Clear Sky Science · en
Endothelial IRE1 signaling maintains blood–brain barrier integrity and limits neuroinflammation after traumatic brain injury
Why Protecting the Brain’s Inner Wall Matters
After a blow to the head, the immediate damage is only part of the story. In the hours and days that follow, the brain mounts a powerful stress and immune response that can quietly extend the injury. This study explores how a tiny stress-sensing system inside the cells lining brain blood vessels helps keep the brain’s inner wall—the blood–brain barrier—sealed and calm after traumatic brain injury. Understanding this hidden defense may point to new treatments that limit lasting disability after head trauma.
The Brain’s Gatekeepers Under Stress
The brain relies on a specialized barrier formed by endothelial cells, which line the inside of blood vessels and tightly control what can pass from blood into brain tissue. Traumatic brain injury (TBI) can disrupt this blood–brain barrier, allowing inflammatory molecules and immune cells to flood in and worsen damage. Inside these endothelial cells lives a quality-control system called the unfolded protein response, which helps cells cope with stress in a structure called the endoplasmic reticulum. One of its key components, known as IRE1, acts like a sensor and regulator when cells are under pressure. The researchers asked: does this endothelial stress sensor help defend the barrier after TBI, or does it make matters worse?
Switching Off a Cellular Sensor in Vessel Cells
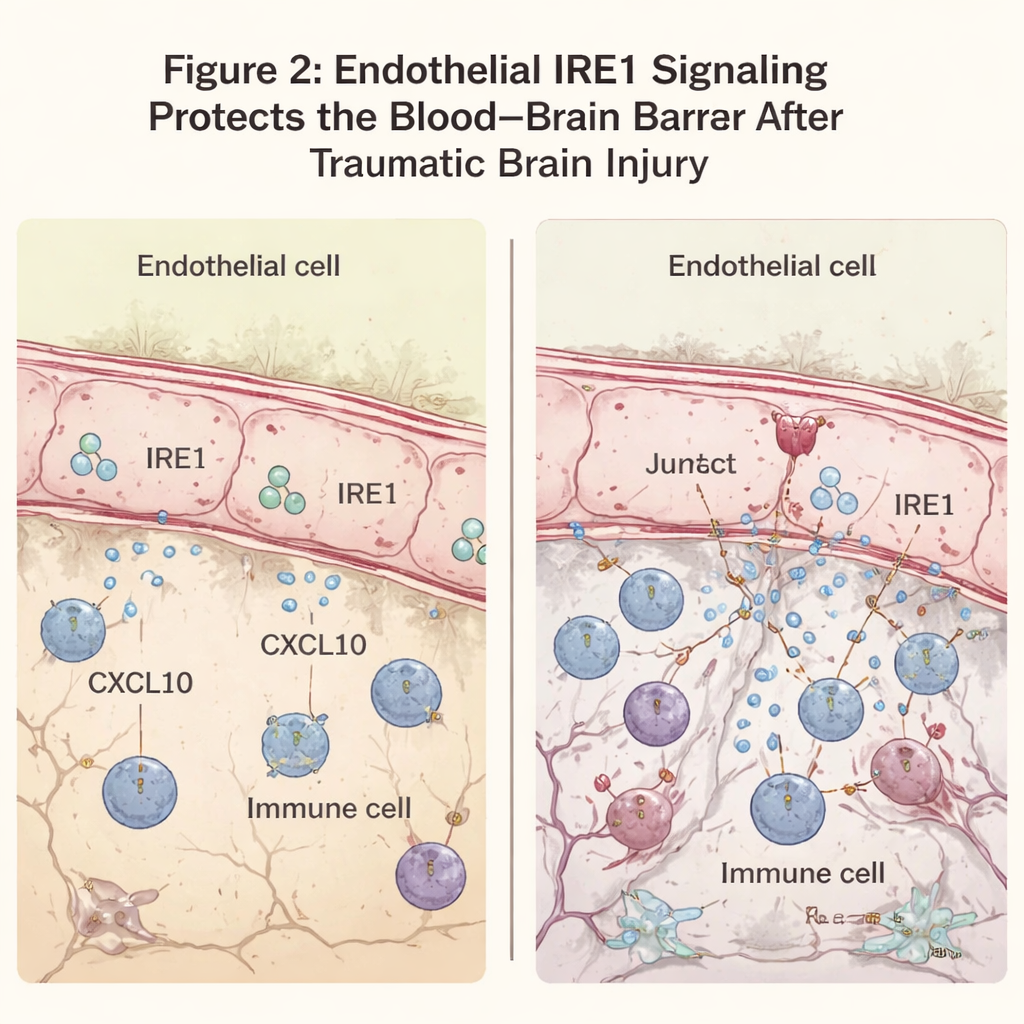
To find out, the team used genetically engineered mice in which IRE1 could be selectively turned off only in endothelial cells. They then created a controlled injury in the motor area of the cortex, mimicking aspects of human TBI, and compared these mice with normal littermates. They tested movement using tasks that measure balance and precise paw placement, and examined brain tissue with high-resolution microscopy and molecular techniques. In normal mice, IRE1 activity rose in blood vessel cells near the injury shortly after trauma, suggesting that endothelial cells quickly sense and respond to stress. When IRE1 was missing specifically in these cells, animals performed worse on motor tests, indicating poorer functional recovery.
Leaky Vessels, More Immune Cells, and Dying Neurons
