Clear Sky Science · en
USP30-mediated Deubiquitination of Hexokinase 2 controls the metabolic fate of glucose and tumor progression
How Cancer Cells Rewire Their Sugar Use
Cancer cells are famous for their “sweet tooth”: they burn sugar in unusual ways to fuel rapid growth. This study uncovers a new switch, a protein called USP30, that helps tumor cells decide how to use glucose. By fine-tuning another protein, hexokinase 2 (HK2), USP30 can push cancer cells to burn more sugar and grow faster, pointing to a fresh target for future anti-cancer drugs. 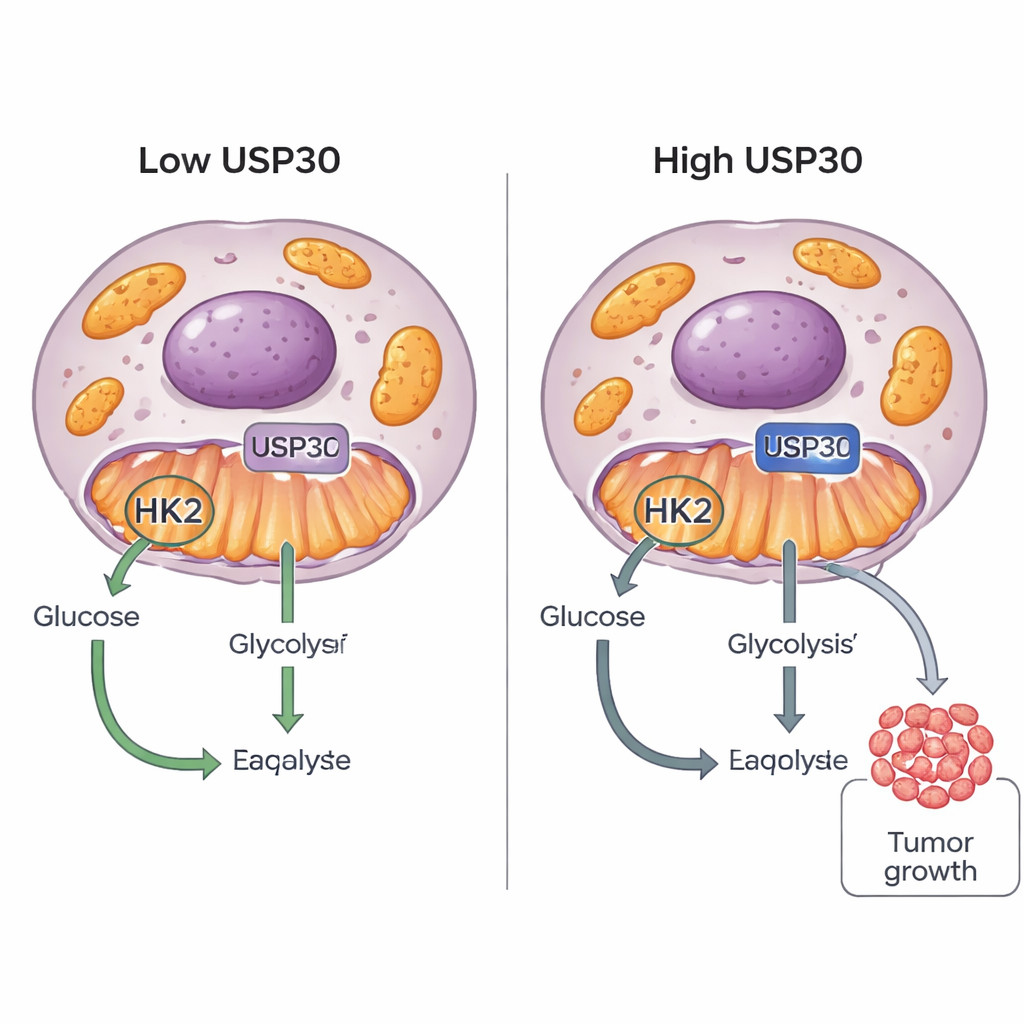
A Sugar-Hungry Lifestyle Inside Tumors
Most healthy cells extract energy from nutrients efficiently, but many cancer cells favor a quick-and-dirty route called aerobic glycolysis, also known as the Warburg effect. They pull in large amounts of glucose and rapidly convert it to lactate, even when oxygen is plentiful. This strategy does more than just make energy: it supplies building blocks for DNA, fats, and proteins, helps tumors survive stress, and can even weaken immune attacks. At the gateway to this pathway sits hexokinase, an enzyme that tags incoming glucose with a phosphate group, committing it to further breakdown and growth-supporting chemistry.
