Clear Sky Science · en
Impaired BDNF-TrkB trafficking and signalling in Down syndrome basal forebrain neurons
Why this brain study matters
People with Down syndrome are living longer than ever, but they also face a very high risk of developing Alzheimer’s-like dementia in midlife. This study looks deep inside individual brain cells to understand why certain neurons are so vulnerable. By tracking how these cells move and respond to a key “brain fertilizer” molecule, the researchers uncover a traffic jam inside neurons that could help explain memory loss and point toward new treatment strategies.
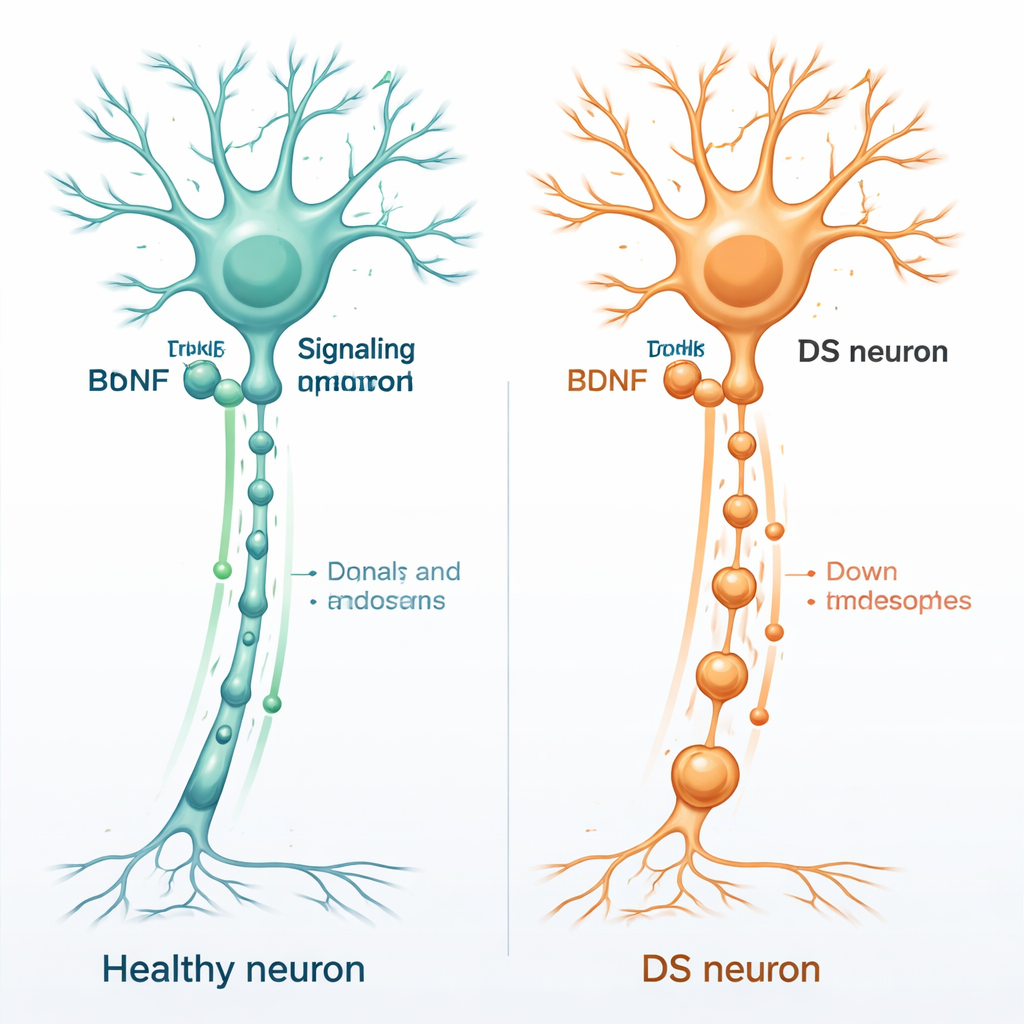
Brain fertilizer and nerve cell health
Healthy brain cells depend on a set of support molecules called neurotrophins, which act like fertilizer for neurons. One of the most important is brain-derived neurotrophic factor, or BDNF. BDNF binds to a docking protein on neurons called TrkB, usually at the far ends of nerve fibers. Once BDNF attaches, the whole BDNF–TrkB complex is pulled inside small membrane bubbles and carried back along the long nerve fiber to the cell body. These moving bubbles, known as signaling endosomes, tell the nucleus which genes to switch on to keep the neuron alive, connected, and adaptable.
When cellular shipping containers grow too big
The team focused on basal forebrain neurons, a group of cells that strongly supports memory and attention and is among the first to degenerate in both Down syndrome and Alzheimer’s disease. Using a mouse model of Down syndrome called Dp1Tyb, they stained neurons for an early endosome marker and compared them with normal neurons. The Down syndrome neurons contained far more early endosomes that were more than 50 percent larger than usual. These structures are controlled by Rab5, a molecular switch that helps form and sort these internal “shipping containers.” The researchers found that Rab5 was stuck in an overactive state in Down syndrome neurons, even before BDNF stimulation, suggesting that the endosomal system was already pushed into overdrive.
Traffic jams along the nerve fiber
Next, the scientists asked whether this distorted internal landscape interfered with the movement of BDNF signals. They grew neurons in microfluidic devices that separate the cell bodies from their axon terminals, allowing them to stimulate only the axon tips. By following harmless fluorescent tracers that share the same transport route as BDNF–TrkB complexes, they measured how fast the signaling endosomes traveled back toward the cell bodies. In normal neurons, adding BDNF made the endosomes move about 30 percent faster and pause less often, reflecting a robust response to growth signals. In Down syndrome neurons, however, BDNF failed to speed up transport or reduce pausing. A separate experiment that tracked real TrkB receptors confirmed that fewer of them successfully reached the cell body in Down syndrome cells.
