Clear Sky Science · en
CD36 enhances sensitivity of triple negative breast cancer cells to palmitate-induced ferroptosis
Why fat around tumors can be both friend and foe
Breast tumors grow in tissue that is naturally rich in fat, and cancer cells are known to “feed” on nearby fatty acids. This study explores a surprising twist: under the right conditions, one of the most common fats in the body, palmitic acid, can actually help kill an especially aggressive form of breast cancer by triggering a specific kind of cell death. Understanding how this happens could point to new treatments for patients who currently have few options.
A hard-to-treat breast cancer under the microscope
Triple-negative breast cancer (TNBC) lacks the hormone and growth-factor receptors that many modern drugs target, making it one of the most difficult breast cancer types to treat. These tumors live in close contact with fat cells, which release large amounts of fatty acids, including the saturated fat palmitic acid. The researchers wanted to know whether palmitic acid simply fuels tumor growth, or whether it might also create a weakness that could be exploited to kill cancer cells.
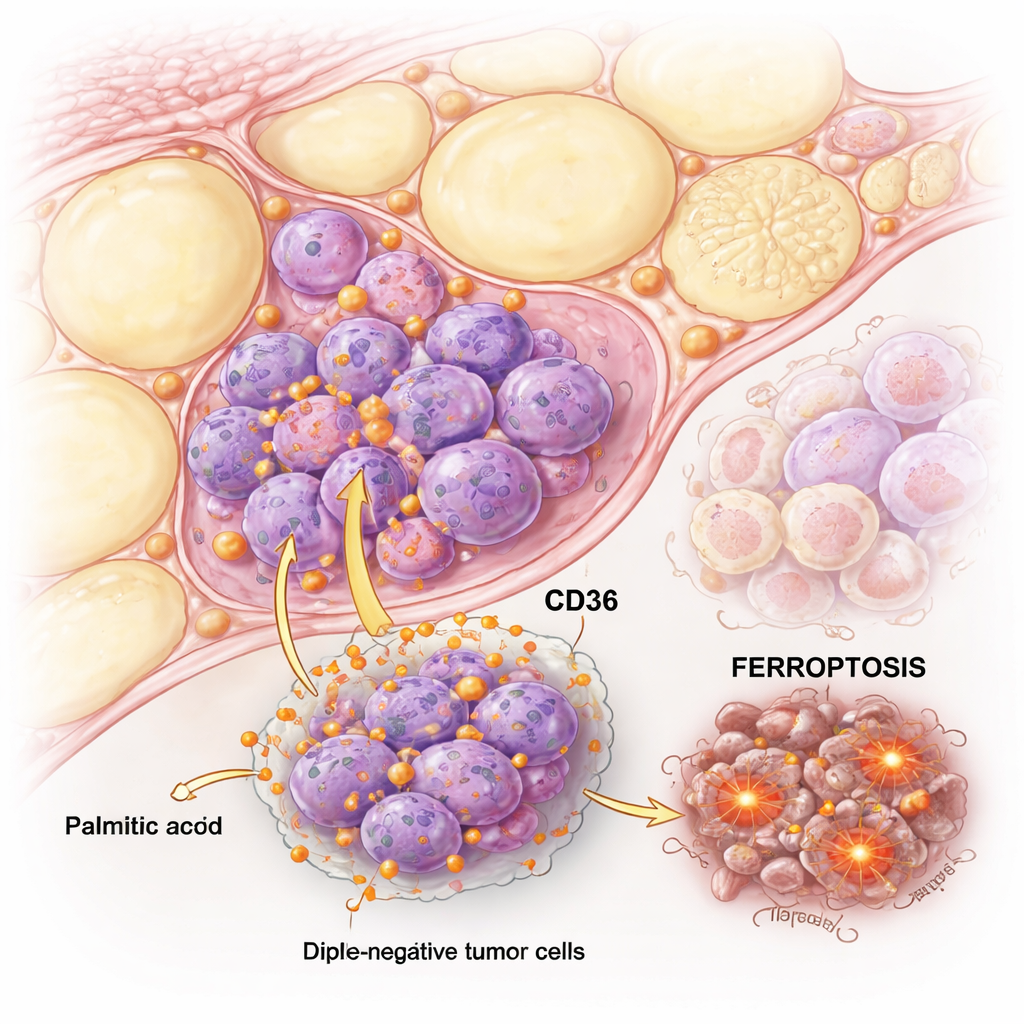
When palmitic acid pushes cancer cells over the edge
The team compared TNBC cells with hormone-sensitive “luminal” breast cancer cells and exposed them to increasing amounts of palmitic acid. Both types of cells could be harmed by high doses, but TNBC cells were far more sensitive. Detailed tests showed that in luminal cells palmitic acid mainly triggered classical apoptosis, a tidy form of cell death. In TNBC cells, however, palmitic acid set off both apoptosis and another, less familiar pathway called ferroptosis, which depends on iron and the destruction of cell membrane fats.
A fatty acid doorway called CD36
To understand why TNBC cells reacted so differently, the researchers focused on CD36, a protein on the cell surface that acts like a doorway for long-chain fatty acids. TNBC cells naturally carried much more CD36 than luminal cells, and exposure to palmitic acid pushed CD36 levels even higher. This meant TNBC cells pulled in more palmitic acid, accumulated more damaged lipids, produced more reactive oxygen in their mitochondria, and built up more free iron inside the cell—all key signals of ferroptosis. When CD36 was blocked with a drug, or reduced with genetic tools, palmitic-acid–induced ferroptosis dropped sharply.