A Mitochondrial Enzyme Steps Into the Spotlight
USP30 is an enzyme perched on the outer surface of mitochondria, the cell’s power plants. It belongs to a family of “deubiquitinases” that remove tiny protein tags called ubiquitin from other proteins, often changing their stability, location, or activity. USP30 was already known for roles in brain cells and in controlling mitochondrial quality, but its impact on cancer was unclear. By mining large cancer gene databases, the researchers noticed that tumors with higher USP30 levels tended to show stronger glucose-burning signatures and weaker fat-burning signatures, hinting that USP30 might be helping cancers shift toward a sugar-fueled lifestyle.
Connecting USP30 to the Cell’s Sugar Gatekeepers
To probe this link, the team lowered or removed USP30 in several cancer cell lines and measured how they processed energy. Using instruments that track acid production and oxygen use in real time, they found that loss of USP30 sharply reduced both glycolysis and mitochondrial respiration. Lactate production and glucose consumption dropped, showing that the cells’ sugar-burning engine had slowed. A battery of mass spectrometry experiments then revealed that USP30 physically interacts with several enzymes tied to glucose metabolism, especially the hexokinases HK1 and HK2. Follow-up tests showed that this interaction depends on USP30’s catalytic activity and occurs directly, not just through indirect partners.
A Precise Molecular Handle on Hexokinase 2
Diving deeper, the researchers found that USP30 removes specific types of ubiquitin chains—so-called atypical linkages—from HK1 and HK2. For HK2, this editing occurs at a single crucial amino acid, lysine 144 (K144). When K144 is mutated so it can no longer carry ubiquitin, HK2 becomes more stable, binds more strongly to a channel protein called VDAC1 on mitochondria, and shows higher enzyme activity. Cells engineered to carry this