Clear Sky Science · en
PDHA1–acetylation signaling suppresses cuproptosis to attenuate anti-androgen effect in prostate cancer
Why this research matters
Prostate cancer is one of the most common cancers in men, and many advanced cases are treated with drugs that shut down male hormone signals. A key drug, enzalutamide, often works at first but then stops helping as tumors become resistant. This study uncovers a surprising reason why some prostate cancers escape enzalutamide’s effects and shows how blocking a single metabolic pathway could re‑sensitize tumors, opening the door to more durable treatments.
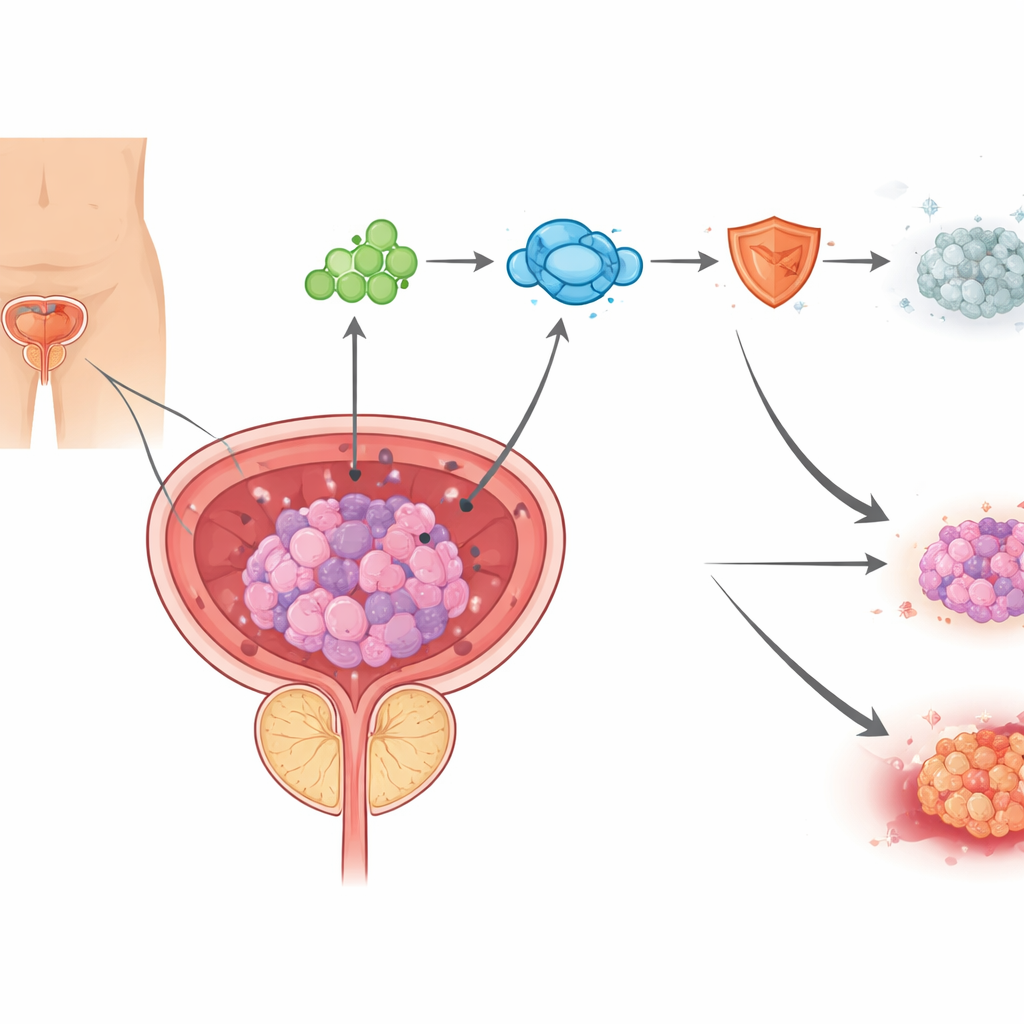
A new kind of cell death driven by copper
Cells can die in many different ways, and modern cancer research increasingly tries to turn these natural death programs back on in tumor cells. The authors focus on a newly described form of cell death called cuproptosis, which is triggered when copper builds up inside mitochondria, the cell’s power plants. In prostate cancer samples from patients treated with androgen‑blocking drugs, the team found signs of copper accumulation and mitochondrial damage, along with protein changes that are hallmarks of cuproptosis. In cell cultures, enzalutamide boosted copper levels and damaged mitochondria, and a copper‑binding compound could largely rescue cells, showing that enzalutamide can kill prostate cancer cells in part by pushing them into copper‑driven death.
How tumors blunt copper’s toxic punch
Yet not all cancer cells succumb. By mining several datasets of enzalutamide‑resistant prostate tumors and focusing on genes linked to cuproptosis, the researchers pinpointed one standout protein: PDHA1, a core part of the machinery that converts sugar‑derived fuel into acetyl‑CoA, a key cellular building block. PDHA1 levels were higher in resistant tumors, associated with worse survival and more aggressive disease, and were increased by enzalutamide itself. In lab experiments, dialing PDHA1 down made cancer cells far more sensitive to both enzalutamide and a direct cuproptosis‑inducing drug, while boosting PDHA1 made them harder to kill. In mice, tumors with reduced PDHA1 shrank much more under enzalutamide treatment, confirming that this enzyme helps tumors withstand therapy in living organisms.