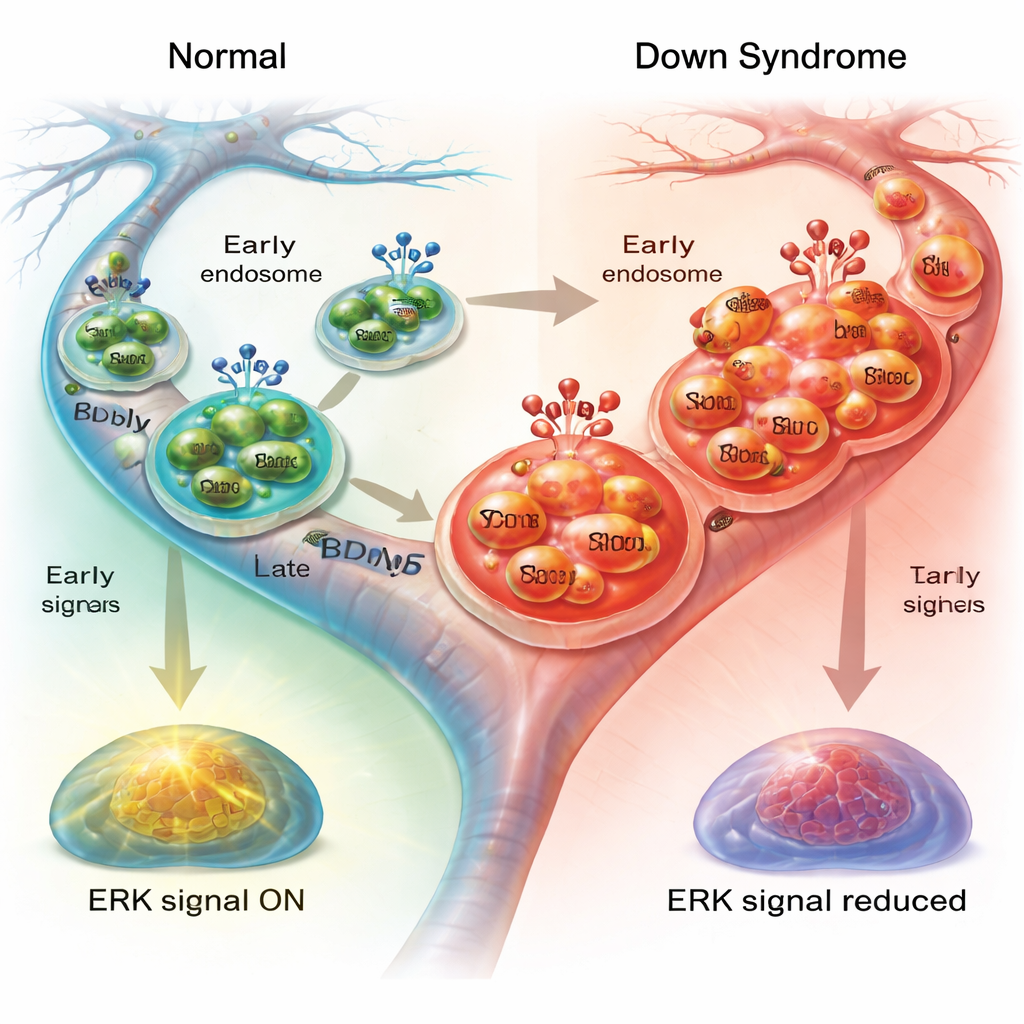
Blunted downstream signals inside the cell
For these traveling bubbles to support neuron health, they must activate internal signaling pathways once they reach their destination. One key route involves ERK1/2, a pair of enzymes that relay BDNF’s message to the nucleus and help regulate the cell’s skeleton and transport machinery. When the researchers measured activated ERK1/2 after BDNF treatment, they found a clear rise in normal neurons but a much weaker response in Down syndrome neurons, especially in the axons. Blocking ERK1/2 in healthy neurons partially reproduced the sluggish transport seen in Down syndrome cells, slowing endosomes and making them pause more often. In contrast, ERK1/2 blockade had little additional effect in Down syndrome neurons, consistent with signaling that was already dampened.
What this means for brain aging in Down syndrome
Taken together, the findings suggest a self-reinforcing problem in vulnerable neurons: Rab5 is overactive, early endosomes swell, and BDNF–TrkB complexes become trapped in compartments that do not move efficiently. As a result, fewer growth signals reach the cell body, ERK1/2 signaling is weakened, and the machinery that powers long-distance transport is further compromised. Over years, this faulty communication system may contribute to the early and severe loss of basal forebrain neurons seen in people with Down syndrome–related Alzheimer’s disease. By highlighting this link between endosomal “traffic control” and growth-factor signaling, the study points toward new therapeutic ideas that aim to normalize Rab5 activity, restore endosome function, and strengthen BDNF responses to help protect these critical neurons.
Citation: Blackburn, E., Birsa, N., Lopes, A.T. et al. Impaired BDNF-TrkB trafficking and signalling in Down syndrome basal forebrain neurons. Cell Death Dis 17, 214 (2026). https://doi.org/10.1038/s41419-026-08464-z
Keywords: Down syndrome, Alzheimer’s disease, BDNF, axon transport, endosomes