K144 change sent more HK2 to mitochondria, burned more glucose, released more lactate, and displayed faster growth and movement in lab dishes. In mice, tumors grown from cells with the K144 mutant HK2 grew larger and faster than those with normal HK2, underscoring the power of this tiny molecular switch. 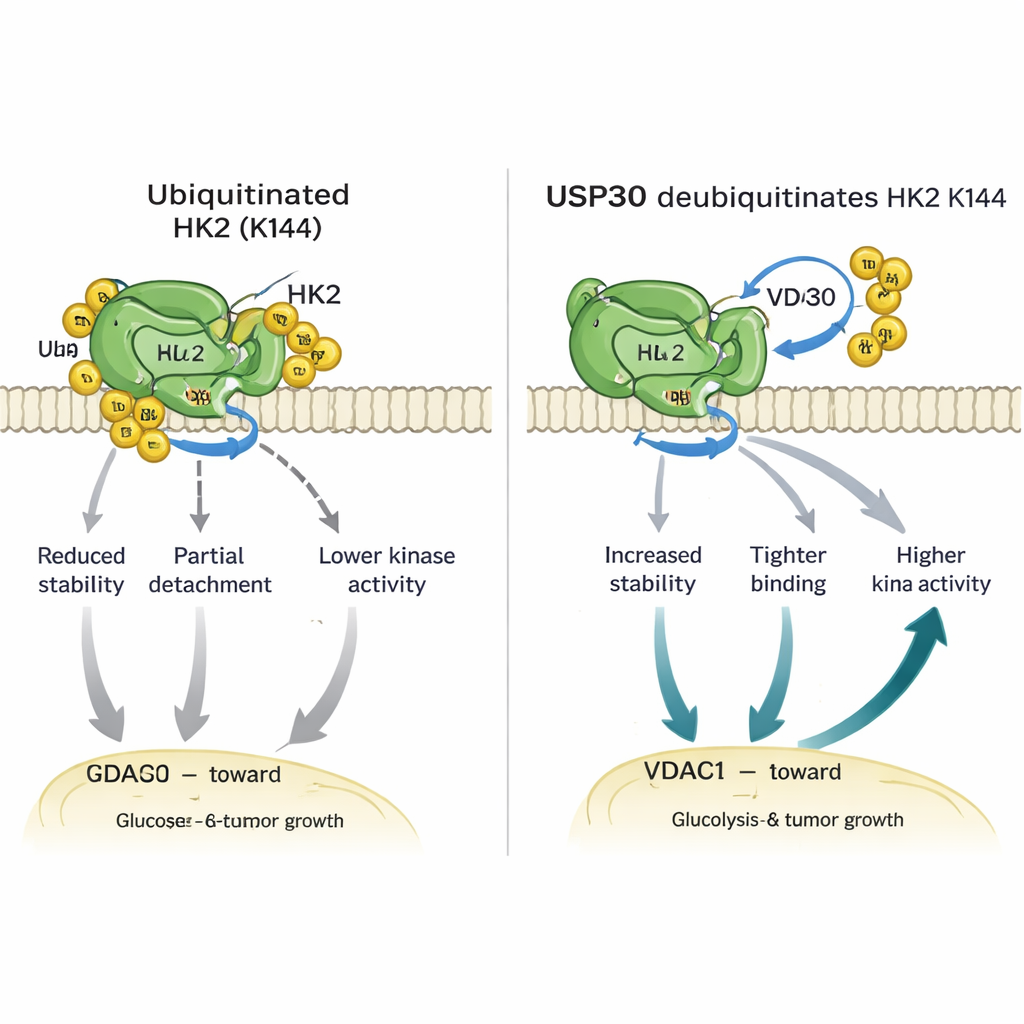
Turning a Metabolic Switch Into a Growth Advantage
These results map out a clear chain of events: USP30 binds to HK2, strips ubiquitin tags from K144, and in doing so keeps HK2 active and anchored on mitochondria. This boosts glycolysis, feeds tumor cell division and migration, and ultimately accelerates tumor growth. When USP30 is missing or inactive, HK2 becomes less stable and less effective, and cancer cells lose some of their metabolic edge. From a lay perspective, USP30 acts like a mechanic that keeps the cancer cell’s sugar engine tuned and bolted in place—remove the mechanic, and the engine sputters.
What This Means for Future Cancer Treatments
To a non-specialist, the key takeaway is that cancer cells rely on finely tuned control over how they burn sugar, and USP30 is a newly discovered knob on that control panel. By stabilizing HK2 at one specific site, USP30 helps tumors run their glucose-hungry metabolism and grow more aggressively. Drugs that block USP30, or that disrupt its grip on HK2 at lysine 144, could weaken tumors by starving their sugar engine without necessarily harming normal cells to the same extent. This work therefore adds an important piece to the puzzle of how cancer rewires metabolism and suggests a promising new angle for targeted therapies.
Citation: Haowei, Z., Li, X., Liao, W. et al. USP30-mediated Deubiquitination of Hexokinase 2 controls the metabolic fate of glucose and tumor progression. Cell Death Dis 17, 225 (2026). https://doi.org/10.1038/s41419-026-08459-w
Keywords: cancer metabolism, glycolysis, hexokinase 2, USP30, Warburg effect