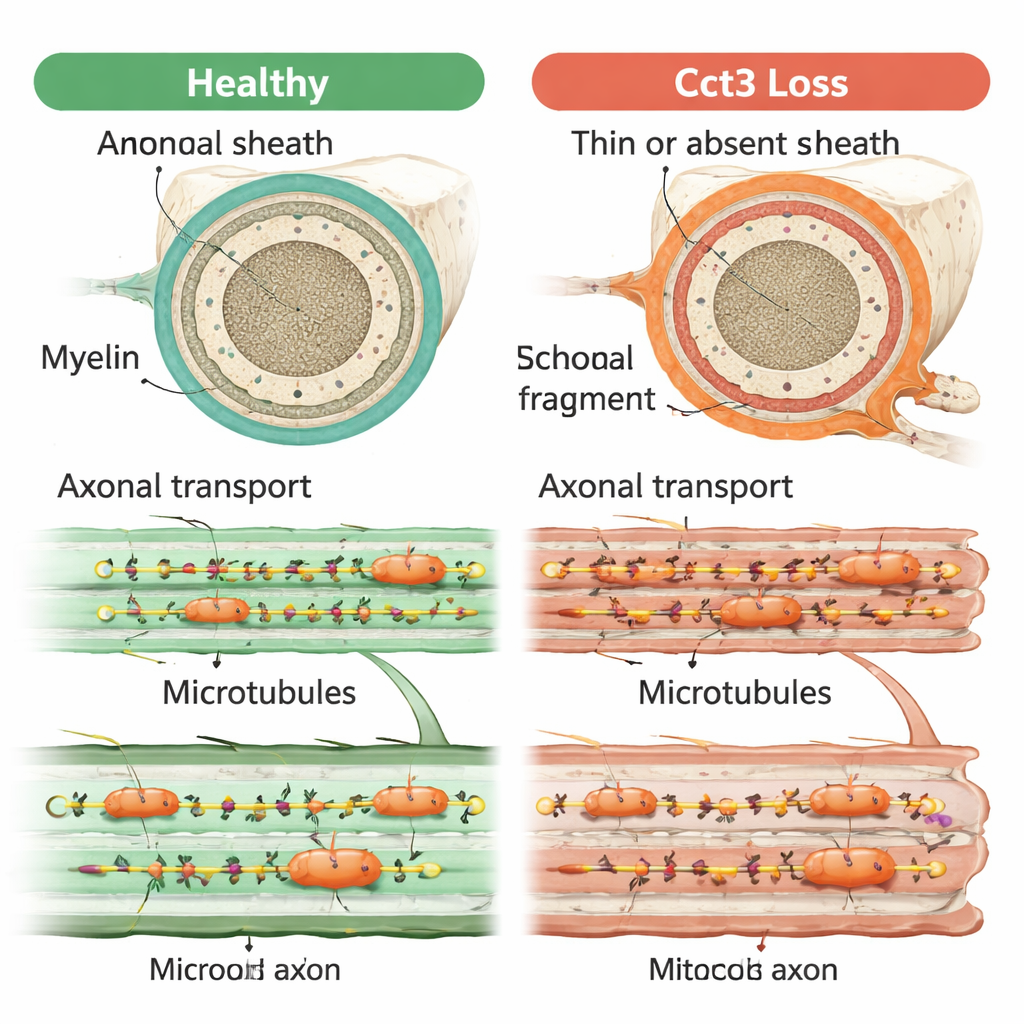
The team used CRISPR/Cas9 gene editing to create zebrafish that lack functional cct3. These tiny fish showed smaller brains and eyes, fluid buildup around the heart, and weak responses to touch, and they died within a few days. When the researchers restored normal cct3 by injecting its messenger RNA, many of these defects improved, confirming that Cct3 loss was the root cause. Focusing on myelin, they used fluorescent markers and electron microscopy to visualize the insulating layers around nerves. In normal fish, myelin-forming cells in the brain and peripheral nerves wrapped axons with neat, layered sheaths by day four of development. In contrast, cct3 mutants had far fewer myelinating cells and essentially no proper myelin around many axons, despite those axons appearing normally shaped on the surface.
Fragile Support Cells and Strained Nerve–Muscle Links
To find out why myelin was missing, the authors examined Schwann cells and related neural crest cells—the support cells that wrap peripheral nerves. In normal fish, these cells extended long, slim processes and then refined their shape as they formed myelin segments. In mutants, they rounded up, shortened their processes, and fragmented. Their nuclei broke apart in a pattern typical of programmed cell death, and a key death marker (activated Caspase 3) appeared, showing that many of these cells were dying early. Meanwhile, at the neuromuscular junctions where nerves meet muscle, the pattern of receptor “hot spots” on muscles failed to refine. Instead of many small, precise clusters, mutants had fewer, larger patches of receptors, echoing the simplified synaptic folds seen in a human patient with a CCT3 mutation.
Broken Inner Tracks and Slowed Cargo Traffic
Because TRiC helps fold actin and tubulin, the authors looked closely at the inner framework of motor axons. They found that overall levels of tubulin were sharply reduced in mutants, and the number of normal microtubules—the hollow tubes that act as tracks for transport—dropped dramatically in both central and peripheral nerves. Chemical “tags” on tubulin that are important for stable, transport-ready microtubules were also severely altered. Using live imaging of glowing mitochondria and endosomes inside motor axons, they watched how these cargoes moved. In healthy fish, organelles traveled swiftly and directionally along the axon. In mutants, many organelles stalled or merely wobbled back and forth, consistent with damaged tracks and disrupted transport.
One Core Problem Behind Many Symptoms
Taken together, the findings show that Cct3 is essential for several interconnected aspects of nerve health: it supports survival of myelin-forming support cells, enables proper myelin wrapping, and allows maturing neuromuscular junctions to become fine-tuned. All of these roles can be traced back to its job in folding and maintaining the proteins that build the cytoskeleton and the microtubule tracks used for axonal transport. When those tracks fail, key signals and materials cannot reach the right places along the nerve, likely undermining both communication with myelin-forming cells and the shaping of nerve–muscle contacts. This work suggests that impaired axonal transport may be the common thread linking the diverse brain and nerve problems seen in TRiC-related disorders.
Citation: Zhang, X., Zajt, K.K., Palaz, T. et al. The chaperonin TRiC component Cct3 is required for axonal transport, myelination, and neuromuscular junction refinement. Cell Death Dis 17, 221 (2026). https://doi.org/10.1038/s41419-026-08465-y
Keywords: myelination, axonal transport, neuromuscular junction, molecular chaperones, zebrafish model