The researchers next examined how well the blood–brain barrier held up. Normally, large blood proteins like antibodies stay inside vessels. After TBI, some leakage is expected near the lesion, but mice lacking endothelial IRE1 showed much more widespread seepage of these proteins into the brain. Microscopy revealed that a key junction molecule, VE-cadherin, which helps seal endothelial cells together, was reduced in the injured area when IRE1 was absent, even though the overall number of vessels looked similar. This leakiness was accompanied by a surge of immune cells entering the brain around the lesion and higher levels of inflammatory signals. In these same regions, neurons were more likely to show signs of degeneration and programmed cell death, and the zone of damaged tissue surrounding the core injury was noticeably larger.
A Stress Drug Calms the Signal Storm
To understand the molecular underpinnings, the team analyzed which genes were switched on after injury. In mice lacking endothelial IRE1, genes involved in antiviral and inflammatory responses were strongly increased. Standing out among them was CXCL10, a chemokine—a kind of molecular flare—that attracts immune cells and can weaken the barrier further. CXCL10 was particularly elevated in endothelial cells near the lesion when IRE1 was missing. In cultured brain endothelial cells exposed to an inflammatory trigger, reducing endoplasmic reticulum stress with a drug called TUDCA lowered both IRE1 activity and CXCL10 production. When given to injured mice, TUDCA reduced CXCL10 and immune cell markers in the damaged cortex and improved motor performance, suggesting that dampening this stress pathway can ease secondary injury.
What This Means for People With Head Injuries
Put simply, this work suggests that the stress sensor IRE1 in the cells lining brain blood vessels acts as a guardian after traumatic brain injury. When it is present and functioning, it helps keep the blood–brain barrier tight, reduces the release of chemical signals that summon immune cells, and limits the spread of inflammation and neuron loss around the lesion. When it is disabled, the barrier becomes leakier, immune cells pour in, and more brain tissue is damaged. Because blood vessel cells are relatively accessible to drugs circulating in the bloodstream, targeting their stress responses—with compounds like TUDCA or more precise future therapies—could become a practical strategy to reduce long-term harm after head trauma.
Citation: Fan, Q., Takarada-Iemata, M., Tanaka, T. et al. Endothelial IRE1 signaling maintains blood–brain barrier integrity and limits neuroinflammation after traumatic brain injury. Cell Death Dis 17, 210 (2026). https://doi.org/10.1038/s41419-026-08461-2
Keywords: traumatic brain injury, blood–brain barrier, endothelial cells, neuroinflammation, cellular stress