A metabolic and epigenetic shield against treatment
The team then unraveled how PDHA1 builds this shield. Because PDHA1 feeds into acetyl‑CoA production, they tested whether it might change how DNA is packaged and read. When PDHA1 was high, acetyl‑CoA levels and chemical “acetyl” tags on histone proteins rose, especially on a mark associated with active genes. One key beneficiary was SLC7A11, a transporter that pulls cysteine into cells to make glutathione, a major antioxidant. With more PDHA1, cells made more glutathione; with less PDHA1, cysteine and glutathione dropped. Glutathione in turn bound and neutralized copper, reducing the stress that triggers cuproptosis. Restoring glutathione or SLC7A11 could undo the heightened copper stress seen when PDHA1 was blocked, showing that a PDHA1–acetyl‑CoA–histone–SLC7A11–glutathione chain lets cancer cells mop up copper and dodge death.
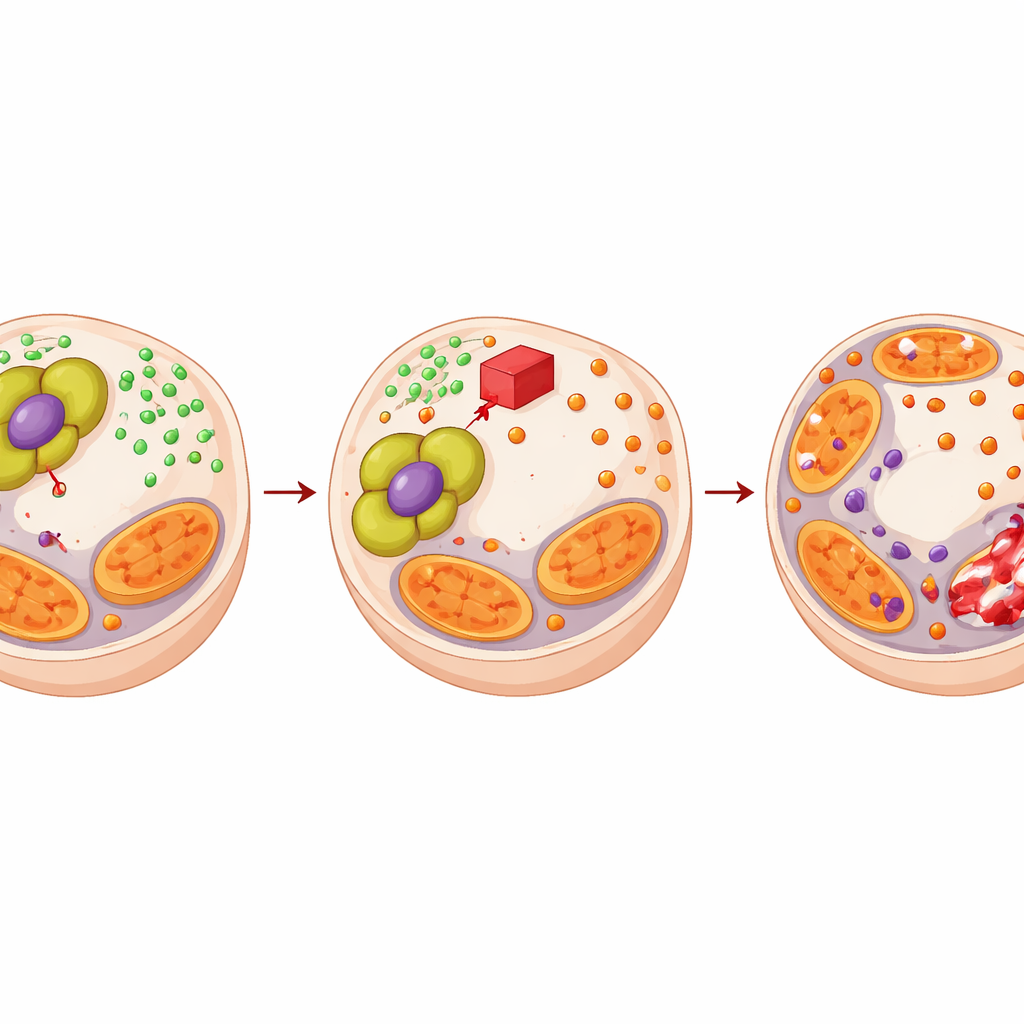
Turning a vulnerability into a therapeutic strategy
Because PDHA1 helps tumors resist enzalutamide by strengthening this copper‑buffering system, the authors asked whether a PDHA1‑targeting drug could flip the script. They used CPI‑613, a compound that disrupts the same metabolic complex. CPI‑613 alone increased copper stress and damaged mitochondrial proteins in prostate cancer cells but spared normal prostate cells. When combined with enzalutamide, CPI‑613 reduced cancer cell growth far more than either treatment alone, with mathematical analysis confirming true synergy. In mouse tumor models and patient‑derived tumor implants, the drug pair produced smaller tumors, more dead tissue, fewer dividing cells, and stronger signatures of copper‑driven cell death.
What this means for patients
Together, these findings show that some prostate cancers escape enzalutamide by rewiring their metabolism to generate more acetyl‑CoA, rewrite gene activity, and boost glutathione, which soaks up toxic copper and blocks cuproptosis. By inhibiting PDHA1, this protective circuit can be dismantled, allowing copper‑driven cell death to proceed and restoring the impact of hormone‑blocking therapy. While clinical trials are still needed, the work points to a concrete, testable strategy: pairing enzalutamide with PDHA1‑targeting drugs like CPI‑613 to overcome resistance in men with advanced, hard‑to‑treat prostate cancer.
Citation: Zhuang, R., Zhou, Q., Cheng, B. et al. PDHA1–acetylation signaling suppresses cuproptosis to attenuate anti-androgen effect in prostate cancer. Cell Death Dis 17, 243 (2026). https://doi.org/10.1038/s41419-026-08462-1
Keywords: prostate cancer, drug resistance, copper-induced cell death, cancer metabolism, epigenetic regulation