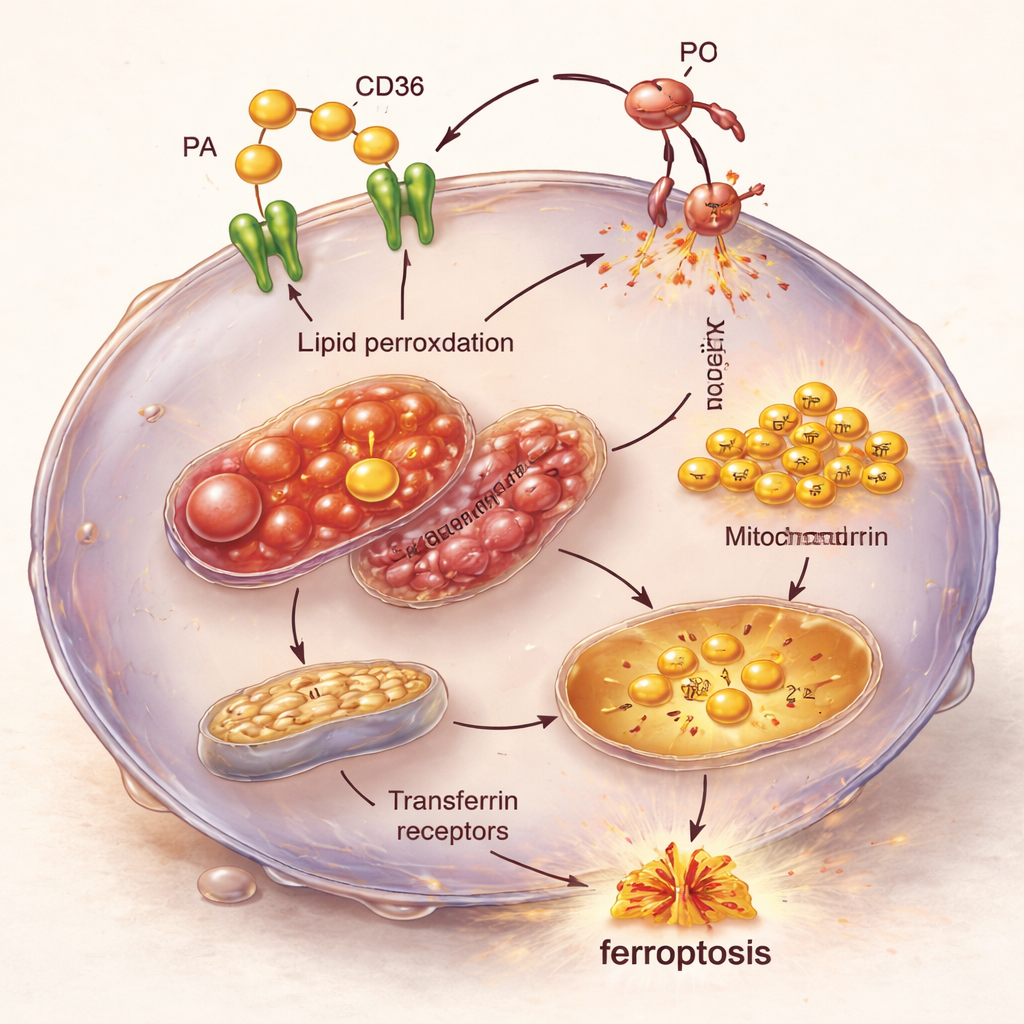
Zooming in on the chain reaction inside the cell
Microscopy and gene-activity analyses revealed the internal chain reaction behind this vulnerability. TNBC cells overloaded with palmitic acid and CD36 showed swollen, structurally damaged mitochondria and rising levels of iron brought in via transferrin receptors. At the same time, genes that encourage ferroptosis were switched on, while genes that normally protect against this form of cell death were dialed down. The result is a perfect storm: excess fat coming in, more iron available to drive chemical reactions, and weakened defenses against lipid damage, together pushing TNBC cells into ferroptosis.
Which patients might benefit the most?
Cancer is not uniform, even within TNBC. Using tumor samples grown in mice and large patient databases, the authors found that CD36 was especially high in a particular TNBC subtype called luminal androgen receptor (LAR), which had already been noted as more sensitive to ferroptosis. Other TNBC subtypes with high iron uptake also tended to show higher CD36 levels. This pattern suggests that CD36 could serve as a marker to identify patients whose tumors are naturally primed for ferroptosis-based therapies.
Turning a common fat into a therapeutic ally
In plain terms, this work shows that an abundant dietary and body fat, palmitic acid, can help kill certain triple-negative breast cancer cells when they express high levels of the fat transporter CD36. By driving fat overload, iron buildup, and damage to cell membranes, CD36 makes these cancer cells more likely to undergo ferroptosis, a destructive fate they cannot escape. If future treatments can safely boost this pathway—or combine it with drugs that further weaken the cells’ defenses—clinicians may be able to turn a metabolic weakness into a new, more precise way to attack some of the most aggressive breast tumors.
Citation: Closset, L., Foy, JP., Louadj, L. et al. CD36 enhances sensitivity of triple negative breast cancer cells to palmitate-induced ferroptosis. Cell Death Dis 17, 219 (2026). https://doi.org/10.1038/s41419-026-08460-3
Keywords: triple-negative breast cancer, ferroptosis, CD36, palmitic acid, tumor metabolism